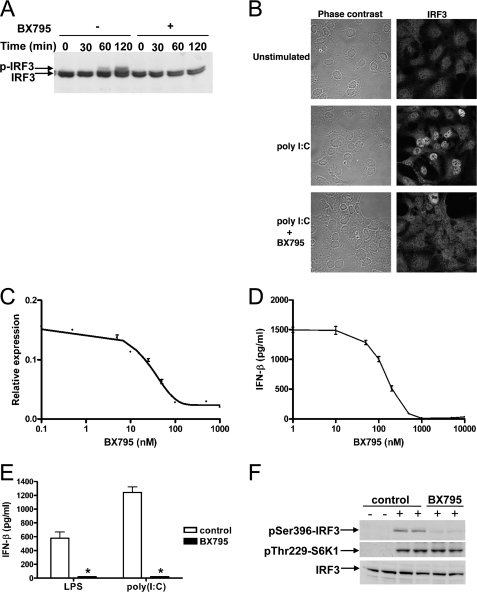
FIGURE 1.
BX795 blocks the phosphorylation, nuclear translocation, and transcriptional activity of IRF3 and production of interferon β in response to TLR3 and TLR4 agonists. A, HEK293-TLR3 cells were incubated for 60 min without (–) or with (+)1 μm BX795 and subsequently stimulated with 50 μg/ml poly(I:C) for the times indicated. Cell extract protein (40 μg) was subjected to SDS-PAGE and immunoblotted for IRF3. B, HEK293-TLR3 cells were stimulated for 2 h with poly(I:C) as in A. The cells were fixed, stained for IRF3, and visualized by confocal microscopy. C, HEK293-TLR3 cells were co-transfected with DNA encoding an IRF3 luciferase reporter construct and pTK-Renilla luciferase plasmid DNA. 24 h post-transfection cells were incubated for 1 h with varying concentrations of BX795 before stimulation for 6 h with 50μg/ml poly(I:C). Luciferase activity was measured and normalized to Renilla luciferase activity (mean ± S.E., n = 4). D, RAW264.7 cells were incubated for 1 h at various concentrations of BX795 and then stimulated for 6 h with 100 ng/ml LPS. The concentration of IFN-β released into the culture medium was measured by enzyme-linked immunosorbent assay. (mean ± S.E., n = 4). E, the experiment was carried out as in D except that cells were incubated without (white bars) or with (black bars)1 μm BX795 and stimulated with either 100 ng/ml LPS or 10 μg/ml of poly(I:C). (mean ± S.E., n = 3; *, p < 0.01). F, the effects of BX795 on TLR signaling do not result from inhibition of PDK1. RAW264.7 macrophages were treated for 30 min without (control) or with 1 μm BX795 then stimulated for 45 min without (–) or with (+) 100 ng/ml LPS. Cell extracts (40 μg protein) were immunoblotted with antibodies recognizing IRF3, IRF3 phosphorylated at Ser-396, and S6 kinase 1 (S6K1) phosphorylated at Thr-229.