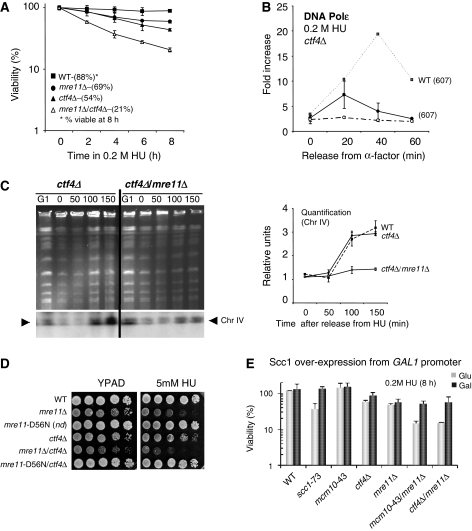
Figure 7.
The MRX complex functions on a pathway parallel to SCC factors to maintain forks during replication stress. (A) Cell viability was monitored as described earlier in Figure 5B for the following strains: wild type (JC467), mre11Δ (JC371), ctf4Δ (JC648), mre11Δ/ctf4Δ(JC662). (B) ChIP using αMyc (9E10) was performed on Myc-Polɛ in wild-type (JC285) and ctf4Δ (JC670) cells as described in Figure 2. (C) Ethidium bromide stained PFGE for samples arrested in G1 with α-factor for 90 min (G1) before treatment with 0.2 M HU for 180 min (0). HU was removed and cells were washed and released into YPAD media for 50, 100 and 150 min. Southern blot analysis was performed to monitor ChrIV replication (lower panel) with quantification of triplicate experiments for Chr IV replication shown in relative units compared with the signal in G1 using Quantity One (Biorad) strains used are ctf4Δ (JC648) and ctf4Δ/mre11Δ (JC662) with all experiments perform at the same time as Figure 3C. (D) Drop assays on YPAD±5 mM HU were performed from exponentially growing cultures with 1:10 serial dilutions at 30°C comparing wild type (JC467), mre11Δ (JC371), mre11D56N(JC606), ctf4Δ (JC648), mre11Δ/ctf4Δ (JC662) and mre11D56N/ctf4Δ (JC647). (E) Cell viability was monitored as colony outgrowth on SCC1overexpression induced (gal) or repressed (glu) from asynchronous cultures grown at permissive temperature (25°C) before transient exposure to 0.2 M HU at 30°C for 8 h. Galactose-induced SCC1 overexpression and glucose repressed SCC1 started 90 min before HU exposure and was maintained throughout all the experiments. The percentage survival is normalized to the cellular viability at time 0 for the following strains: wild type (JC784), scc1-73 (JC786), mcm10-43 (JC787), ctf4Δ (JC795), mre11Δ (JC801), mre11Δ/mcm10-43 (JC803), mre11Δ/ctf4Δ (JC797).