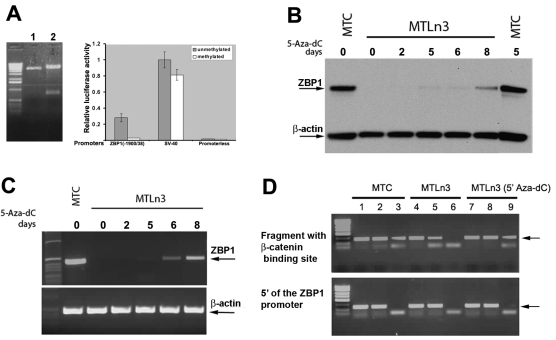
Fig. 3.
Repression of ZBP1 expression in metastatic cells results from promoter methylation, which prevents the binding of the promoter to β-catenin. (A) In vitro methylation of the ZBP1 promoter leads to repression of its transcriptional activity. Three luciferase constructs were in vitro methylated by SssI methyltransferase. Left: completion of in vitro methylation was evaluated by restriction digestion of methylated (lane 1) or unmethylated (lane 2) constructs with EagI, which recognizes unmethylated CGGCCG sequences. Right: after transfecting the reporters into mTC cells, luciferase activity was measured and normalized. Relative luciferase activity is represented as a percentage of the unmethylated pSV-40 construct. Values are the average of two independent assays ± s.e.m. (B) Western blot analysis of ZBP1 and β-actin protein expression in MTC and MTLn3 cells treated or non-treated with 5-Aza-dC. The arrows indicate the positions of the detected proteins. (C) Analyses of ZBP1 and β-actin mRNA expression in the same cell cultures as shown in B. The arrows indicate the positions of the ZBP1 and β-actin transcripts. (D) MTC cells and MTLn3 cells incubated with or without 5-Aza-dC were subjected to chIP using antibodies against acetylated histone and β-catenin. Total nuclear lysates were used as an input control to purified chromatin DNA. Two primer pairs, one amplifying a region of the ZBP1 promoter containing a putative CTTTG-TC binding site for β-catenin (upper) and the other amplifying a region 2-kb upstream of the ZBP1 transcription site (lower), were used in the experiments. Lanes 1, 4 and 7: inputs of MTC cells, MTLn3 cells and MTLn3 cells treated with 5-Aza-dC. Lanes 2, 5 and 8: precipitates with antibodies against acetylated histone. Lanes 3, 6 and 9: precipitates with antibodies against β-catenin. Anti-β-catenin antibody specifically pulled down the ZBP1 promoter in MTC and 5-Aza-dC-treated MTLn3 cells, but not in parental MTLn3 cells because of the promoter methylation.