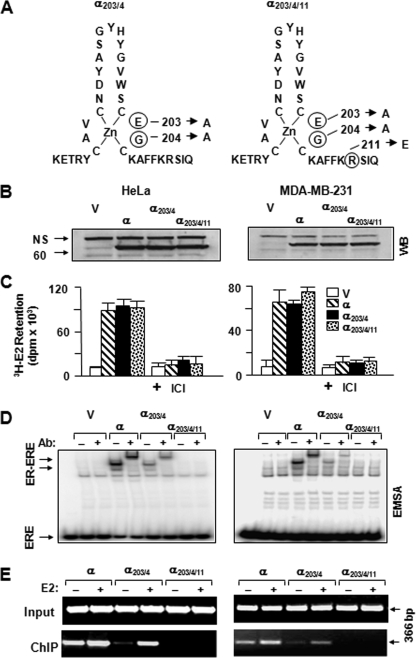
FIGURE 1.
Generation of ERE-binding defective ERα variants. A, ERα203/4 was engineered by changing glutamic acid and glycine at positions 203 and 204 of the DNA-binding helix of the ERα to alanine residues, whereas ERα203/4/11 contains an additional change that replaces arginine at position 211 with glutamic acid. B, synthesis of ERα species. HeLa (left panels) or MDA-MB-231 cells (right panels) were transiently transfected with an expression vector bearing none (V) or an ERα cDNA. Cell extracts (10 μg) were subjected to WB using a horseradish peroxidase-conjugated monoclonal FLAG antibody. Molecular mass in kDa is indicated. NS, nonspecific. C, in situ E2 binding assay. Twenty four hours after transient transfections with an expression vector bearing none (V) or an ERα cDNA, HeLa or MDA-MB-231 cells were incubated in medium containing 10-7 m of [3H]E2 for 1 h. Medium containing the radioactive [3H]E2 was then removed. Cells were extensively washed with fresh medium and dislodged. Radioactivity retained in cells was quantified by scintillation counting. The specific retention of [3H]E2 was assessed by the co-incubation of cells with 10-6 m ICI. The graph represents the mean ± S.E. of three independent experiments performed in duplicate. D, cell extracts (10 μg) of transfected cells were also subjected to EMSA without (-) or with (+) a FLAG antibody (Ab). ERE specifies unbound and ER-ERE denotes ER-bound radiolabeled ERE. A representative result from three independent experiments of WB or EMSA is shown. E, ChIP assay. HeLa or MDA-MB-231 cells were transiently transfected with an expression vector bearing an ERα cDNA together with a reporter plasmid bearing the simple TATA-box promoter with one ERE. Cells were treated without (-) or with (+)10-7 m E2 for 1 h prior to ChIP using FLAG antibody-conjugated agarose beads. Sizes of DNA fragments in base pair are indicated.