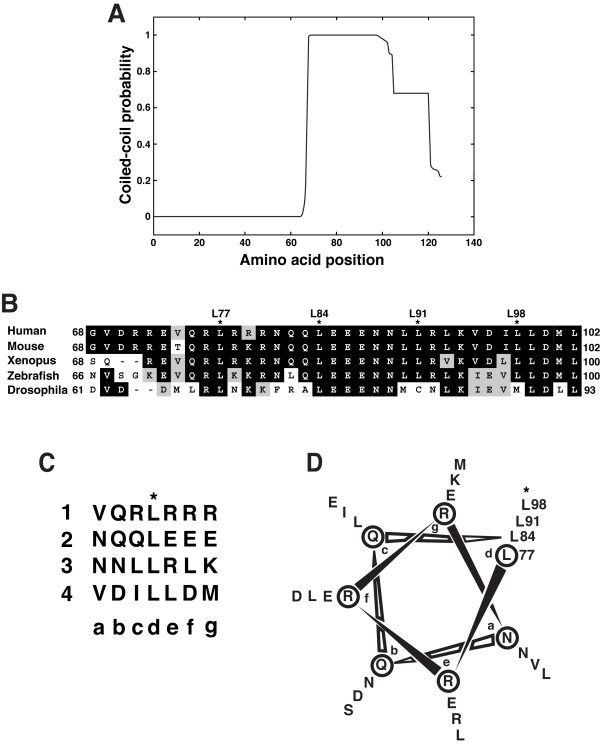
Figure 3.
The C-terminal region of Cby harbors a putative coiled-coil motif. (A) The human Cby protein sequence (126 amino acid residues) was analyzed by the COILS program at a window size of 21 residues. Note that high coiled-coil probabilities exceeding 0.9 were evident from amino acid 68 to 102. (B) Sequence alignment of the Cby coiled-coil domain across species. Identical and similar residues are highlighted in black and gray, respectively. Four leucines, indicated by asterisks, appear at every seventh position, indicative of a leucine-zipper motif. (C) The putative coiled-coil motif of human Cby is depicted as heptad repeats of seven amino acids. The letters a through g designate the positions of residues within the heptad with hydrophobic residues generally found at the a and d positions. The four consecutive heptads are numbered 1 through 4. Note that the d positions are occupied by leucine residues.(D) Helical wheel diagram of the coiled-coil domain of human Cby (amino acids 77–98).