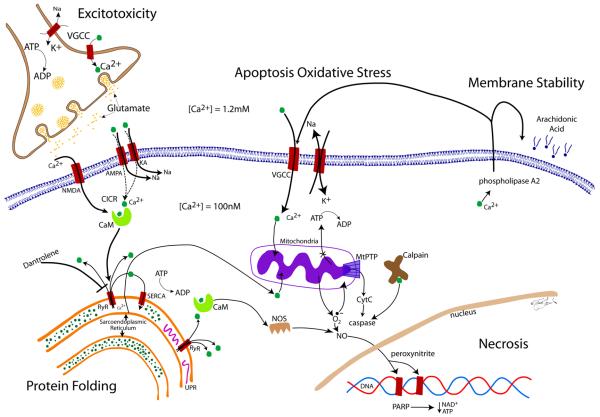
FIGURE. The role of calcium in cellular injury.
Under normal conditions, there is a 10,000-fold Ca2+-gradient between the intra- and extracellular space. When cellular oxygen levels reach below-threshold levels, the Na+-K+-pump, which consumes 70% of neuronal ATP, fails, leading to depolarization of the membrane and influx of Ca2+ through VGCC and glutamate release into the synaptic cleft. Glutamate binds to NMDA, AMPA and KA receptors, leading to direct and indirect influx of Ca2+ into the cell. Elevated levels of intracellular Ca2+ lead to further calcium-induced calcium release (CICR) via binding of Ca2+ to CaM. CICR is mediated by RyR binding and calcium release from the sarcoendoplasmatic reticulum, the largest store of intracellular Ca2+. Increases in intracellular Ca2+ cannot be compensated for by the energy-dependent SERCA pump, located in the membrane of the sarcoendoplasmatic reticulum. A reduction in endoplasmic Ca2+ results in misfolding of proteins, leading to UFR as a cellular stress response. Influx of Ca2+ into mitochondria leads to formation of oxygen radicals and energy failure through decreased production of ATP. Binding of Ca2+ to CaM also leads to activation of NOS and formation of NO. Oxygen radicals react with NO to form peroxinitrite, a highly reactive molecule that damages DNA and proteins. DNA cleavage activates the DNA-repair enzyme PARP, which requires ATP for its action. PARP-related energy depletion adds further to cellular stress causing necrosis. The increased (toxic) levels of intracellular Ca2+ lead to increased mitochondrial permeability via MtPTP. This causes mitochondria to become permeable to molecules smaller than 1.5 kDa, which increases the osmolar load inside the mitochondria, causing mitochondrial swelling and outer membrane rupture, releasing CyC. CyC activates pro-apoptotic factors (caspases) by reacting with Ca2+-activated calpain. Elevated intracellular Ca2+ activates phospholipase A2 which cleaves cell membrane phospholipids, releasing arachidonic acid, which in turn can be further activated to proinflammatory molecules, such as prostaglandins, leukotrienes, and thromboxanes (not shown). Dantrolene is neuroprotective by blocking ryanodine receptors at the sarcoendoplasmatic reticulum, and thereby inhibiting further increase in intracellular Ca2+-levels.