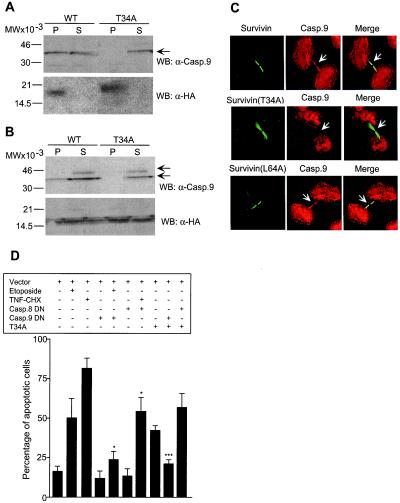
Figure 5.
Modulation of survivin-caspase-9 complex by survivin phosphorylation on Thr34. (A) Immunoprecipitation from nonadherent cells. HeLa cells transfected with HA-survivin (WT) or HA-survivin(T34A) (T34A) were harvested after mitotic shake off, immunoprecipitated with anti-HA followed by Western blotting (WB) with an antibody to caspase-9 (casp.9). (B) Immunoprecipitation from synchronized cells. HeLa cells were transfected with HA-survivin (WT) or HA-survivin(T34A) (T34A), immunoprecipitated with anti-HA 12-h after thymidine release, and analyzed for coassociated caspase-9 by Western blotting. Arrows, position of 46-kDa proform caspase-9 and ≈35-kDa active caspase-9. P, pellet; S, supernatant. (A and B) Blots were sequentially immunoblotted with anti-HA. (C) Mislocalization of caspase-9 from midbodies in survivin(T34A)-expressing cells. HeLa cells transfected with HA-survivin (Survivin), survivin(T34A), or survivin(L64A) were labeled for survivin (FITC, green) with a mAb to HA or mAb 8E2 (Bottom), and caspase-9 (Casp.9, TR, red), and analyzed by confocal microscopy. Image-merging analysis is shown on the right. Arrows, differential localization of wild-type survivin, survivin(T34A), or survivin(L64A) with caspase-9 at midbodies. Experiments were repeated at least four times with comparable results. (D) Apoptosis in survivin(T34A)-expressing cells is mediated by caspase-9. HeLa cells transfected with the various indicated combinations of GFP-constructs, with or without etoposide (10 μg/ml) or TNFα (TNF, 10 ng/ml) plus cycloheximide (CHX, 10 μg/ml), were morphologically scored for nuclear fragmentation by DAPI staining. DN, dominant negative. *, P < 0.05; ***, P < 0.0005. Data are the mean ± SEM of four independent experiments.