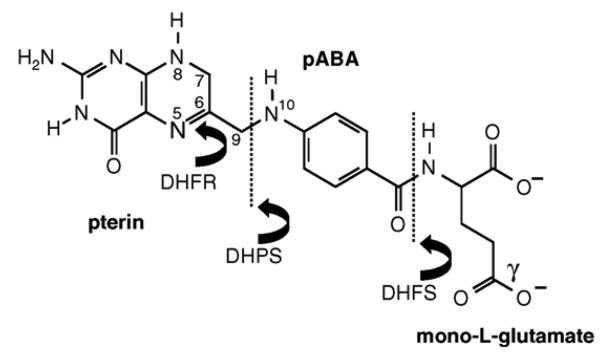
Fig. 1.
Structure of dihydrofolate (DHF) indicating its construction from the pterin, pABA and l-glutamate moieties. The enzyme DHPS links pABA to an activated pterin ring to give dihydropteroate, which is converted to dihydrofolate by addition of a single l-glutamate residue by DHFS. The 5,6 double bond in DHF is reduced by DHFR to give tetrahydrofolate (THF); all active folate cofactors are found in this fully reduced state. They are also subject to polyglutamation by the FPGS activity, whereby additional glutamate residues are linked via peptide bond formation at the gamma position. Important atomic positions on the dihydropteroate moiety are numbered. For enzyme abbreviations, see the legend to Fig. 2.