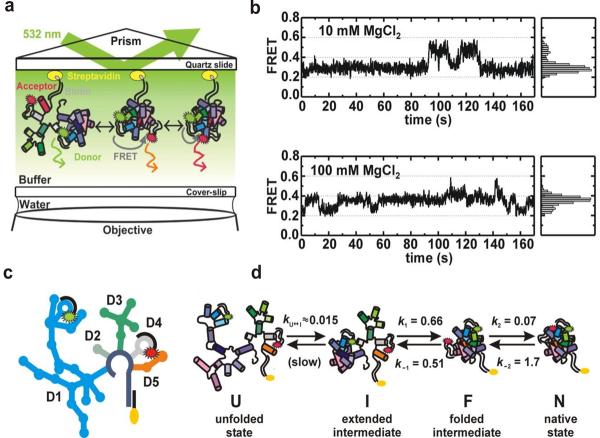
Fig. 3.
Single molecule FRET studies of group II intron ribozymes. (a) Experimental setup of the total internal reflection fluorescence spectroscopy (TIRF) based single molecule FRET. Fluorophore-labeled ribozyme construct is immobilized on a quartz slide through a biotin-streptavidin linkage. (b) Single molecule time trajectories and histograms at different Mg2+ concentrations showing the presence of three different FRET states (∼0.25, 0.4 and 0.6). (c) A schematic structure of the Cy3 and Cy5 fluorophore-labeled D135-L14 ribozyme containing five domains (D1, D2, D3, D4 and D5). (d) Folding pathway of the D135-L14 ribozyme consisting of four different states: the unfolded state (U), the extended-intermediate state (I), the folded-state (F), and the native state (N). Reprinted with permission from reference [34], M Steiner et. al., PNAS 105, 13853 (2008). Copyright (2008) National Academy of Sciences, U.S.A.