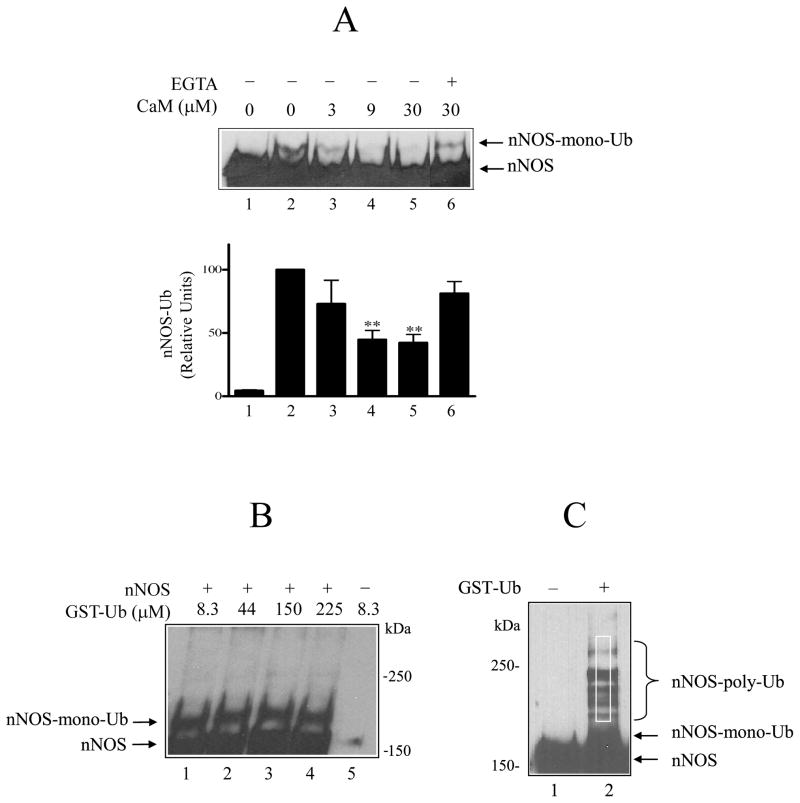
FIGURE 1.
Calmodulin inhibition of nNOS ubiquitination by the DE52-retained fraction of rabbit reticulocyte lysate. A, nNOS monoubiquitin conjugates (nNOS-mono-Ub) were detected by Western blot. nNOS was incubated for 1 h at 37 °C with a DE52-retained fraction of rabbit reticulocyte lysate, ATP, GST-ubiquitin and the indicated concentrations of calmodulin as described under Methods. Samples were Western blotted by probing with anti-nNOS. Lane 1, incubation time 0; lanes 2–6, incubation time 1 h. For bar graph, the relative amount of nNOS-Ub in replicate experiments as in A was determined by scanning and expressed as % of the 1 h control without calmodulin. The values are the mean ± S.E. (n=3). ** denotes significantly (p<0.01) lower nNOS-Ub conjugates relative to the 1 h control without CaM. B, the effect of increasing the ubiquitin concentration on the ubiquitination of nNOS. The studies were as in A, except that the indicated concentrations of GST-ubiquitin were used (lanes 1–4). Lane 5, nNOS was omitted. The data are representative of three identical experiments. C, nNOS polyubiquitin conjugates (nNOS-poly-Ub) produced by the purified ubiquitinating enzyme mixture without (lane 1) or with (lane 2) GST-ubiquitin. The white rectangle above the monoubiquitinated nNOS represents the region scanned to determine relative densities of polyubiquitination in experiments with the purified ubiquitination system.