Figure 5. Cooperation amongst intrinsically disordered and coiled-coil segments within IC promotes binding to LC8.
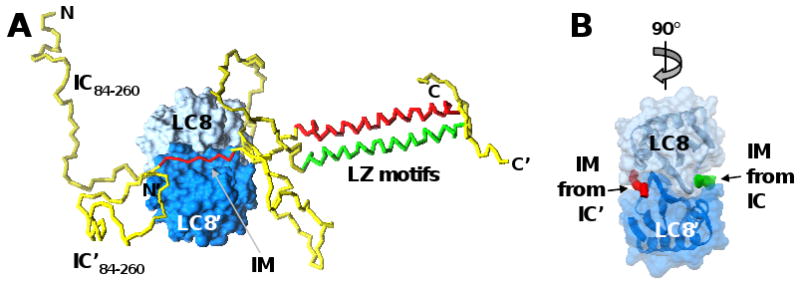
(A) A short interaction motif (IM) from two molecules of the intrinsically disordered protein, IC (residues 84-260 illustrated), adopt rigid, extended structure when bound on opposite faces of the folded, dimeric protein, LC8. While not directly involved in binding to LC8, two leucine-zipper (LZ) motif-containing segments of IC, that are unfolded and monomeric in the absence of LC8, form a coiled-coil dimer when the IM segments of IC bind to LC8. IC is illustrated as a yellow tube, with the IM segments and LZ motifs colored red or green, respectively, in the two molecules. The two subunits of the LC8 dimer are shown in surface representation in dark and light blue, respectively. (B) The LC8 dimer was rotated 90° relative to (A) and only the IM segments of the two IC molecules are illustrated as red and green tubes, respectively. [Modeled after Figure 2 in ref. 69, with permission from the author.]
