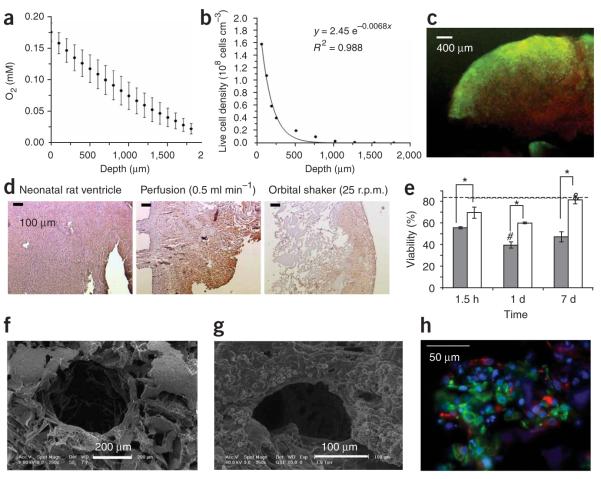
Figure 1.
Cultivation of cardiac tissue constructs. In (a–c) static and (d–h) perfusion culture. Neonatal rat cardiomyocytes after one pre-plating were seeded on collagen sponge scaffolds (~108 cells cm−3). (a) Owing to the high metabolic demand for oxygen and diffusion as the main mode of mass transfer within the interior of the scaffold dissolved oxygen concentration decreased linearly within the scaffold interior. (b) Cell density, as measured from histological cross-section, exhibited an exponential decay in the scaffold interior. (c) Live/dead staining indicated that most of the cells in the construct interior were dead (red). (d) Cardiac Troponin I staining of cardiomyocytes cultivated in collagen scaffolds in perfusion shows uniform and high cell density throughout the scaffold. (e) The viability of cells cultivated in perfusion (white bars) was significantly higher than those cultivated under static conditions (gray bars) at every time point tested. (f) A parallel channel array was laser-bored in poly(glycerol sebacate) scaffolds, seeded with cells and cultivated in perfusion in culture medium containing perfluorocarbon (PFC) oxygen carriers. (g) The channels remained open upon cultivation. (h) Double staining for cardiomyocytes (Troponin I, green) and fibroblasts (vimentin red) in constructs cultivated in the presence of PFC oxygen carriers on channeled PGS (poly (glycerol sebacate)) scaffolds. Nuceli are counterstained blue with 4,6-diamidino-2-phenylindole (DAPI).