Abstract
OBJECTIVE
Up to 30% of women with recent gestational diabetes mellitus (GDM) remain glucose intolerant after delivery. However, the rate of postpartum oral glucose tolerance tests (ppOGTTs) is low. Our aim in this study was to develop a model for risk assessment to target women with high risk for postpartum diabetes.
RESEARCH DESIGN AND METHODS
In 605 Caucasian women with GDM, antenatal obstetrical and glucose data and the glucose data of the ppOGTTs performed 13 weeks (median) after delivery were prospectively collected.
RESULTS
A total of 132 (21.8%) women had an abnormal ppOGTT (2.8% impaired fasting glucose, 13.6% impaired glucose tolerance, and 5.5% diabetes). Independent risk factors were BMI ≥30 kg/m2 (prevalence of abnormal ppOGTT 36.0 vs. 17.3%), gestational age at diagnosis <24 weeks (32.4 vs. 18.0%), 1-h antenatal value >200 mg/dl (11.1 mmol/l) (35.2 vs. 14.8%), and insulin therapy (30.3 vs. 14.5%). The prevalence of an abnormal ppOGTT was assessed according to the number of risk factors: 0, 9.2% (14 of 153); 1, 13.4% (25 of 186); 2, 28.5% (43 of 151); 3, 45.6% (26 of 57); and 4, 68.4% (13 of 19). Subjects were divided according to a significant increase of prevalence and risk for a ppOGTT: low risk (59.9% of subjects), <2 risk factors, 11.6%, odds ratio 1.3; intermediate risk, 2 risk factors, 28.5%, 4.0; and high risk, >2 risk factors, 51.3%, 10.5. The intermediate/high-risk group included 86.6% of those with diabetes and 67% of all those with abnormal ppOGTTs.
CONCLUSIONS
Women with ≥2 risk factors have a high risk for an abnormal ppOGTT, and 86% of postpartum diabetes is diagnosed within this group. Targeting women for ppOGTTs based on a risk assessment using available antenatal risk factors might reduce the number of missed cases of postpartum diabetes.
Gestational diabetes mellitus (GDM) implies a substantial risk of later diabetes. Diabetes rates are reported to be between 9 and 43% within 5–10 years after the index pregnancy (1–4), and the incidence of diabetes in women after GDM pregnancies appears to be increasing (2). There is evidence that in women with GDM an underlying preexisting β-cell defect is demasked by poor pancreatic β-cell compensation for physiological insulin resistance in pregnancy. The prevalence of diabetes within 1 year after delivery in women with recent GDM is reported to vary between 3 and 24% (3,4), depending on the population characteristics, timing of postpartum testing, and applied diagnostic criteria for diabetes outside pregnancy.
Recommendations of the 5th Workshop-Conference on GDM (5) are that women with GDM undergo postpartum glucose tolerance testing with an oral glucose tolerance test (OGTT) at 6–12 weeks, 1 year after delivery, and every 3 years thereafter. The rational for this recommendation is based on the potential to identify women with apparent diabetes as well as women with impaired glucose tolerance (IGT) in whom diabetes can be delayed or prevented by lifestyle intervention or moderate drug therapy (6).
In reality, the rates of postpartum glucose testing are low and dependent on the compliance of caregivers and patients (7–10). Testing is complicated by discontinuity of care after delivery and mothers underestimating their diabetes risk and stress to adapt to the challenge of caring for a young baby. More exact quantification of the individual risk for diabetes and targeting women at high risk may increase the compliance and efficacy not to miss women with postpartum persistent glucose intolerance. Several risk factors had been identified for an increased risk for persistent glucose intolerance in the 1st year after delivery (10–15). The aim of our study was to use the knowledge of risk factors to develop a model for risk stratification based on the combination of antenatal risk factors that might allow us to distinguish between women with high, intermediate, or low risk for postpartum diabetes within 1 year after GDM.
RESEARCH DESIGN AND METHODS
For this prospective multicenter study, data from Caucasian women whose GDM was diagnosed from 1 January 2000 through December 2005 from three different German institutions with diabetes and pregnancy specialist services were analyzed. Those services were 1) the Clinic of Diabetes at the Department of Obstetrics of the Vivantes Medical Center, Berlin, 2) the Clinic of the Department of Diabetology at the University Medical Center of Bonn, and 3) a private clinic that is run by two diabetologists in Kiel.
The women were selected from an ongoing database in which clinical, glycemic, and delivery data for all women were prospectively entered. Inclusion criteria for this study were 1) maternal glucose intolerance first diagnosed in pregnancy, 2) availability of clinical data regarding maternal characteristics, glycemic data, and neonatal parameters, and 3) a documented maternal postpartum OGTT (ppOGTT) within 1 year of delivery. The study protocol was approved by local ethics committees. Informed consent was obtained from the women at the time of ppOGTT.
Diabetes care and postpartum testing
Diabetes care was uniformly managed at all sites by a standardized management protocol based on German guidelines for diagnosis and therapy of GDM derived from the recommendations of the 5th Workshop-Conference on GDM (5). Diagnosis of glucose intolerance was established by a 2-h 75-g OGTT performed between 24 and 28 weeks of gestation. In women with a high risk for diabetes, testing was done earlier. According to the policy in Germany at the time of the study, GDM testing was focused on women with risk factors for GDM. After GDM diagnosis, women were given dietary instructions and advised to perform blood glucose monitoring with glucose profiles and preprandial and 2-h postprandial measurements twice a week or daily using memory-based glucometers if insulin therapy was required. Insulin therapy was initiated when either the average glucose value of a profile exceeded 100 mg/dl (5.5 mmol/l) or a fasting glucose value >95 mg/dl (5.3 mmol/l) and/or 2-h postprandial value >120 mg/dl (6.6 mmol/l) was seen after a 2-week trial of diet (5).
In women with GDM requiring insulin, at least one glucose profile was performed before hospital discharge, and patients with fasting capillary whole blood glucose >110 mg/dl (6.1 mmol/l) and 2-h postprandial glucose >160 mg/dl (8.8 mmol/l) were asked to continue glucose testing and were scheduled for a visit to the diabetes clinic 1 week after discharge. All remaining women were offered an OGTT within 6–12 weeks after delivery. Diabetes was diagnosed by either fasting venous plasma glucose ≥126 mg/dl (7.0 mmol/l) or a 2-h value ≥200 mg/dl (11.1 mmol/l), impaired fasting glucose (IFG) tolerance by fasting glucose >110 mg/dl (6.1 mmol/l), and IGT by 2-h glucose >140 mg/dl (7.7 mmol/l) (16).
Data collection and statistical analysis
Antenatal maternal clinical, glycemic, delivery, neonatal, and ppOGTT data were derived from the database of each institution. Neonatal macrosomia was defined as birth weight >90th percentile derived from a German national survey (17). As a primary measure of outcome, ppOGTT data diagnosed as IFG, IGT, or diabetes were combined to create a binary variable coded as abnormal. An OGTT fulfilling the criteria of both IFG and IGT was classified as IGT. Women with repeat elevated results in the postpartum glucose profiles (performed because of insulin-requiring GDM) were included in the group with abnormal postpartum glucose tolerance and were assigned to the groups mentioned above, depending on the elevated values in the profiles.
Variables considered as potentially predictive for an abnormal ppOGTT were analyzed with the statistical package SPSS 12.0 (SPSS, Chicago, IL). Data are presented as numbers and proportion for categorical variables or as means ± SD for continuous variables, respectively. Accordingly, a Pearson χ2 test or two-sided Mann-Whitney U test was used to test for differences between women with normal or abnormal ppOGTT. Differences with P < 0.05 were regarded as statistically significant.
Forward stepwise multivariate logistic regression analysis including all significant parameters from the univariate analysis was performed to identify independent predictors of an abnormal ppOGTT termed as risk factors. For this reason, variables were dichotomized according to conventionally used thresholds and/or statistical considerations (e.g., BMI ≥30 kg/m2 or antenatal 1-h OGTT >200 mg/dl). Results for each risk factor are presented as odds ratios (ORs) with 95% CI. Furthermore, based on the number of risk factors identified for each subject, the prevalence of an abnormal ppOGTT was assessed and used for the stratification of the study cohort into three groups according to low, intermediate, or high risk of an abnormal ppOGTT, respectively.
RESULTS
In 1,184 women (Kiel 331, Bonn 336, and Berlin 517) with GDM, the overall return rate for postpartum testing was 51.1% (Kiel 79.9%, Bonn 65.0%, and Berlin 23.4%). At the Berlin site, the lowest return rate of 13% was seen in women with Turkish or Arabian background who accounted for 32.8% of the population. A total of 605 women (Kiel 265, Bonn 219, and Berlin 121) fulfilled the inclusion criteria and qualified for the analysis. ppOGTT was found to be abnormal in 132 women (21.8%) and was classified as IFG in 17 (2.8%) and as IGT in 82 (13.6%); 33 women (5.5%) had diabetes. Diagnosis was based on abnormal glucose profiles in 17 women within 1 week after discharge. The rate of abnormal ppOGTT was similar in two of the participating centers (Kiel 17.7% and Bonn 21.5%) but was significantly elevated in the third center (Berlin 31.4%, P = 0.027). By univariate analyses, significant differences between women with normal and abnormal ppOGTTs were found for prepregnancy BMI, gestational age at diagnosis, all glucose values for the antenatal OGTT, gestational age at delivery, insulin therapy, and neonatal macrosomia (Table 1).
Table 1.
Maternal characteristics and delivery data of 605 women after pregnancies with GDM with normal and abnormal ppOGTT performed within 1 year after delivery
| Normal | Abnormal | P * | |
|---|---|---|---|
| n | 473 | 132 | |
| History | |||
| Prior GDM (%) | 14.8 | 19.1 | 0.233 |
| Prior macrosomia (%) | 5.7 | 7.6 | 0.416 |
| Family history of diabetes (%) | 56.6 | 60.5 | 0.541 |
| Parity | 2.2 ± 1.3 | 2.5 ± 1.6 | 0.060 |
| Age (years) | 32.7 ± 4.5 | 32.2 ± 5.6 | 0.651 |
| Prepregnancy BMI (kg/m2) | 25.8 ± 5.5 | 28.1 ± 6.1 | <0.001 |
| Weight gain (kg) | 11.9 ± 4.6 | 12.6 ± 5.1 | 0.457 |
| Antenatal glucose metabolism | |||
| Gestational age at diagnosis of GDM | 27.3 ± 6.1 | 24.2 ± 7.8 | <0.001 |
| Insulin therapy (%) | 141.0 | 64.1 | <0.001 |
| apOGTT fasting (mg/dl) | 88 ± 14 | 96 ± 20 | <0.001 |
| apOGTT 1 h | 191 ± 22 | 211 ± 34 | <0.001 |
| apOGTT 2 h | 144 ± 31 | 166 ± 41 | <0.001 |
| Highest fasting glucose in profiles during pregnancy (mg/dl) | 87 ± 11 | 90 ± 14 | 0.341 |
| Delivery | |||
| Gestational age at delivery | 39.5 ± 1.4 | 38.9 ± 2.3 | 0.005 |
| Macrosomia (>90th percentile) (%) | 8.9 | 16.0 | 0.022 |
| ppOGTT | |||
| Time point (month pp) | 3.7 ± 3.1 | 5.4 ± 6.7 | 0.186 |
| ppOGTT fasting (mg/dl) | 84 ± 9 | 100 ± 25 | <0.001 |
| ppOGTT 1 h | 148 ± 31 | 199 ± 45 | <0.001 |
| ppOGTT 2 h | 104 ± 19 | 158 ± 38 | <0.001 |
Data are means ± SD or %.
*P values (two-sided): χ2 test or Mann-Whitney U test, respectively. ap, antepartum; pp, postpartum.
The study site in Berlin had a very low return rate. To exclude a possible selection bias by overrepresentation of women at highest risk, we compared returners and nonreturners at this study site. There was no significant difference in maternal characteristics, glycemic parameters, or insulin use (31.0 vs. 32.8%) besides the 2-h OGTT value (154.9 vs. 146.4 mg/dl, P = 0.02).
In 566 subjects (93.6%), complete datasets for multivariate analysis were available. We found four independent risk factors: prepregnancy BMI ≥30 kg/m2 (prevalence of abnormal ppOGTT 36.0 vs. 17.3%), gestational age at diagnosis ≤24 weeks (32.4 vs. 18.0%), 1-h antenatal OGTT >200 mg/dl (35.2 vs. 14.8%), and insulin therapy (30.3 vs. 14.5%). A 1-h glucose value >200 mg/dl was the strongest predictor of postpartum glucose intolerance (Table 2). BMI ≥30 kg/m2 was present in 22.5% of the women, gestational age at diagnosis was ≤24 weeks in 28.1%, insulin therapy was being used in 45.8%, and a 1-h postchallenge glucose value >200 mg/dl was seen in 31.9%.
Table 2.
Independent risk factors for abnormal postpartum glucose tolerance in 605 mothers with GDM
| Factor | OR (95% CI) | P |
|---|---|---|
| Antenatal OGTT 1 h >200 mg/dl (11.1 mmol/l) | 2.73 (1.77–4.21) | <0.001 |
| Insulin therapy | 2.12 (1.36–3.30) | 0.001 |
| Prepregnancy BMI ≥30 kg/m2 | 2.12 (1.33–3.40) | 0.002 |
| Gestational age at GDM diagnosis ≤24 weeks | 1.81 (1.16–2.85) | 0.010 |
A total of 566 datasets (93.6%) with complete parameters were used for the multiple logistic regression analysis.
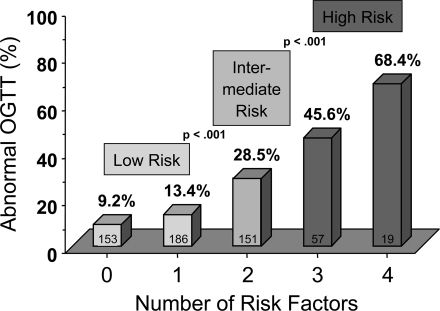
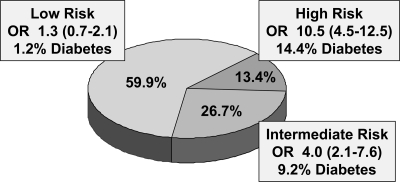
Based on the results of the analysis above, we calculated the prevalence of an abnormal ppOGTT according to the number of identified risk factors present in each of the 566 women (Fig. 1). The majority of the women (339 of 566, 59.9%) had <2 risk factors. A significant increase in the prevalence of abnormal ppOGTTs was seen between <2 and 2 risk factors and between 2 and >2 risk factors (Fig. 1). The proportion of subjects with either <2 risk factors (low risk), 2 risk factors (intermediate risk), or >2 risk factors (high risk), and the corresponding ORs for postpartum glucose intolerance are presented in Fig. 2. The prevalence of abnormal postpartum glucose tolerance was 11.5% (39 of 339) in the low-risk group, 28.5% (43 of 151) in the intermediate-risk group, and 51.3% (39 of 76) in the high-risk group.
Figure 1.
Prevalence of abnormal postpartum glucose tolerance in women after GDM divided by the number of risk factors present. There was a significant increase in abnormal postpartum glucose tolerance between women with low risk (<2 risk factors) and intermediate (2 risk factors) (P < 0.001), and intermediate (2 risk factors) and high risk (>2 risk factors) (P < 0.001).
Figure 2.
Proportion of subjects stratified by the number of independent risk factors: low-risk group (<2 risk factors), intermediate-risk group (2 risk factors), and high-risk group (3 or 4 risk factors). The risk of an abnormal ppOGTT is estimated by OR (95% CI) according to the risk in 153 subjects without any positive factor. Overall 86.2% of the overt diabetes (25 of 29) and 67.8% of all abnormal OGTTs (82 of 121) were identified within the intermediate/high-risk group.
When OGTT testing is concentrated on women with intermediate/high risk (≥2 risk factors, 40.1% of subjects), a total rate of 36.1% abnormal glucose tolerance could be expected, compared with a reduced rate of 11.5% in women with <2 risk factors. Of the women with low risk, only 4 (1.2%) had overt diabetes, in contrast with 25 woman (11.0%) in the intermediate/high-risk group. That means ∼85 low-risk women would need to be tested to uncover 1 case of diabetes compared with 10 women per case in the intermediate/high-risk women. In total, 86.2% of cases of overt diabetes (25 of 29) and 67.8% of all abnormal OGTTs (82 of 121) were identified within the intermediate/high-risk group.
CONCLUSIONS
In a large-scale, multicenter study, we found 21% persistent glucose intolerance within 1 year after a pregnancy with GDM in Caucasian women. However, we saw that the return rate was different at the three study sites. We suspect that the return rate might have been influenced by the population composite and the care setting. Based on identified risk factors, we developed an algorithm for postpartum glucose testing that allows us to target women who should be intensively followed and motivated to return for testing because of their high risk for abnormal glucose tolerance. A combination of ≥2 risk factors, which was present in 40% of the population, was highly predictive for the development of diabetes within 1 year after delivery, and 86% of diabetes would be detected.
The prevalence of abnormal glucose testing observed in our population was within the range reported from other studies. The prevalence of isolated IFG varies between 3 and 6%, the prevalence of IGT varies between 7 and 29%, and the prevalence of diabetes varies between 5 and 12% (4,7,10–13,15,18,19). Particular risk factors associated with abnormal postpartum glucose tolerance varied among studies, but early diagnosis of GDM and high maternal BMI as potential signs of preexisting insulin resistance and insulin use as a marker of the degree of hyperglycemia had been proven to be predictive in most of the studies. Age, prior GDM, family history of diabetes, weight gain, or neonatal macrosomia often lost their predictive value when maternal glucose values were included in the multivariate analysis (1,3). We identified four independent antenatal risk factors; besides maternal BMI >30 kg/m2, gestational age at diagnosis <24 weeks, and insulin use, a 1-h glucose value >200 mg/dl increased the risk by almost threefold. There is controversy in the literature over whether the antenatal fasting (7,15,20) or postchallenge values (10–12), or both (18), or the number of abnormal values of an OGTT (13) is more predictive for postpartum diabetes. Similar to our data, in one of the large studies that included almost 3,000 women, the 1-h OGTT value was proven to be superior to the fasting glucose value (11). Recent data from Canada demonstrated lower postpartum insulin sensitivity in women with an isolated abnormal 1-h glucose value compared with those with only elevated 2- or 3-h high values (21). The postchallenge hyperglycemia reflects the first-phase release. Insulin clamps during pregnancy in women with GDM showed that a low first-phase intravenous glucose tolerance test response is independently associated with a high risk for type 2 diabetes within 6 months after delivery (14).
The presence of almost 22% abnormal glucose tolerance within the 1st year after GDM underlines the importance of early postpartum testing. Despite the knowledge that after GDM women have a substantial risk of staying glucose intolerant or developing diabetes with the highest conversion rate in the 1st years after the index pregnancy (3), physician and patient compliance for early postpartum glucose testing is low. Only 20% of the obstetricians, even in an academic medical center, ordered postpartum diabetes screening tests for their patients (22). With a few exceptions (11,12), most studies reported a return rate less than or ∼50% (7,9,10,19,23,24).
Overall, 51% of the women in our study returned for testing, but we noted a remarkable diversity between the study sites that might be influenced by the social and ethnic composition of the population. The highest recall rates were achieved in the setting of a private clinic in an urban middle-class population, whereas in a community obstetrical hospital in an underprivileged Berlin district with a high rate of immigrants (41.2%) only 23% of all women returned for testing and only 13% of women with Turkish or Arabian background returned. On the patient side, it is likely that different overall health awareness and increased family obligations in large families may contribute to low testing rates. Unfortunately, it seems that, especially in populations with high prevalence of postpartum glucose intolerance, many obstacles have to be overcome. Some of these are related to the health system, some are internal to the patient, and some are socioeconomic or cultural.
An algorithm to target women at high risk for postpartum glucose intolerance might ensure that the majority of postpartum diabetes is identified. Ideally, it should be possible to calculate the individual risk of each woman for diabetes during the early postpartum period based on easily available antenatal risk factors. Based on the prevalence of abnormal ppOGTTs according to the number of risk factors present, we defined three risk categories. Women with low risk (OR 1.3) had a low prevalence of abnormal ppOGTTs (11%), whereas in women with intermediate or high risk we expect to see 36% with persistent glucose intolerance. These women accounted for 40% of our population, but the rate of women at high risk may vary in other populations depending, for example, on the obesity and underlying type 2 diabetes prevalence. If our model of risk factor–based postpartum screening holds in other populations, we would detect 86% of postpartum diabetes by focusing on intensive counseling and following women who are likely to remain diabetic. Concentrating our efforts on motivating these women to return for testing and developing a system of cooperation with other caregivers, e.g., the pediatrician who sees the children, might reduce the number of missed cases of postpartum diabetes. Missing the opportunity to identify pre-diabetes or diabetes in underserved women may mean that diabetes is not identified until a much later stage than in women who access health care.
The limitation of our study is that we do not have information about the postpartum glucose tolerance status of all of the women who did not return for postpartum testing. We have relatively complete data for two centers, but because of the low return rates in Berlin, the incidence of abnormal ppOGTTs might be overestimated. Because nonreturners at the site with a low return rate were not different from returners regarding risk factors for diabetes we consider our results to be representative for the whole population. However, further studies with prospective use of our model in different populations have to be conducted to prove the validity and transferability of our data.
Considering the high rate of early glucose intolerance, it is imperative that we ensure that women who develop GDM understand that glucose intolerance may persist or return after pregnancy and that this risk for diabetes can be modified by lifestyle changes (6), and, therefore, postpartum testing is essential. However, widespread implementation of ppOGTT is difficult. The model of risk assessment we presented, which is based on easily available antenatal data, may allow us to target women with a high need of intensive motivation for postpartum testing and therefore may offer a chance to reduce the number of missed cases of postpartum diabetes in women with recent GDM.
Acknowledgments
No potential conflicts of interest relevant to this article were reported.
Parts of this study were presented in abstract form at the 66th Scientific Sessions of the American Diabetes Association, Washington, DC, 9–13 June 2006.
Footnotes
The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C. Section 1734 solely to indicate this fact.
References
- 1. Löbner K, Knopff A, Baumgarten A, Mollenhauser U, Marienfeld S, Garrido-Franco M, Bonifacio E, Ziegler A: Predictors of postpartum diabetes in women with gestational diabetes mellitus. Diabetes 2006;55:792–797 [DOI] [PubMed] [Google Scholar]
- 2. Lauenborg J, Hansen T, Jensen D, Vestergaard H, Molsted-Pedersen L, Hornnes P, Locht H, Pedersen O, Damm P: Increasing incidence of diabetes after gestational diabetes. Diabetes Care 2004;27:1194–1199 [DOI] [PubMed] [Google Scholar]
- 3. Kim C, Newton K, Knopp R: Gestational diabetes and the incidence of type 2 diabetes. Diabetes Care 2002;25:1862–1868 [DOI] [PubMed] [Google Scholar]
- 4. Kitzmiller J, Dang-Kilduff L, Taslimi M: Gestational diabetes after delivery. Diabetes Care 2007;30(Suppl. 2):S225–S235 [DOI] [PubMed] [Google Scholar]
- 5. Metzger B, Buchanan T, Coustan D, De Leiva A, Dunger D, Hod M, Kitzmiller J, Kjos S, Oats J, Pettitt D, Sacks D, Zoupas C: Summary and recommendations of the 5th Workshop-Conference on Gestational Diabetes Mellitus. Diabetes Care 2007;30(Suppl. 2):S251–S260 [DOI] [PubMed] [Google Scholar]
- 6. Knowler W, Barett-Connor E, Fowler S, Hamman R, Lachin J, Walker E, Nathan D: Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med 2002;346:393–403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Schaefer-Graf U, Buchanan T, Xiang A, Peters R, Kjos S: Clinical predictors for a high risk for development of diabetes mellitus in the early puerperium in women with recent gestational diabetes. Am J Obstet Gynecol 2002;186:751–756 [DOI] [PubMed] [Google Scholar]
- 8. Clark H, Van Walraven C, Code C, Karovitch A, Keely E: Did duplication of clinical practice guideline recommendation to screen for type 2 diabetes in women with gestational diabetes change practice? Diabetes Care 2003;26:265–268 [DOI] [PubMed] [Google Scholar]
- 9. Smirnakis K, Chasan-Taber L, Wolf M, Markenson G, Ecker J, Thadhani R: Postpartum diabetes screening in women with history of gestational diabetes. Obstet Gynecol 2005;106:1297–1303 [DOI] [PubMed] [Google Scholar]
- 10. Greenberg LR, Moore TR, Murphy H: Gestational diabetes mellitus: antenatal variables as predictors of postpartum glucose intolerance. Obstet Gynecol 1995;86:97–101 [DOI] [PubMed] [Google Scholar]
- 11. Wein P, Beischer N, Sheedy M: Studies of postnatal diabetes mellitus in women who had gestational diabetes. Part 2. Prevalence and predictors of diabetes mellitus after delivery. Aust N Z J Obstet Gynaecol 1997;37:420–423 [DOI] [PubMed] [Google Scholar]
- 12. Aberg A, Jönsson E, Eskilsson I, Landin-Olsson M, Frid A: Predictive factors of developing diabetes mellitus in women with gestational diabetes. Acta Obstet Gynecol Scand 2002;81:11–16 [DOI] [PubMed] [Google Scholar]
- 13. Pallardo F, Herranz L, Garcia-Ingelmo T, Grande C, Martin-Vaquero P, Janez M, Gonzalez A: Early postpartum metabolic assessment in women with prior gestational diabetes. Diabetes Care 1999;22:1053–1058 [DOI] [PubMed] [Google Scholar]
- 14. Buchanan T, Xiang A, Kjos S, Lee W, Trigo E, Nader I, Bergner A, Palmer J, Peters R: Gestational diabetes: antepartum characteristics that predict postpartum glucose intolerance and type 2 diabetes in Latino women. Diabetes 1998;47:1302–1310 [DOI] [PubMed] [Google Scholar]
- 15. Catalano P, Vargo K, Bernstein I, Amini S: Incidence and risk associated with abnormal glucose tolerance in women with gestational diabetes. Am J Obstet Gynecol 1991;165:914–919 [DOI] [PubMed] [Google Scholar]
- 16. American Diabetes Association. Position statement: Gestational diabetes mellitus. Diabetes Care 2000;23(Suppl. 1):S77–S79 [PubMed] [Google Scholar]
- 17. Voigt M, Schneider K, Jährig K: Analysis of a 1992 birth sample in Germany. 1: New percentile values of the body weight of newborn infants. Geburtshilfe Frauenheilkd 1996;56:550–558 [in German] [DOI] [PubMed] [Google Scholar]
- 18. Weijers R, Bekedam D, Goldschmidt H, Smulders Y: The clinical usefulness of glucose tolerance testing in gestational diabetes to predict early postpartum diabetes mellitus. Clin Chem Lab Med 2006;44:99–104 [DOI] [PubMed] [Google Scholar]
- 19. Jang H, Yim C, Han K, Yoon H, Han I, Kim M, Yang J, Cho N: Gestational diabetes mellitus in Korea: prevalence and prediction of glucose intolerance at early postpartum. Diabetes Res Clin Pract 2003;61:117–124 [DOI] [PubMed] [Google Scholar]
- 20. Kjos SL, Buchanan TA, Greenspoon JS, Montoro M, Bernstein GS, Mestman JH: Gestational diabetes mellitus: the prevalence of glucose intolerance and diabetes mellitus in the first two months post partum. Am J Obstet Gynecol 1990;163:93–98 [DOI] [PubMed] [Google Scholar]
- 21. Retnakaran R, Qi Y, Sermer M, Connelly P, Zinman B, Hanley A: Isolated hyperglycemia at 1 hour on oral glucose tolerance test in pregnancy resembles gestational diabetes mellitus in predicting postpartum metabolic dysfunction. Diabetes Care 2008;31:1275–1281 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Almario C, Ecker T, Moroz L, Bucovetsky L, Berghella V, Baxter J: Obstetricians seldom provide postpartum diabetes screening for women with gestational diabetes. Am J Obstet Gynecol 2008;198:528.e1–e5 [DOI] [PubMed] [Google Scholar]
- 23. Kaufmann R, Smith T, Bochantin T, Khardori R, Evans M, Steahly L: Failure to obtain follow-up testing for gestational diabetic patients in a rural population. Obstet Gynecol 1999;93:734–737 [DOI] [PubMed] [Google Scholar]
- 24. Hunt K, Conway D: Who returns for postpartum glucose screening following gestational diabetes mellitus? Am J Obstet Gynecol 2008;198:404.e1–e6 [DOI] [PMC free article] [PubMed] [Google Scholar]