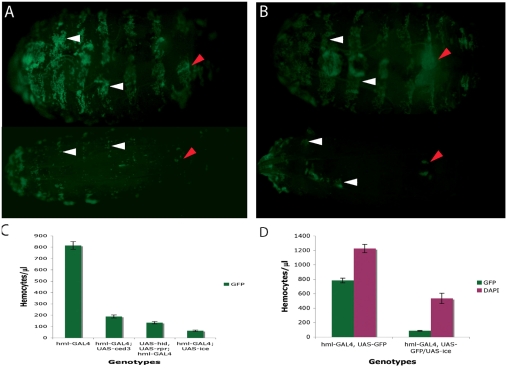
Fig. 1.
Ablation of haemocytes using the GAL4-UAS system. (A) hmlGAl4 and (B) pxnGAL4 were crossed to a UAS line expressing the human apoptotic gene ICE. The cross was hml-GAL4, UAS-GFP or pxn-GAL4, UAS-GFP × TM6B/UAS-ICE (hml-GAL4, UAS-GFP; UAS-ICE /+ or pxn-GAL4, UAS-GFP; UAS-ICE /+, bottom panels of A and B, respectively). Larvae in top panels are the hmlGAL4, UAS-GFP; TM6B/+ and pxnGAL4, UAS-GFP; TM6B/+ siblings of the ablated larvae, hence their Tubby appearance. Visualisation of ablation through a UAS-GFP transgene showed that circulating blood cells and a large number of sessile haemocytes were eliminated (compare top and bottom panels in both A and B). However, some groups of sessile haemocytes were resistant (white arrowheads). The larval haematopoietic organ (red arrowheads) was not deleted but was severely reduced. (C) Quantification of circulating haemocytes marked with GPF in hmlGAL4, UAS-GFP larvae after expression of C elegans ced-3, Drosophila hid and reaper (hid, rpr) and human ICE measured by the number of cells/μl of blood. The hmlGAL4, UAS-GFP strain (without any of the pro-apoptotic gene UAS constructs) was used as the wild-type control (hml). Six independent experiments were performed. The graph represents the mean values ± s.d. Similar results were obtained with pxnGAL4 and srpD-GAL4 (not shown). (D) Comparison between the total number of circulating haemocytes (DAPI) and those labelled by GFP in hmlGAL4, UAS-GFP larvae (left column) or hmlGAL4, UAS-GFP; UAS-ICE larvae (right column). Approximately 59% of circulating haemocytes were deleted by ablation.