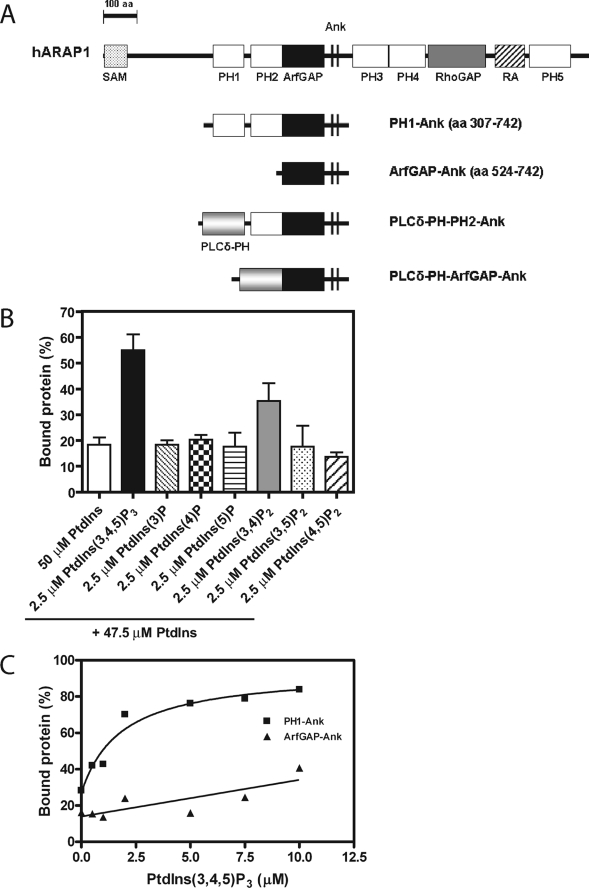
FIGURE 1.
ARAP1 binding to phospholipids. A, schematic of the recombinant proteins used in this study. Domain abbreviations: Ank, ankyrin repeat; PLCδ-PH, PH domain of phospholipase C δ; RA, Ras association motif; RhoGAP, Rho GTPase-activating domain. B, ARAP1 phosphoinositide binding specificity. 500 nm PH1-Ank recombinant protein was incubated with sucrose-loaded LUVs formed by extrusion through a 1-μm pore filter. LUVs contained PtdIns alone or PtdIns with 2.5 μm PtdIns(3,4,5)P3, 2.5 μm PtdIns(3)P, 2.5 μm PtdIns(4)P, 2.5 μm PtdIns(5)P, 2.5 μm PtdIns(3,4)P2, 2.5 μm PtdIns(3,5)P2, or 2.5 μm PtdIns(4,5)P2 with a total phosphoinositide concentration of 50 μm and a total phospholipid concentration of 500 μm. Vesicles were precipitated by ultracentrifugation, and associated proteins were separated by SDS-PAGE. The amount of precipitated protein was determined by densitometry of the Coomassie Blue-stained gels with standards on each gel. C, PtdIns(3,4,5)P3-dependent binding of ARAP1 to LUVs. 1 μm PH1-Ank or ArfGAP-Ank recombinant protein was incubated with 1 mm sucrose-loaded LUVs formed by extrusion through a 1-μm pore size filter containing varying concentration of PtdIns(3,4,5)P3. Precipitation of LUVs and analysis of associated proteins were performed as described in B. The average ± S.E. of three independent experiments is presented.