Table 3.
Effect of Changing Ring Substituents, or the Substitution of C-4 of the Ring by N, on the Equilibrium Constant for Addition of Glycine to Benzaldehyde to Form the Corresponding Imine.a
| aldehyde | (Kadd)Xb | ΔΔGXc (kcal/mol) |
|
ΔΔGXe (kcal/mol) |
p(Ka)X-IMDf | Δ(pKa)Xg | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
 |
44 | 1.0 | 0 | 0.0033 | 1.0 | 0 | 6.29 | 0 | |||
 |
1.76 | 0.040 | 1.9 | 72 | 22,000 | −5.9 | 12.13 | 5.8 | |||
 |
0.49 | 0.011 | 2.7 | 0.29 | 88 | −2.7 | 10.16 | 3.9 | |||
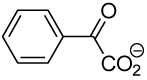 |
1.5h | 0.034 | 2.0 | 1 × 10−4h | 0.030 | 2.1 | 6.19h | −0.1 | |||
 |
500 | 11.4 | −1.4 | 0.002 | 0.61 | 0.3 | 4.95 | −1.3 |
Data from Table 1 for reactions in D2O, unless noted otherwise.
Equilibrium constants for addition of glycine anion to the aldehyde in the first Table row to form the corresponding unprotonated imine.
The effect of the substituent X on the change in Gibbs energy for formation of the imine from the free aldehyde and amino acid.
Equilibrium constants for addition of glycine zwitterion to the aldehyde in the first Table row to form the corresponding iminium ion.
The effect of the substituent X on the change in Gibbs energy for formation of the iminium ion from the free aldehyde and amino acid.
Apparent acidity constants for the substituted iminium ions.
The effect of the substituent X on the pKa for ionization of the iminium ion.
Data from ref 9 for reactions in D2O at 25 °C and I = 1.0 (KCl).
