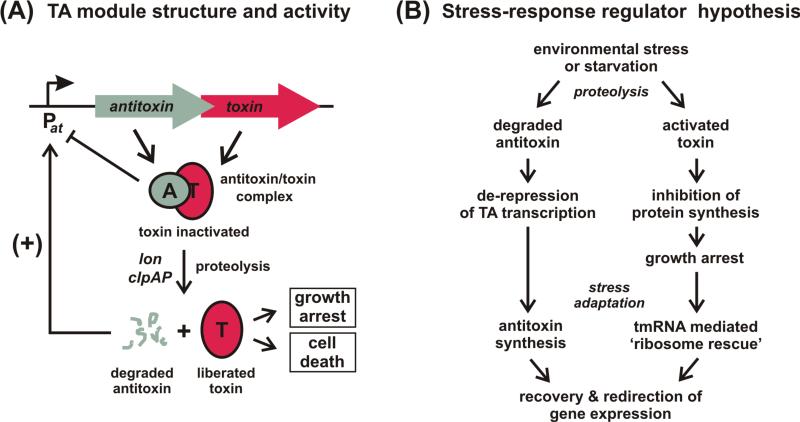
Figure 2. Toxin-antitoxin (TA) module organization and the stress-response regulator hypothesis.
(A) TA module genetic organization. TA systems are typically two-gene operons with the antitoxin encoded upstream of the toxin gene. Proteic antitoxins bind their cognate toxins and inhibit toxin activity. Antitoxins and toxin-antitoxin complexes repress TA module transcription. In Escherichia coli, toxin activation involves degradation of antitoxin by either the Lon or ClpAP proteases. Activated toxins inhibit cell growth and/or lead to cell death.
(B) The stress-response regulator hypothesis of chromosomal TA function. Environmental stresses such as starvation activate chromosomal TA modules through antitoxin degradation. Many toxins are mRNA-specific RNases that rapidly inhibit protein synthesis, inducing a bacteriostatic state. Upon adaptation (or removal of the stress), antitoxin synthesis resumes to counteract toxin activity, and tmRNA activity rescues ribosomes arrested on toxin-cleaved messages.