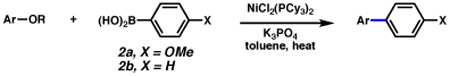
Table 1.
Cross-coupling of aryl carbamates and carbonates with arylboronic acids 2a or 2b.a
 | ||||
|---|---|---|---|---|
| entry | Ar–OR | (HO)2B–Ar | product | yieldc |
| 1b | 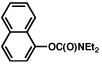 |
2a | 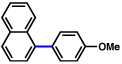 |
51% |
| 2 | 86% | |||
| 3 | 2a | 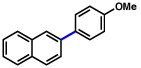 |
47% | |
| 4 | 2b | 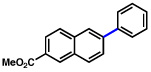 |
54% | |
| 5 | 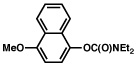 |
2b | 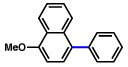 |
77% |
| 6 | 2a | 52% | ||
| 7 | 2b | 41% | ||
| 8 | 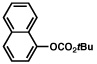 |
2a | 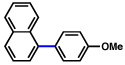 |
72% |
| 9 | 2a | 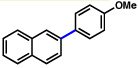 |
85% | |
| 10 | 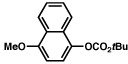 |
2b | 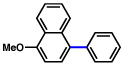 |
65% |
Conditions: NiCl2(PCy3)2 (10 mol%), ArB(OH)2 (4 equiv), K3PO4 (7.2 equiv), toluene (0.3 M), 130 °C for 24 h.
Conditions: NiCl2(PCy3)2 (5 mol%), ArB(OH)2 (2.5 equiv), K3PO4 (4.5 equiv), toluene (0.3 M), 110 °C, 24 h.
Isolated yields.
