Abstract
The present study was conducted to examine post-thaw in vitro developmental competence of buffalo embryos cryopreserved by cytoskeletal stabilization and vitrification. In vitro produced embryos were incubated with a medium containing cytochalasin-b (cyto-b) in a CO2 incubator for 40 min for microfilament stabilization and were cryopreserved by a two-step vitrification method at 24℃ in the presence of cyto-b. Initially, the embryos were exposed to 10% ethylene glycol (EG) and 10% dimethylsulfoxide (DMSO) in a base medium for 4 min. After the initial exposure, the embryos were transferred to a 7 µl drop of 25% EG and 25% DMSO in base medium and 0.3 M sucrose for 45 sec. After warming, the embryos were cultured in vitro for 72 h. The post-thaw in vitro developmental competence of the cyto-b-treated embryos did not differ significantly from those vitrified without cyto-b treatment. The hatching rates of morulae vitrified without cyto-b treatment was significantly lower than the non-vitrified control. However, the hatching rate of cyto-b-treated vitrified morulae did not differ significantly from the non-vitrified control. This study demonstrates that freezing of buffalo embryos by cytoskeletal stabilization and vitrification is a reliable method for long-term preservation.
Keywords: buffalo, cytochalasin-b, embryo, vitrificationm
Introduction
The major obstacle to the large-scale commercial application of in vitro produced (IVP) embryos, particularly in buffalos, is the lack of a suitable cryopreservation method. Approaches like slow freezing, fast cooling, slow or fast warming and vitrification have been employed to cryopreserve the embryos of domestic animals. Vitrification is a physical process in which a solution is transformed into a stable amorphous glass by rapid cooling, bypassing ice crystal formation while maintaining the properties of the liquid in a solidified form [16]. The glass is devoid of ice crystals and embryos are not subjected to cellular damage associated with ice crystal formation [17]. The in vitro and in vivo survival rates of vitrified embryos are reasonable in cattle [2,9,15] and buffaloes [8,10,11].
Cryopreservation can be extremely disruptive to the cellular organization of embryos [5]. To minimize cytoskeletal disruption of the embryos during and after cryopreservation, cytoskeletal stabilizers are added to the vitrification solution. Information on cryopreservation of IVP buffalo embryos by vitrification is scarce. The present study was carried out to examine the post-thaw in vitro developmental competence of buffalo embryos cryopreserved by cytoskeletal stabilization and vitrification.
Materials and Methods
Media and chemicals were obtained from Sigma-Aldrich (USA) unless otherwise indicated. Filters having a 0.22 µm pore size were from Millipore (USA). Large, square and circular 35 mm-diameter disposable Falcon plastic dishes, and plastic tubes were purchased from Becton Dickinson Labware (USA). Steer serum (SS) was collected from the institute herd by clotting the blood from a long-term castrated bullock, which was then heat inactivated at 56℃ for 30 min, sterilized by filtration and stored in 2 ml aliquots at -20℃ until use. The same pool of serum was used throughout the study.
In vitro embryo production
Ovaries were collected from mature, non-pregnant buffaloes (Bubalus bubalis) from the Corporation slaughterhouse, Bangalore, and brought to the laboratory in normal saline (32~33℃) supplemented with gentamicin (50 µg/ml) within 2 h after slaughter. Ovaries were washed three times in normal saline in the laboratory and extra-ovarian tissues were removed. The oocytes were aspirated from the visible surface antral follicles using a 20 gauge hypodermic needle attached to a 5 ml syringe containing 0.5~1.0 ml of aspiration medium consisting of TCM 199 supplemented with 5% SS.
Oocytes were identified and their morphology evaluated using a stereo zoom microscope; oocytes having a compact cumulus investment were retained for use. The oocytes were washed once with an aspiration medium and twice in a culture medium consisting of TCM 199, 10% SS, 50 µM of cysteamine and 0.02 U of porcine follicle stimulating hormone/ml. Oocytes in groups of 5~10 were transferred to 50 µl droplets of culture medium. The oocyte-containing droplets were covered with warm (38.5℃) mineral oil and in vitro maturation of the oocytes proceeded during incubation at 38.5℃ and 90~95% relative humidity in an atmosphere of 5% CO2 for 24 h. The in vitro fertilization (day 0) of oocytes and culture of embryos was performed as previously described [11]. Good quality compact morulae on day 6 and blastocysts on day 7 were selected for vitrification.
Microfilament stabilization and vitrification
Embryos were incubated in a CO2 incubator for 45 min in an embryo culture medium consisting of TCM 199 and 10% SS supplemented with 7.5 µg/ml cytochalasin-b (cyto-b) for microfilament stabilization. These embryos were vitrified following a two-step addition of cryoprotectant in the presence of cyto-b. Initially, the embryos were exposed to 10% ethylene glycol (EG) and 10% dimethylsulfoxide (DMSO) in base medium for 4 min. After the initial exposure, the embryos were transferred to a 7 µl drop of 25% EG and 25% DMSO in base medium and 0.3 M sucrose for 45 sec. During the second exposure step, the embryos were loaded into the middle part of 0.25 ml straws (IMV; 3~4 embryos per straw) in a small volume of vitrification solution. The rest of the straw was filled with a dilution medium consisting of 0.5 M sucrose in base medium that was separated by air bubbles from the column of vitrification solution containing the embryos. The straw was then heat-sealed and, 45 sec later, was plunged vertically, sealed end first, into liquid nitrogen to cover the embryos. The rest of the straw was then slowly immersed into the liquid nitrogen within 30 sec. In the vitrified control group, the embryos were vitrified without the inclusion of microfilament stabilization and cyto-b in the vitrification medium. After 1 week of cryopreservation, the straws were warmed in air (24℃) for 10 sec and then in water bath horizontally at 20℃ until the ice disappeared. The straw was held at the sealed end and shaken to mix the columns. The embryos were expelled into a 30-mm diameter Petri dish and maintained at 24℃ for 5 min. To remove cyto-b, the embryos were washed three times in 200 µl drops of embryo culture medium. The embryos were cultured for further development in the embryo culture medium on a 7-day-old monolayer of oviductal epithelial cells in a CO2 incubator. Re-expansion and hatching rates for morulae and blastocysts were evaluated at 72 h post-thaw in vitro culture. For the non-vitrified control, non-vitrified morulae and blastocysts from the same batch as vitrified samples were cultured on a monolayer of oviductal epithelial cells in a CO2 incubator for 72 h after being held at 24℃ during the entire vitrification procedure.
Statistical analyses
The percentage values were subjected to arcsine transformation before being statistically analyzed. The data on post-thaw in vitro development of morulae and blastocysts were analyzed by one way ANOVA to verify differences between the groups followed by the Bonneforni multiple comparison test between group means by using statistical software (GraphPad Prism; GraphPad, USA). Significance of differences between the mean values was determined at p < 0.05. Results are expressed as mean ± SE.
Results
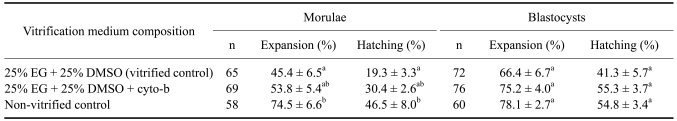
The expansion and hatching rate of cyto-b-treated vitrified morulae did not differ significantly from non-vitrified controls on post-thaw in vitro culture (Table 1). However, the expansion and hatching rate of morulae vitrified without cyto-b treatment was significantly lower than the non-vitrified control. The development of blastocysts vitrified with or without cyto-b treatment to expansion and hatching stages did not differ significantly from non-vitrified control. However, hatching rates of cyto-b-treated morulae and blastocysts were not significantly higher than those vitrified without cyto-b treatment.
Table 1.
Post-thaw in vitro development of buffalo embryos after cryopreservation by vitrification under the influence of microfilament stabilizer cyto-b
Means within the same column with different superscript differ significantly (p < 0.05). EG: ethylene glycol. G: glycerol. DMSO: dimethylsulfoxide. cyto-b: cytochalasin-b.
Discussion
The present study examined the post-thaw in vitro developmental competence of buffalo embryos cryopreserved by cytoskeletal stabilization and vitrification. Post-thaw in vitro developmental competence of the embryos vitrified after cytoskeletal stabilization in the presence of cyto-b did not differ significantly than those vitrified without microfilament stabilization and cyto-b in the vitrification medium. Embryo survival depends on the cryoprotectant used, species involved, and stage of development, as well as the culture systems used for IVP embryos. Cryoprotectant mixtures like EG and DMSO may have some advantages over solutions containing only one penetrating cryoprotectant [3,14,20]. The ability of embryos to hatch after 48 h post-thaw in vitro culture is improved when two (EG, DMSO) or three (EG, DMSO and 1,3-butanediol) permeating cryoprotectants are included in the vitrification media rather than a single permeating cryoprotectant like EG [15]. The use of two permeable cryoprotectants rather than a single cryoprotectant such as EG enables the use of lower concentrations of each cryoprotectant, thereby reducing cryoprotectant toxicity. The composition of a vitrification solution (permeating cryoprotectants and non-permeating macromolecules or saccharides) is among the factors influencing cryosurvival of embryos. The incorporation of DMSO into an EG-containing medium has at least two advantages. Firstly, vitrification is facilitated because of the greater glass-forming characteristics of DMSO [1]. Secondly, permeability of each cryoprotectant is enhanced in the presence of the other [21]. In buffaloes, the morulae and blastocyts vitrified by a 4 min exposure to 25% EG and 25% DMSO display a better hatching rate than other combinations of vitrification medium composition and exposure time [10].
Cytoskeletal stabilizers such as the cytochalasins have been used extensively in reproductive biotechnology for studies dealing with micromanipulation of embryonic development [12,18]. It is generally accepted that cytochalasin is a microfilament inhibitor capable of disrupting actin polymerization by blocking monomer addition at the fast-growing end of the polymers [19], thus preventing cytokinesis without affecting karyokinesis [4,13]. Treatment of cells with cytochalasins makes the plasma membrane less rigid and more elastic so that microfilaments are disrupted during micromanipulation [12]. In the present study, the post thaw in vitro developmental competence of cyto-b-treated embryos was not affected.
Cryoprotectants-the organic solutes that help to protect cellular organelles during cryopreservation-can also damage the cytoskeletal system, as they can be toxic and cause disruptive osmotic damage to the cell. Although many cryoprotectants depolymerize microfilaments and microtubules prior to cryopreservation, they might cause irreparable damage to cytoskeletal components. An intact cytoskeleton is essential for cytokinesis and karyokinesis; irreversibly disruption of the cytoskeleton will abrogate the mitotic cell cycle, could compromise junctional complexes and affects solute transport systems. Maintaining the integrity of the cytoarchitecture within an embryo during cryopreservation is, thus, of utmost importance. In porcines, cyto-b-treated embryos display better development than those vitrified without cyto-b treatment [6]. Similarly, in the present study the hatching rates of cyto-b-treated morulae and blastocysts were not significantly higher than those vitrified without cyto-b treatment.
In conclusion, the present study demonstrates that cryopreservation of buffalo embryos by cytoskeletal stabilization and vitrification is a reliable method for long-term preservation. IVP buffalo embryos cryopreserved by cytoskeletal-stabilization and vitrification achieved a maximum hatching rate of 30% and 55% on post-thaw in vitro culture of morulae and blastocysts.
Acknowledgments
We thank Dr. K. T. Sampath, Director, National Institute of Animal Nutrition and Physiology, Bangalore, India for providing the necessary facilities to complete this research.
References
- 1.Ali J, Shelton JN. Design of vitrification solutions for the cryopreservation of embryos. J Reprod Fertil. 1993;99:471–477. doi: 10.1530/jrf.0.0990471. [DOI] [PubMed] [Google Scholar]
- 2.Campos-Chillòn LF, Walker DJ, de la Torre-Sanchez JF, Seidel GE., Jr In vitro assessment of a direct transfer vitrification procedure for bovine embryos. Theriogenology. 2006;65:1200–1214. doi: 10.1016/j.theriogenology.2005.07.015. [DOI] [PubMed] [Google Scholar]
- 3.Critser JK, Agca Y, Gunasena KT. The cryobiology of mammalian oocytes. In: Karow AM, Critser JK, editors. Reproductive Tissue Banking: Scientific Principles. San Diego: Academic Press; 1997. pp. 332–358. [Google Scholar]
- 4.Cuthbertson KSR. Parthenogenetic activation of mouse oocytes in vitro with ethanol and benzyl alcohol. J Exp Zool. 1983;226:311–314. doi: 10.1002/jez.1402260217. [DOI] [PubMed] [Google Scholar]
- 5.Dobrinsky JR. Cellular approach to cryopreservation of embryos. Theriogenology. 1996;45:17–26. [Google Scholar]
- 6.Dobrinsky JR, Pursel VG, Long CR, Johnson LA. Birth of piglets after transfer of embryos cryopreserved by cytoskeletal stabilization and vitrification. Biol Reprod. 2000;62:564–570. doi: 10.1095/biolreprod62.3.564. [DOI] [PubMed] [Google Scholar]
- 7.Gasparrini B. In vitro embryo production in buffalo species: state of the art. Theriogenology. 2002;57:237–256. doi: 10.1016/s0093-691x(01)00669-0. [DOI] [PubMed] [Google Scholar]
- 8.Hufana-Duran D, Pedro PB, Venturina HV, Hufana RD, Salazar AL, Duran PG, Cruz LC. Post-warming hatching and birth of live calves following transfer of in vitro-derived vitrified water buffalo (Bubalus bubalis) embryos. Theriogenology. 2004;61:1429–1439. doi: 10.1016/j.theriogenology.2003.08.011. [DOI] [PubMed] [Google Scholar]
- 9.Kaidi S, Donnay I, Van Langendonckt A, Dessy F, Massip A. Comparison of two co-culture systems to assess the survival of in vitro produced bovine blastocysts after vitrification. Anim Reprod Sci. 1998;52:39–50. doi: 10.1016/s0378-4320(98)00089-x. [DOI] [PubMed] [Google Scholar]
- 10.Manjunatha BM, Gupta PS, Ravindra JP, Devaraj M, Nandi S. Effect of vitrification medium composition and exposure time on post-thaw development of buffalo embryos produced in vitro. Vet J. 2009;179:287–291. doi: 10.1016/j.tvjl.2007.08.031. [DOI] [PubMed] [Google Scholar]
- 11.Manjunatha BM, Gupta PSP, Ravindra JP, Devaraj M, Nandi S. In vitro embryo development and blastocyst hatching rates following vitrification of river buffalo embryos produced from oocytes recovered from slaughterhouse ovaries or live animals by ovum pick-up. Anim Reprod Sci. 2008;104:419–426. doi: 10.1016/j.anireprosci.2007.06.030. [DOI] [PubMed] [Google Scholar]
- 12.McGrath J, Solter D. Nuclear transplantation in the mouse embryo by microsurgery and cell fusion. Science. 1983;220:1300–1302. doi: 10.1126/science.6857250. [DOI] [PubMed] [Google Scholar]
- 13.Modliński JA. Preimplantation development of microsurgically obtained haploid and homozygous diploid mouse embryos and effects of pretreatment with Cytochalasin B on enucleated eggs. J Embryol Exp Morphol. 1980;60:153–161. [PubMed] [Google Scholar]
- 14.Paynter SJ, Cooper A, Fuller BJ, Shaw RW. Cryopreservation of bovine ovarian tissue: structural normality of follicles after thawing and culture in vitro. Cryobiology. 1999;38:301–309. doi: 10.1006/cryo.1999.2170. [DOI] [PubMed] [Google Scholar]
- 15.Pugh PA, Tervit HR, Niemann H. Effects of vitrification medium composition on the survival of bovine in vitro produced embryos, following in straw-dilution, in vitro and in vivo following transfer. Anim Reprod Sci. 2000;58:9–22. doi: 10.1016/s0378-4320(99)00087-1. [DOI] [PubMed] [Google Scholar]
- 16.Rall WF. Factors affecting the survival of mouse embryos cryopreserved by vitrification. Cryobiology. 1987;24:387–402. doi: 10.1016/0011-2240(87)90042-3. [DOI] [PubMed] [Google Scholar]
- 17.Rall WF, Fahy GM. Ice-free cryopreservation of mouse embryos at -196℃ by vitrification. Nature. 1985;313:573–575. doi: 10.1038/313573a0. [DOI] [PubMed] [Google Scholar]
- 18.Surani MAH, Barton SC, Norris ML. Development of reconstituted mouse eggs suggests imprinting of the genome during gametogenesis. Nature. 1984;308:548–550. doi: 10.1038/308548a0. [DOI] [PubMed] [Google Scholar]
- 19.Theodoropoulos PA, Gravanis A, Tsapara A, Margioris AN, Papadogiorgaki E, Galanopoulos V, Stournaras C. Cytochalasin B may shorten actin filaments by a mechanism independent of barbed end capping. Biochem Pharmacol. 1994;47:1875–1881. doi: 10.1016/0006-2952(94)90318-2. [DOI] [PubMed] [Google Scholar]
- 20.Vajta G, Rindom N, Peura TT, Holm P, Greve T, Callesen H. The effect of media, serum and temperature on in vitro survival of bovine blastocysts after open pulled straw (OPS) vitrification. Theriogenology. 1999;52:939–948. doi: 10.1016/S0093-691X(99)00184-3. [DOI] [PubMed] [Google Scholar]
- 21.Vicente JS, García-Ximénez F. Osmotic and cryoprotective effects of a mixture of DMSO and ethylene glycol on rabbit morulae. Theriogenology. 1994;42:1205–1215. doi: 10.1016/0093-691x(94)90869-9. [DOI] [PubMed] [Google Scholar]