Abstract
Invariant natural killer T (iNKT) cells develop in the thymus upon recognition of CD1d expressed on developing thymocytes. Although CD4 and CD8 coreceptors are not directly involved in the interaction between CD1d and the T cell receptors (TCRs) of iNKT cells, a conspicuous lack of CD8+ iNKT cells in mice raised the question of whether CD8+ iNKT cells are excluded due to negative selection during their thymic development, or if there is no lineage commitment for the development of murine CD8+ iNKT cells. To address this question, we analyzed iNKT cell-specific TCR Vα14+ transgenic mice, where the Vα14 transgene forces the generation of iNKT cells. This allows detailed study of the iNKT cell repertoire. We were able to identify CD8+ iNKT cells which respond to the NKT cell-specific glycolipid ligand α-galactosylceramide. Unlike conventional iNKT cells, CD8+ iNKT cells produce predominantly IFN-γ but not IL-4 upon antigen stimulation. We also confirmed the presence of CD8+ iNKT cells in wild type mice. Our results suggest that CD8+ NKT cells do exist in mice, although their population size is quite small. Their Th1-skewed phenotype might explain why the population size of this subtype needs to be controlled tightly.
Keywords: antigens, CD1d; CD8-positive T-lymphocytes; α-galactosylceramide; mice, transgenic; natural killer T-cells
Introduction
Natural killer T (NKT) cells are distinguished from conventional T lymphocytes by their function and T cell receptor (TCR) usage (Bendelac et al., 1997). They are divided by their TCR repertoires into 2 groups, Type I and Type II NKT cells. Both NKT cell types undergo CD1d-dependent development in the thymus and recognize glycolipid antigens presented on CD1d, a nonpolymorphic class Ib molecule. Murine type I NKT cells express an invariant TCR Vα14 chain and restricted TCR Vβ chains and are thus called invariant NKT (iNKT) cells, while Type II NKT cells express diverse Vα chains (Godfrey et al., 2004). As Type I but not type II NKT cells recognize α-galactosylceramide (α-GalCer), they can be detected by using α-galCer-loaded CD1d tetramer. Type I NKT (iNKT) cells rapidly produce large amounts of cytokines such as IFN-γ, IL-4 and IL-10 upon interaction with α-GalCer-loaded CD1d (Kronenberg and Gapin, 2002; Godfrey et al., 2004). Due to their ability to produce both Th1 and Th2-type cytokines, NKT cells are believed to play roles not only in tumor rejection and defense against infectious agents but also in the regulation of autoimmunity (Kronenberg and Gapin, 2002).
Like murine iNKT cells, human iNKT cells also use an invariant Vα24 chain paired with a Vβ11 chain and are activated by glycolipid antigens presented on CD1d. TCR Vα chains of human and murine iNKT cells are highly homologous (Lantz and Bendelac, 1994) and they can cross-react to each other's CD1d molecules (Borg et al., 2007). Despite the strong similarity in iNKT cells between the two species, it is generally accepted that mice do not have CD8+ iNKT cells (Bendelac et al., 1997; Kronenberg and Gapin, 2002), while human and macaque iNKT cells are constituted by CD4 single positive, CD8 single positive and CD4/CD8 double negative (DN) cells (Motsinger et al., 2003; Seino and Taniguchi, 2005). The conspicuous lack of CD8+ iNKT cells in mice might be the result of negative selection during thymic development, lack of lineage commitment for the development of murine CD8+ iNKT cells, or a small population size which might have been previously overlooked. A murine system with a large iNKT cell population would help address this question by allowing differentiation of each subtype of iNKT cells.
TCR Vα14 transgenic mice (Vα14+) strongly express an invariant TCR Vα14 chain used by invariant NKT cells, thus supporting the development of iNKT cells (Bendelac et al., 1996). Since Vα14+ mice produce a large number of iNKT cells, these mice are ideal to analyze the repertoire of iNKT cells.
Through careful analysis of iNKT cells in Vα14+ and wild type (WT) mice, we found, for the first time, that there is indeed lineage commitment toward CD8+ iNKT cells in the murine thymus, and that these cells showed a Th1-skewed phenotype by predominantly producing IFN-γ upon α-GalCer stimulation.
Results
The existence of CD8+ iNKT cells in Vα14+ mice
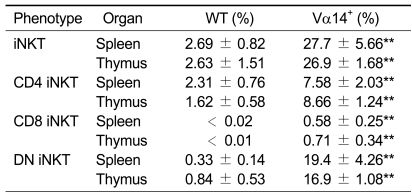
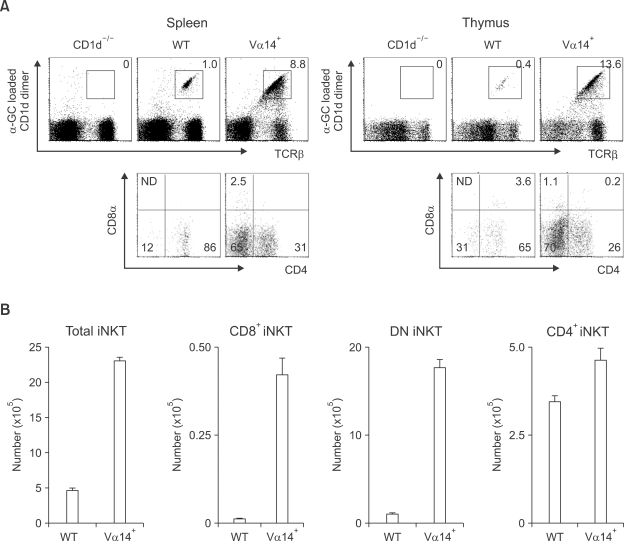
To determine the presence of CD8+ iNKT cells, we first stained splenocytes and thymocytes in CD1d-/-, WT and Vα14+ mice with α-GalCer-loaded CD1d dimer and mAbs against TCRβ, CD4 and CD8α. As previously reported (Bendelac et al., 1996), CD1d/α-GalCer dimer+ T cells (which indicate iNKT cells) in Vα14+ mice were greatly expanded compared to WT mice (8.8% vs. 1.0% in the spleen and 13.6% vs. 0.4% in the thymus). The number of iNKT cells in liver also expanded up to 30-40% of total liver T cells (data not shown). In accordance with the previous report, the relative population size of DN iNKT cells was increased, and the population with a CD4 phenotype was decreased in Vα14+ mice compared to WT mice (Table 1). Additionally we were able to clearly detect CD8+ iNKT cells in Vα14+ mice (2.5% in spleen and 1.1% in thymus, Figure 1A and 2% in liver, data not shown). This subtype of iNKT cells in WT mice was negligible and thus could be considered a non-specific staining. To enumerate the presence of CD8+ iNKT cells, we next determined the absolute number of each subtype of iNKT cells in WT and Vα14+ mice. As expected, while the absolute number of CD4+ iNKT cells was increased marginally, the absolute number of CD8+ iNKT cells along with DN iNKT cells in Vα14+ mice was greatly increased compared to WT mice (Figure 1B). Although it has been shown that the majority of iNKT cells in Vα14+ have the DN phenotype (Bendelac et al., 1996; Baldwin et al., 2005), the existence of CD8+ iNKT cells was not clearly appreciated since this subtype has not been recognized in WT mice and their population size even in Vα14+ mice was still quite small. However, by careful examination of iNKT cells in Vα14+ mice, we found that CD8+ iNKT cells do exist, and there was a substantial increase in the relative ratio and absolute number of CD8+ iNKT cells.
Table 1.
iNKT cells in WT and Vα14+ mice.a
aData show the percentage of each subset of iNKT cells among total splenic and thymic T cells of indicated mice. Data summarized the results of three independent experiments (n = 6-7). Data represent the mean values ± SD. iNKT cells indicated α-GalCer/CD1d dimer+, TCRβ+ cells, DN: CD4CD8 double negative.
Figure 1.
The Existence of CD8+ invariant NKT cells in Vα14+ mice. The size of the iNKT cell population (A) in the spleen and thymus from the indicated mice were analyzed as described in Methods. The numbers in the dot plot indicate the percentage of each cell population. Lower panels show the CD4 and CD8 phenotypes of TCRβ+CD1d/α-GalCer dimer+ cells in each mouse. (B) The absolute number of each iNKT cell subtype in the spleen. Numbers represent mean value ± SD per spleen. ND means not detected.
TCR Vβ usage of CD8+ iNKT cells
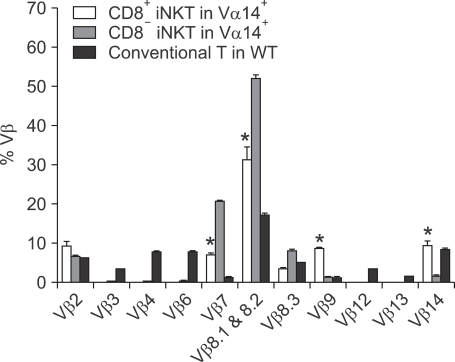
We next examined TCR Vβ usage of CD8+ iNKT cells, since it has been shown that type I iNKT cells express the Vα14 chain combined with restricted Vβ chains (mainly Vβ8, 7 and 2) (Lantz and Bendelac, 1994; Bendelac et al., 1996). We compared Vβ repertoires of CD8+ iNKT and CD8- (CD4+ and DN) iNKT cells. Both subtypes of iNKT cells utilized NKT cell-specific Vβ chains. This is quite different from conventional T cells in WT mice, which express a more even distribution from Vβ2 to Vβ14, without preference for specific Vβ chains. CD8+ NKT cells expressed a similar Vβ repertoire as CD8- iNKT cells in Vα14+ mice, which consisted of three major families: Vβ2, Vβ7 and Vβ8. However, Vβ8 and 7 usage was lower in CD8+ than CD8- iNKT cells, and Vβ9 and 14 usage in the spleen was remarkably increased in CD8+ cells (Figure 2). Interestingly, the increase of Vβ9 and 14 has also been reported previously in Vα14+ mice (Wei et al., 2006) (Figure 2). However, since the authors had not clearly distinguished the CD8+ iNKT cell population from the larger population of DN iNKT cells, it was considered to be an idiosyncrasy of the transgenic mice.
Figure 2.
TCR Vβ usage of CD8+ invariant NKT cells in Vα14+ mice. Splenocytes from the indicated mice were stained with CD1d/α-GalCer dimer and mAbs against TCRβ, CD8α and the Vβ TCR screening panel. The bar chart shows the mean percentage of each subtype of Vβ positive T cells (± SD) among the indicated cells. The percentages represent mean value ± SD per spleen.
Th1-skewed response of CD8+ iNKT cells against α-GalCer
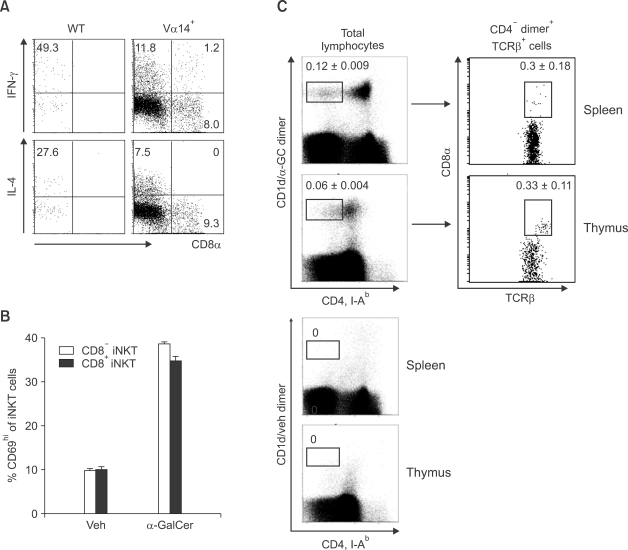
It is known that iNKT cells are activated by α- GalCer derived from a marine sponge (Bendelac et al., 1997). To analyze functional characteristics of CD8+ iNKT cells, we first examined cytokine production by iNKT cells upon α-GalCer stimulation. While CD8- iNKT cells from WT or Vα14+ mice showed the typical cytokine production profile of iNKT cells (both IL-4 and IFN-γ), CD8+ iNKT cells mainly produced IFN-γ but not IL-4 (Figure 3A). These data suggest that CD8+ iNKT cells probably have a more Th1-skewed phenotype than conventional CD8- iNKT cells.
Figure 3.
CD8+ invariant NKT cells in Vα14+ mice reveal a Th1-skewed phenotype. Splenocytes from the indicated mice were co-cultured with DCs pulsed with 100 ng/ml α-GalCer for 6 h. (A) Percentage of IFN-γ and IL-4-secreting cells among gated iNKT cells. (B) Percentage of CD69hi cells in the indicated iNKT cells. The bar chart represents mean value ± SD. (C) Splenocytes and thymocytes from WT mice (n = 3) were stained with CD1d/α-GalCer dimer along with mAbs against CD4, CD8, TCRβ and I-Ab. Vehicle loaded mCD1d-dimer was used as a negative control. After recording more than one million events, CD1d/α-GalCer dimer+ CD4-I-Ab- cells were plotted against TCRβ and CD8α expression. Antibody against I-Ab was used to exclude non-T cell populations in the analysis. The numbers on the figure represent mean percentage ± SD of marked cells from three WT mice. The data shown are representatives of two independent experiments with similar result.
Secondly, we examined the activation status of NKT cells by measuring CD69 on NKT cells upon α-GalCer stimulation, since it was previously reported that α-GalCer stimulation of NKT cells leads to significant up-regulation of CD69 on NKT cells (Testi et al., 1994; Vilanova et al., 1996). CD8+ iNKT cells up-regulated CD69 as much as CD8- iNKT cells upon α-GalCer stimulation, suggesting that CD8+ iNKT cells have the same activation potency as CD8- iNKT cells (Figure 3B).
Presence of CD8+ iNKT cells in the wild type mouse
Since we confirmed the presence of CD8+ iNKT cells in Vα14+ mice, we decided to re-evaluate WT mice for the presence of CD8+ iNKT cells, which might have been ignored in other studies because of their low numbers. We recorded a large number of events (more than 1.5 × 106 cells) in the FACS analysis for iNKT cells and found clearly distinguishable CD8+ iNKT cells in the spleen and thymus from wild type mice, although their population size was very limited (Figure 3C). These results disprove the idea that there is no lineage commitment to CD8+ NKT cells during their development.
Discussion
CD1d-dependent NKT cells appear to be CD4 single positive or CD4/CD8 DN in terms of their coreceptor expression, at least in mice. Because invariant TCRs of NKT cells recognize glycolipid antigens presented on CD1d, a MHC I-like molecule, coreceptor CD4 on the iNKT cells does not have any specific function for CD1d/antigen recognition, although CD4 and DN NKT cells can be functionally and developmentally distinctive (Gumperz et al., 2002; Benlagha et al., 2005). However, the conspicuous lack of CD1d-dependent CD8+ NKT cells despite of the presence CD4+ NKT cells in mice provoked the question of whether CD8+ NKT cells undergoing selection are too scarce to be detected or if NKT cells do not express CD8 for another reason. One common explanation is that CD8 provides too strong affinity for the recognition of CD1d/antigen by the TCRs of iNKT cells, which thus induces negative selection during thymic development of iNKT cells (Ashton-Rickardt et al., 1994; Bendelac et al., 1994; Kronenberg, 2005). However, there has been no experimental proof for the negative selection of this subtype of iNKT cells in mice. Although there were reports of the existence of CD8+ NK1.1+ αβ T cells in mice, these cells are not CD1d-dependent (Hammond et al., 1999; Emoto et al., 2000). Unlike CD1d-dependent NKT cells, which have TCR repertoires restricted to Vα14, murine CD8+NK1.1+TCRβ+ cells have variant Vα chains (Eberl et al., 1999). Further questions arise from the fact that there are CD1d-dependent CD8+ NKT cells in humans (Takahashi et al., 2002).
To address the question whether murine iNKT cells contain a CD8+ subtype, we used the Vα14+ mouse, which has forced expression of the invariant NKT cell receptor Vα14. Our data show that iNKT cells were expanded to up to 28% of total T cells in the spleen of Vα14+ mice, whereas iNKT cells normally constitute less than 2.7% of splenic T cells in the WT mouse. Thus the Vα14+ mouse is an optimal model for the discovery of a rare population of iNKT cells. We identified CD8+ iNKT cells in Vα14+ mice. Even more surprisingly, we were able to detect this population in the wild type mouse by recording more than one million events of FACS staining in both the spleen and thymus. Our data clearly show that CD8+ NKT cells exist as a subset of iNKT cells, although the population size is quite small. This disproves the idea that there is no lineage commitment to CD8+ NKT cells during their development.
CD1d-dependent iNKT cells, defined as α-GalCer/CD1d dimer+ cells, in Vα14+ mice showed a dramatic increase in the absolute number and relative ratio of CD8+ iNKT cells compared with WT mice. CD8+ iNKT cells from Vα14+ mice secreted only IFN-γ upon in vitro α-GalCer activation, unlike CD8- NKT cells from WT or Vα14+ mice, and they showed similar or slightly higher levels of CD69 up-regulation compared to CD8- iNKT cells. Consistent with these results, it has been reported that human CD4- NKT cells, especially CD8+ NKT cells, secrete IFN-γ but not IL-4 after activation (Gumperz et al., 2002; Lee et al., 2002). This study provides the first in vivo evidence that natural Vα14+ CD8+ iNKT cells indeed exist in the WT mouse, and they can be expanded by forced expression of the TCR Vα14+ transgene. Furthermore, we found that these CD8+ iNKT cells have extreme Th1-skewed phenotypes. This extreme phenotype of CD8+ iNKT cells might explain why the number of this subtype is kept low. In other word, it is possible that the interaction between TCR of murine CD8+ iNKT cells and mCD1d during thymic development is stronger than the interaction of human CD8+ iNKT cells and hCD1d. The intrinsic strong interaction affinity between murine CD8+ iNKT cells and CD1d may induce either negative selection of murine CD8+ iNKT cells or positive selection of Th1-skewed CD8+ iNKT cells (though the frequency is low) depending on the natural antigens presented by mCD1d.
It remains to be determined whether there is not enough positive selection for CD8+ iNKT cells or if there is strong and highly specific negative selection for CD8+ iNKT cells by CD1d. It will be of great interest to clarify which cells are responsible for the positive (or negative) selection of CD8+ iNKT cells.
Methods
Mice
C57BL/6 wild type (WT) mice were purchased from The Jackson Laboratory. CD1d-/- and TCR Vα14+ transgenic (Vα14+) mice were provided by Dr. A. Bendelac (University of Chicago, Chicago, IL).
All mice used in this study had a C57BL/6 background, and were raised in a specific pathogen-free environment at Korea University and were used at 6-8 wk of age unless otherwise specified. The experimental protocols adopted in this study were approved by the Institutional Animal Care and Use Committee of Korea University.
FACS analysis
Cells were pre-incubated with an anti-FcγRII/III mAb (2.4G2) and then labeled for 30 min with the appropriate mAbs. The following mAbs were used: fluorescein isothiocyanate (FITC) conjugated anti-CD4 (clone RM4-5), cychrome (CYC) conjugated anti-CD8α (clone 53-6.7), allophycocyanin (APC) conjugated anti-TCRβ (clone H57), APC conjugated anti-IL-4 (clone 11B11) and -IFN-γ (XMG1.2) (all from BD PharMingen).
CD1d Dimer X was purchased from BD Biosciences and α-GalCer was provided by Dr. Bendelac. CD1d/α-GalCer dimer was prepared according to the manufacturer's protocol.
TCR Vβ usage by NKT cells was analyzed among CD1d/α-GalCer-dimer+ cells with a mouse Vβ TCR screening panel (BD biosciences). The stained cells were then analyzed on a FACSCalibur using CellQuest software (both BD Biosciences). Figures with panel sets depict analyses from the same experiment to allow for direct comparison of fluorescence intensity.
DC sorting
Splenic DCs were obtained from naive mice via MACS using magnetic bead-conjugated anti-CD11c mAb (Miltenyi Biotec) in accordance with the manufacturer's instructions.
NKT cell activation
The cell culture medium used in this study was RPMI 1640 complete medium (Life Technologies, Grand Island, NY) containing 10% heat-inactivated FBS (Hyclone, Logan, UT) supplemented with 2 mM sodium bicarbonate, 2 mM L-glutamine, 5µM β-mercaptoethanol, 10 µg/ml gentamicin, 50 U/ml penicillin and 50µg/ml streptomycin (all from Life Technologies). For NKT cell activation assays, DCs were used as antigen presenting cells. DCs were pulsed for 4 h at 37℃ with α-GalCer or vehicle (Veh) and then extensively washed before being plated at 5 × 104 cells/well. Whole splenocytes were seeded at 5 × 105 cells/well in a 96-well plate and then cultured for 6 h with Golgi stop (BD PharMingen).
Analysis of intracellular cytokine production
To determine intracellular cytokine levels, cells cultured for 6 h with Golgi stop (BD PharMingen) were initially stained with the appropriate mAbs, fixed and permeabilized with Cytofix/Cytoperm solution (BD PharMingen) and finally stained with APC-conjugated anti-IL-4 or anti-IFN-γ mAbs for 45 min on ice. The percentage of cells expressing intracellular IL-4 or IFN-γ was determined via flow cytometry (FACSCalibur).
Statistical analysis
Student's t tests were used to determine statistical significance between the two groups. Throughout the text, figures, table and legends, the following symbols were used to denote statistical significance:**, P ≤ 0.0001 or P ≤ 0.001;*, P ≤ 0.01.
Acknowledgments
The authors thank Dr. A. Bendelac for the generous gift of transgenic animals and α-GalCer. This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) (20090081330) and Korea Biotech R&D Group of Next-generation growth engine project (F104AC010002-08A-0301-00230) funded by the Ministry of Education, Science and Technology.
Abbreviations
- α-GalCer
α-galactosylceramide
- DN
CD4/CD8 double negative
- DP
CD4/CD8 double positive
- Veh
vehicle
References
- 1.Ashton-Rickardt PG, Bandeira A, Delaney JR, Van Kaer L, Pircher HP, Zinkernagel RM, Tonegawa S. Evidence for a differential avidity model of T cell selection in the thymus. Cell. 1994;76:651–663. doi: 10.1016/0092-8674(94)90505-3. [DOI] [PubMed] [Google Scholar]
- 2.Baldwin TA, Sandau MM, Jameson SC, Hogquist KA. The timing of TCR alpha expression critically influences T cell development and selection. J Exp Med. 2005;202:111–121. doi: 10.1084/jem.20050359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bendelac A, Killeen N, Littman DR, Schwartz RH. A subset of CD4+ thymocytes selected by MHC class I molecules. Science. 1994;263:1774–1778. doi: 10.1126/science.7907820. [DOI] [PubMed] [Google Scholar]
- 4.Bendelac A, Hunziker RD, Lantz O. Increased interleukin 4 and immunoglobulin E production in transgenic mice overexpressing NK1 T cells. J Exp Med. 1996;184:1285–1293. doi: 10.1084/jem.184.4.1285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bendelac A, Rivera MN, Park SH, Roark JH. Mouse CD1-specific NK1 T cells: development, specificity, and function. Annu Rev Immunol. 1997;15:535–562. doi: 10.1146/annurev.immunol.15.1.535. [DOI] [PubMed] [Google Scholar]
- 6.Benlagha K, Wei DG, Veiga J, Teyton L, Bendelac A. Characterization of the early stages of thymic NKT cell development. J Exp Med. 2005;202:485–492. doi: 10.1084/jem.20050456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Borg NA, Wun KS, Kjer-Nielsen L, Wilce MC, Pellicci DG, Koh R, Besra GS, Bharadwaj M, Godfrey DI, McCluskey J, Rossjohn J. CD1d-lipid-antigen recognition by the semiinvariant NKT T-cell receptor. Nature. 2007;448:44–49. doi: 10.1038/nature05907. [DOI] [PubMed] [Google Scholar]
- 8.Eberl G, Lees R, Smiley ST, Taniguchi M, Grusby MJ, MacDonald HR. Tissue-specific segregation of CD1d-dependent and CD1d-independent NK T cells. J Immunol. 1999;162:6410–6419. [PubMed] [Google Scholar]
- 9.Emoto M, Zerrahn J, Miyamoto M, Perarnau B, Kaufmann SH, Kaufmann SH. Phenotypic characterization of CD8(+)NKT cells. Eur J Immunol. 2000;30:2300–2311. doi: 10.1002/1521-4141(2000)30:8<2300::AID-IMMU2300>3.0.CO;2-2. [DOI] [PubMed] [Google Scholar]
- 10.Godfrey DI, MacDonald HR, Kronenberg M, Smyth MJ, Van Kaer L. NKT cells: what's in a name? Nat Rev Immunol. 2004;4:231–237. doi: 10.1038/nri1309. [DOI] [PubMed] [Google Scholar]
- 11.Gumperz JE, Miyake S, Yamamura T, Brenner MB. Functionally distinct subsets of CD1d-restricted natural killer T cells revealed by CD1d tetramer staining. J Exp Med. 2002;195:625–636. doi: 10.1084/jem.20011786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hammond KJ, Pelikan SB, Crowe NY, Randle-Barrett E, Nakayama T, Taniguchi M, Smyth MJ, van Driel IR, Scollay R, Baxter AG, Godfrey DI. NKT cells are phenotypically and functionally diverse. Eur J Immunol. 1999;29:3768–3781. doi: 10.1002/(SICI)1521-4141(199911)29:11<3768::AID-IMMU3768>3.0.CO;2-G. [DOI] [PubMed] [Google Scholar]
- 13.Kronenberg M. Toward an understanding of NKT cell biology: progress and paradoxes. Annu Rev Immunol. 2005;23:877–900. doi: 10.1146/annurev.immunol.23.021704.115742. [DOI] [PubMed] [Google Scholar]
- 14.Kronenberg M, Gapin L. The unconventional lifestyle of NKT cells. Nat Rev Immunol. 2002;2:557–568. doi: 10.1038/nri854. [DOI] [PubMed] [Google Scholar]
- 15.Lantz O, Bendelac A. An invariant T cell receptor alpha chain is used by a unique subset of major histocompatibility complex class I-specific CD4+ and CD4-8- T cells in mice and humans. J Exp Med. 1994;180:1097–1106. doi: 10.1084/jem.180.3.1097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lee PT, Benlagha K, Teyton L, Bendelac A. Distinct functional lineages of human V(alpha)24 natural killer T cells. J Exp Med. 2002;195:637–641. doi: 10.1084/jem.20011908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Motsinger A, Azimzadeh A, Stanic AK, Johnson RP, Van Kaer L, Joyce S, Unutmaz D. Identification and simian immunodeficiency virus infection of CD1d-restricted macaque natural killer T cells. J Virol. 2003;77:8153–8158. doi: 10.1128/JVI.77.14.8153-8158.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Seino K, Taniguchi M. Functionally distinct NKT cell subsets and subtypes. J Exp Med. 2005;202:1623–1626. doi: 10.1084/jem.20051600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Takahashi T, Chiba S, Nieda M, Azuma T, Ishihara S, Shibata Y, Juji T, Hirai H. Cutting edge: analysis of human V alpha 24+CD8+ NK T cells activated by alpha-galactosylceramide-pulsed monocyte-derived dendritic cells. J Immunol. 2002;168:3140–3144. doi: 10.4049/jimmunol.168.7.3140. [DOI] [PubMed] [Google Scholar]
- 20.Testi R, D'Ambrosio D, De Maria R, Santoni A. The CD69 receptor: a multipurpose cell-surface trigger for hematopoietic cells. Immunol Today. 1994;15:479–483. doi: 10.1016/0167-5699(94)90193-7. [DOI] [PubMed] [Google Scholar]
- 21.Vilanova M, Tavares D, Ferreira P, Oliveira L, Nobrega A, Appelberg R, Arala-Chaves M. Role of monocytes in the up-regulation of the early activation marker CD69 on B and T murine lymphocytes induced by microbial mitogens. Scand J Immunol. 1996;43:155–163. doi: 10.1046/j.1365-3083.1996.d01-25.x. [DOI] [PubMed] [Google Scholar]
- 22.Wei DG, Curran SA, Savage PB, Teyton L, Bendelac A. Mechanisms imposing the Vbeta bias of Valpha14 natural killer T cells and consequences for microbial glycolipid recognition. J Exp Med. 2006;203:1197–1207. doi: 10.1084/jem.20060418. [DOI] [PMC free article] [PubMed] [Google Scholar]