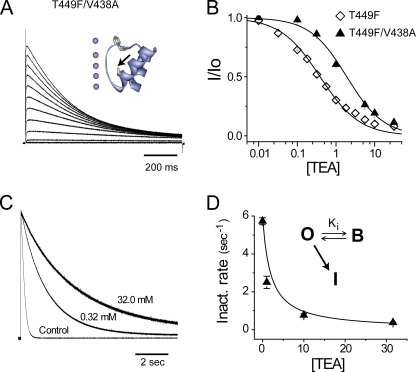
Figure 2.
The V438A mutation enhances slow inactivation, lowers TEA affinity, and restores foot-in-the-door blockade in Shaker T449F channels. (A) Potassium currents from the double V438A/T449F mutant in response to 1-s depolarizations from −80 to +100 mV in 20-mV steps. The inset shows the structure of KcsA (PDB, 1ZW1) with the side chains shown for the aromatic Y82 and E71A (highlighted by arrow). The corresponding Shaker residues are 449 and 438, respectively. (B) Inhibition of the mutants by extracellular TEA. Smooth lines represent standard binding isotherms with inhibitory constants of 0.39 and 1.97 mM for T449F and V438A/T449F, respectively. (C) Normalized outward potassium currents from V438A/T449F channels in increasing TEA concentrations. The V438A mutation speeds slow inactivation from τinact = 9.9 ± 0.4 s to τinact = 0.18 ± .01 s at +40 mV for T449F (n = 14) and V438A/T449F (n = 5) channels, respectively, with the number of cells in parentheses. (D) The speeding can also be explained by a three-state model in which inactivation only occurs from the open state. The smooth line through the data points is a theory curve generated from known values. See equations in the legend of Fig. 1, with kBI = 0.