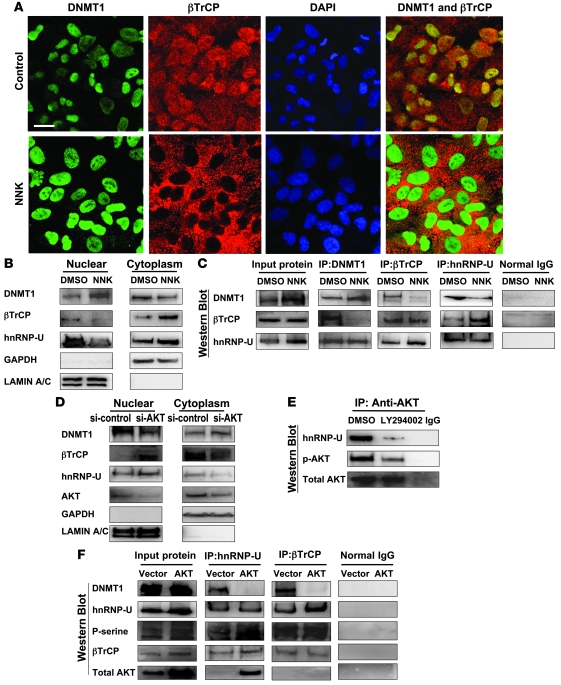
Figure 5. NNK enhances hnRNP-U/βTrCP translocation to the cytoplasm and induces DNMT1 accumulation in the nucleus, but this effect is attenuated by AKT inhibition.
(A) Immunofluorescence confocal microscopy to localize DNMT1 (green), βTrCP (red), and nucleus (blue; DAPI). The merged figures showed colocalization of DNMT1 with βTrCP protein in A549 cells treated with DMSO control. Mutually exclusive localization of DNMT1 and βTrCP was observed in cells treated with NNK. Original magnification, ×630. Scale bar: 20 μm. (B) Cell fractionation assay to analyze distribution of nuclear and cytoplasmic proteins, using anti-DNMT1, anti-βTrCP, anti–hnRNP-U, and anti-GSK3βSer9 antibodies. Anti-GAPDH and anti–lamin A/C antibodies were used as cytoplasmic and nuclear protein markers, respectively. The distribution of βTrCP and hnRNP-U proteins was contrary to that of DNMT1 protein. (C) DNMT1 and βTrCP interaction was interrupted by NNK treatment. In contrast, βTrCP and hnRNP interaction was increased by NNK treatment. (D) Cell fractionation assay was performed in A549 cells with or without AKT siRNA. Inhibition of AKT induced both hnRNP-U and βTrCP proteins predominantly located in the nucleus, where DNMT1 protein level decreased. (E) IP assay using anti-AKT antibody showed that hnRNP formed a complex with AKT, and the interaction was attenuated by LY294002. (F) An increase of hnRNP-U phosphorylation was mediated by exogenous AKT, which increased the interaction between hnRNP-U and βTrCP. In addition, AKT decreased the interaction of DNMT1 with hnRNP-U and βTrCP.