Abstract
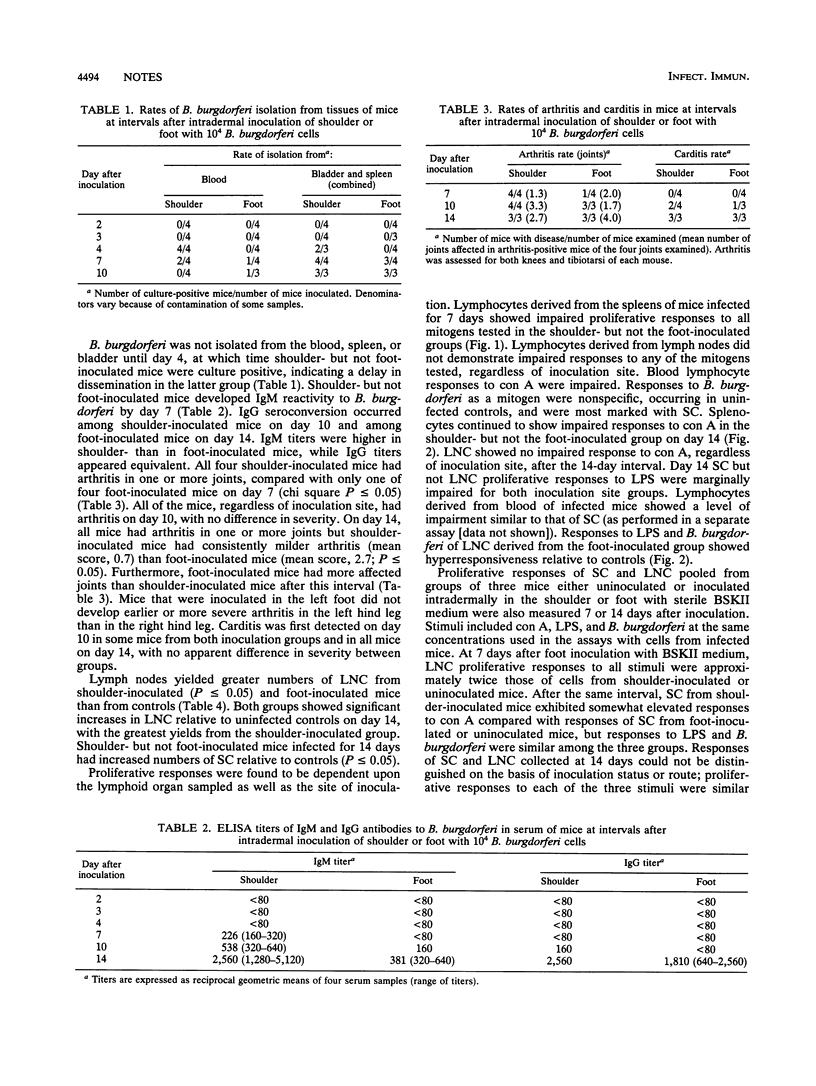
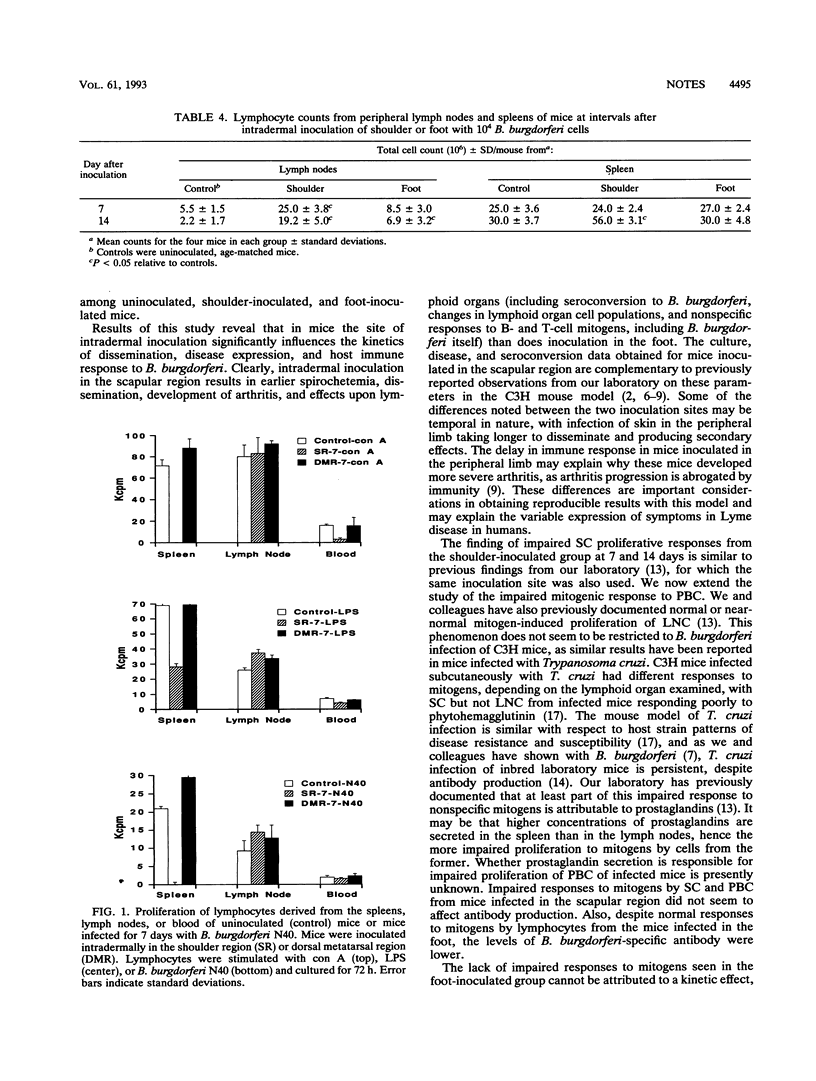
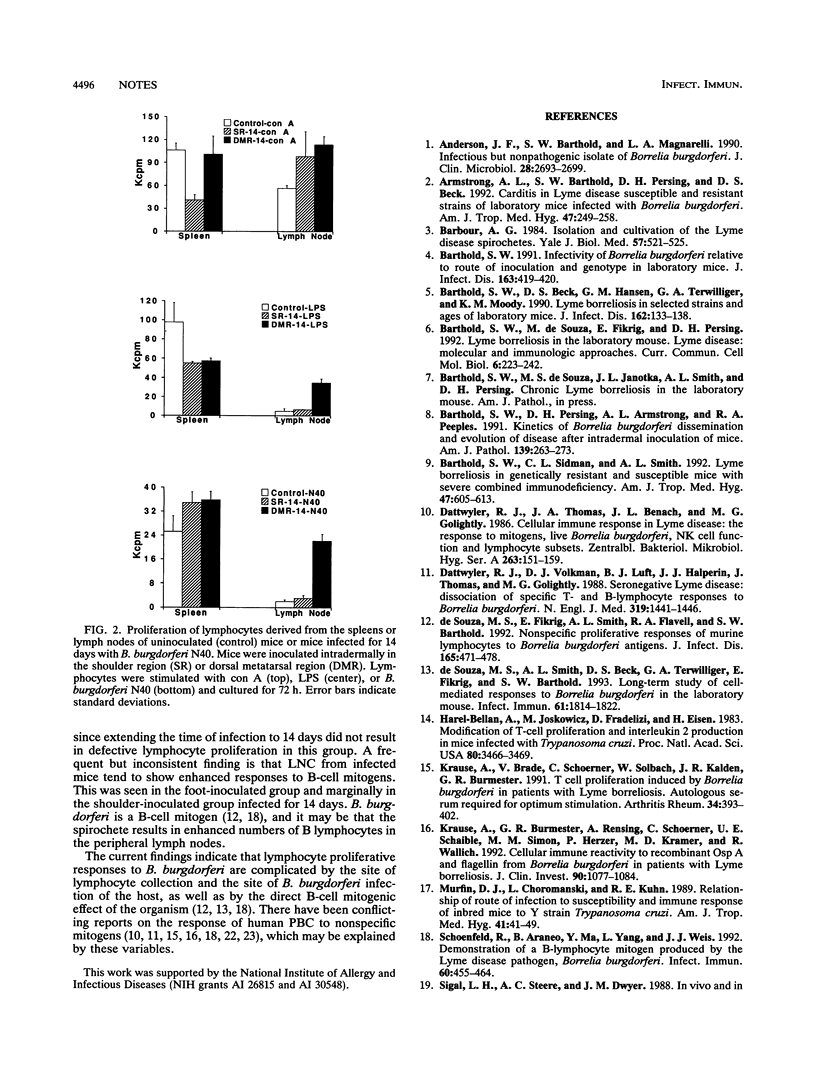
C3H/He mice inoculated intradermally at one of two sites with Borrelia burgdorferi responded differently to infection. Shoulder-inoculated mice developed spirochetemia, B. burgdorferi-specific antibody, and arthritis earlier than foot-inoculated mice. Lymphocyte populations derived from spleen tissue were elevated in the shoulder- but not the foot-inoculated mice, and those from lymph nodes were increased in both groups. Lymphocytes derived from blood and spleen tissue showed impaired proliferative responses to all mitogens for shoulder-inoculated mice only, whereas proliferation of lymph node cells was not affected, regardless of route. These results demonstrate that the site of initial B. burgdorferi inoculation is an important determinant in the pathogenesis of B. burgdorferi infection.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson J. F., Barthold S. W., Magnarelli L. A. Infectious but nonpathogenic isolate of Borrelia burgdorferi. J Clin Microbiol. 1990 Dec;28(12):2693–2699. doi: 10.1128/jcm.28.12.2693-2699.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armstrong A. L., Barthold S. W., Persing D. H., Beck D. S. Carditis in Lyme disease susceptible and resistant strains of laboratory mice infected with Borrelia burgdorferi. Am J Trop Med Hyg. 1992 Aug;47(2):249–258. doi: 10.4269/ajtmh.1992.47.249. [DOI] [PubMed] [Google Scholar]
- Barbour A. G. Isolation and cultivation of Lyme disease spirochetes. Yale J Biol Med. 1984 Jul-Aug;57(4):521–525. [PMC free article] [PubMed] [Google Scholar]
- Barthold S. W., Beck D. S., Hansen G. M., Terwilliger G. A., Moody K. D. Lyme borreliosis in selected strains and ages of laboratory mice. J Infect Dis. 1990 Jul;162(1):133–138. doi: 10.1093/infdis/162.1.133. [DOI] [PubMed] [Google Scholar]
- Barthold S. W. Infectivity of Borrelia burgdorferi relative to route of inoculation and genotype in laboratory mice. J Infect Dis. 1991 Feb;163(2):419–420. doi: 10.1093/infdis/163.2.419. [DOI] [PubMed] [Google Scholar]
- Barthold S. W., Persing D. H., Armstrong A. L., Peeples R. A. Kinetics of Borrelia burgdorferi dissemination and evolution of disease after intradermal inoculation of mice. Am J Pathol. 1991 Aug;139(2):263–273. [PMC free article] [PubMed] [Google Scholar]
- Barthold S. W., Sidman C. L., Smith A. L. Lyme borreliosis in genetically resistant and susceptible mice with severe combined immunodeficiency. Am J Trop Med Hyg. 1992 Nov;47(5):605–613. doi: 10.4269/ajtmh.1992.47.605. [DOI] [PubMed] [Google Scholar]
- Dattwyler R. J., Thomas J. A., Benach J. L., Golightly M. G. Cellular immune response in Lyme disease: the response to mitogens, live Borrelia burgdorferi, NK cell function and lymphocyte subsets. Zentralbl Bakteriol Mikrobiol Hyg A. 1986 Dec;263(1-2):151–159. doi: 10.1016/s0176-6724(86)80118-3. [DOI] [PubMed] [Google Scholar]
- Dattwyler R. J., Volkman D. J., Luft B. J., Halperin J. J., Thomas J., Golightly M. G. Seronegative Lyme disease. Dissociation of specific T- and B-lymphocyte responses to Borrelia burgdorferi. N Engl J Med. 1988 Dec 1;319(22):1441–1446. doi: 10.1056/NEJM198812013192203. [DOI] [PubMed] [Google Scholar]
- Harel-Bellan A., Joskowicz M., Fradelizi D., Eisen H. Modification of T-cell proliferation and interleukin 2 production in mice infected with Trypanosoma cruzi. Proc Natl Acad Sci U S A. 1983 Jun;80(11):3466–3469. doi: 10.1073/pnas.80.11.3466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krause A., Brade V., Schoerner C., Solbach W., Kalden J. R., Burmester G. R. T cell proliferation induced by Borrelia burgdorferi in patients with Lyme borreliosis. Autologous serum required for optimum stimulation. Arthritis Rheum. 1991 Apr;34(4):393–402. doi: 10.1002/art.1780340404. [DOI] [PubMed] [Google Scholar]
- Krause A., Burmester G. R., Rensing A., Schoerner C., Schaible U. E., Simon M. M., Herzer P., Kramer M. D., Wallich R. Cellular immune reactivity to recombinant OspA and flagellin from Borrelia burgdorferi in patients with Lyme borreliosis. Complexity of humoral and cellular immune responses. J Clin Invest. 1992 Sep;90(3):1077–1084. doi: 10.1172/JCI115923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murfin D. J., Choromanski L., Kuhn R. E. Relationship of route of infection to susceptibility and immune response of inbred mice to Y strain Trypanosoma cruzi. Am J Trop Med Hyg. 1989 Jul;41(1):41–49. [PubMed] [Google Scholar]
- Schoenfeld R., Araneo B., Ma Y., Yang L. M., Weis J. J. Demonstration of a B-lymphocyte mitogen produced by the Lyme disease pathogen, Borrelia burgdorferi. Infect Immun. 1992 Feb;60(2):455–464. doi: 10.1128/iai.60.2.455-464.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steere A. C., Dwyer E., Winchester R. Association of chronic Lyme arthritis with HLA-DR4 and HLA-DR2 alleles. N Engl J Med. 1990 Jul 26;323(4):219–223. doi: 10.1056/NEJM199007263230402. [DOI] [PubMed] [Google Scholar]
- de Souza M. S., Fikrig E., Smith A. L., Flavell R. A., Barthold S. W. Nonspecific proliferative responses of murine lymphocytes to Borrelia burgdorferi antigens. J Infect Dis. 1992 Mar;165(3):471–478. doi: 10.1093/infdis/165.3.471. [DOI] [PubMed] [Google Scholar]
- de Souza M. S., Smith A. L., Beck D. S., Terwilliger G. A., Fikrig E., Barthold S. W. Long-term study of cell-mediated responses to Borrelia burgdorferi in the laboratory mouse. Infect Immun. 1993 May;61(5):1814–1822. doi: 10.1128/iai.61.5.1814-1822.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]



