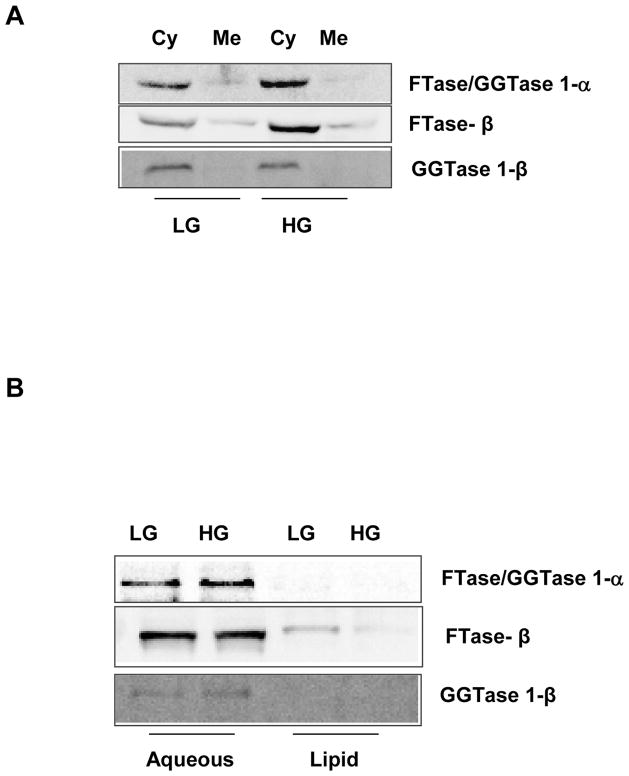
Figure 1.
Figure 1A. Sub-cellular distribution of FTase and GGTase subunits in INS-832/13 following exposure to basal or stimulatory glucose concentrations
INS-832/13 cells were cultured overnight in low glucose-low serum media. Cells were further incubated in presence of either basal [2.5 mM; LG] or high glucose [20 mM; HG] for 45 min. Homogenates of these cells were centrifuged at 100,000g for 90 min and the total cytosolic [Cy] and membrane [Me] fractions were separated by SDS-PAGE, transferred to a membrane and probed with corresponding antibodies for detection of FTase/GGTase 1- α, FTase β and GGTase 1-β subunits. Data are representative of two separate experiments yielding identical results.
Figure 1B. Phase partition of prenyltransferase subunits in INS-832/13 cells following exposure to basal or stimulatory glucose concentrations
INS-832/13 cells were cultured overnight in low glucose-low serum media. Cells were further incubated in presence of either basal glucose [2.5 mM; LG) or stimulatory glucose [20 mM; HG] for 45 min. Homogenate proteins were partitioned into hydrophobic and hydrophilic compartment using Triton X-114 partition method as described in Methods section. Fractions were separated by SDS-PAGE, transferred to a membrane and probed with corresponding antibodies for detection of FTase/GGTase 1- α, FTase β and GGTase 1-β subunits. Data are representative of two separate experiments yielding identical results.