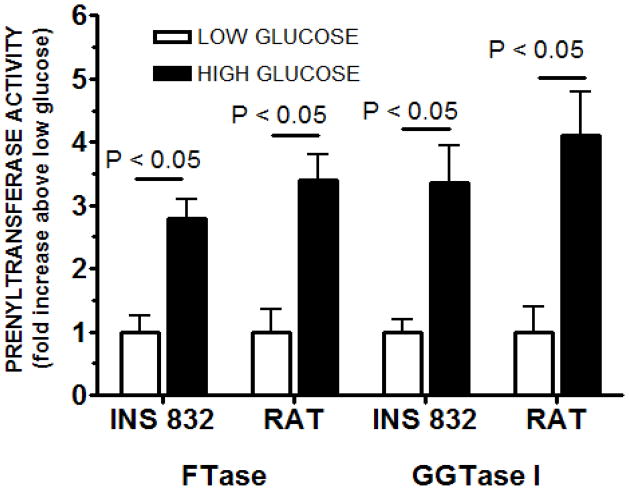
Figure 3. FTase and GGTase-I activities in INS 832/13 cells or isolated rat islets following exposure to basal [2.5 mM] or stimulatory [20 mM] glucose.
FTase and GGTase-I activities were determined using a modified filtration assay as previously described in Methods. Briefly, cells were grown to confluence and treated with low [2.5 mM] or stimulatory [20 mM] glucose for 45min. Subsequently, cells were lysed, sonicated and centrifuged 10,000 × g. Protein concentrations were determined from lysate supernatants. The in vitro filtration assay was initiated by adding a 5-μl aliquot of diluted and normalized extract to 45 μl of reaction assay solution, which contained tritiated farnesyl- or geranylgeranyl pyrophosphate and allowed to incubate for 30 minutes. The assay was stopped using ice-cold 1 M HCl in ethanol. Reactions solutions were individually filtered through Whatman GF/C glass-fiber filters, air dried, placed in scintillation fluid and quantified by scintillation spectrometry. Activities were first calculated in dpm/μg protein/unit time and then converted to relative amounts. FTase and GGTase-I activities are expressed as fold increase above low glucose level in the same treatment group and represent the mean ± SEM (n = 6 for each treatment).