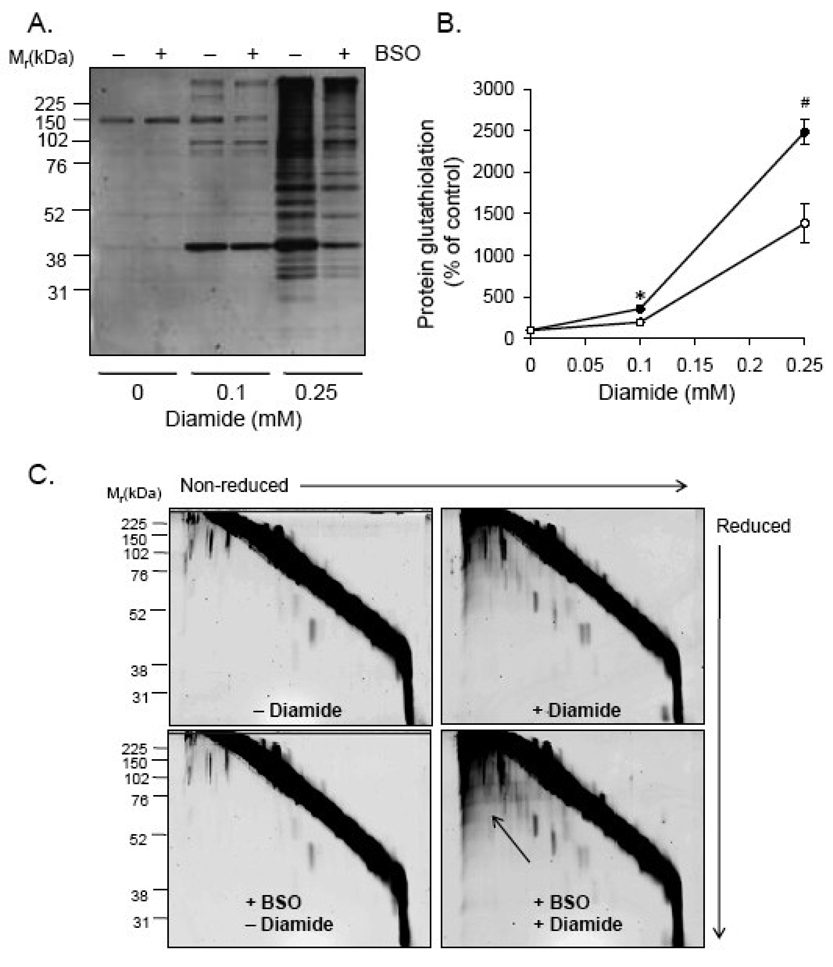
Fig. 5. Glutathione depletion decreases diamide-induced protein-glutathione adduct formation.
Protein-glutathione adducts and protein-protein dithiol formation in glutathione-depleted cells treated with diamide. (A) Protein-glutathione adducts in cells depleted of glutathione: Cells were treated with 100 µM BSO for 24 h followed by diamide (0–0.25 mM) for 40 min. Equal amounts of protein (10 µg) from cell lysates were separated by SDS-PAGE, and glutathiolated proteins were detected by chemifluorescent Western blotting with anti-PSSG antibodies. (B) Group data from panel A. n = 3 per group; *p < 0.05 vs. glutathione-depleted cells treated with 0.1 mM diamide; #p<0.05 vs. glutathione-depleted cells treated with 0.25 mM diamide. (C) Diagonal electrophoresis gels in control cells (upper left panel), cells treated with 0.25 mM diamide (upper right panel), glutathione-depleted cells (lower left panel), and glutathione-depleted cells treated with 0.25 mM diamide. The arrow points to the area of the gel showing increased protein-protein dithiol formation.