Abstract
Thirty-days-old female rats were chronically exposed, for 60 days, to 1or 2 mg/kg/day of mercuric chloride or an equivalent volume of water, via gavage. At 90 days of age they were mated with unexposed males. At approximately day 13 of gestation necropsies were performed on the females. Data were collected on the number of implantations and non-viable implantations in the uterus. No physical signs of Hg intoxication were seen except in weight gain. There were significantly fewer implantations in the high HgCl2 group, with significantly more non-viable implantations in the low and high HgCl2 groups, compared to controls. Lower levels of progesterone and higher levels of pituitary luteinizing hormone (LH) were found in the high HgCl2 group compared to controls, whereas pituitary follicle stimulating hormone levels (FSH), while not significant, showed a dose–response relationship to HgCl2 levels. No difference was found in the number of corpora lutea. The experiment indicated low level chronic ingestion of mercuric chloride, in female rats, while not effecting ovulation, produced disruption of implantation and fetal viability. Lower progesterone levels, higher LH, and possibly FSH levels, indicate that mercuric chloride may have a disruptive effect in the corpora lutea which manifests itself after ovulation.
Keywords: Inorganic mercury, Reproduction, Female, Fertility, Implantation, Rats
1. Introduction
Mercury (Hg) is a naturally occurring metallic element that is ubiquitous throughout the environment. It is present in several inorganic forms including metallic (Hg0), mercurous (Hg+), and mercuric (Hg2+) valence states (Daintith, 1996). Metallic Hg is found in such household products as barometers, blood pressure instruments, and switches in automobiles (Davis et al., 2001). It is also found in older products such as switches in children’s shoes that light up, thermometers, as well as being used frequently in dental amalgams. Mercuric Hg is found influorescent lights, (Davis et al., 2001), including the newer type that are replacing incandescent light bulbs.
Research on the reproductive effects of inorganic Hg is sparse although several studies in the 1980s documented reproductive problems of women working in Hg contaminated environments. These included reproductive failures, menstrual cycle disorders, primary subfecundity, and adverse pregnancy outcomes (De Rosis et al., 1985; Sikorski et al., 1985), however, a relationship with the level of Hg exposure was not always clear (De Rosis et al., 1985). A more recent study involving females in an Hg exposed work environment also revealed a higher frequency of adverse reproductive outcomes, especially congenital anomalies, (Elghany et al., 1997). Ovarian function in women can be compromised by exposure to toxic chemicals in the environment through direct effects on the ovaries or indirectly via the hypothalamus–pituitary–ovarian axis (Hoyer, 2005). Inorganic Hg in placental cord blood can be 20– 65% higher than in maternal venous blood (Grandjean et al., 1992).
In animal models, exposure to inorganic Hg has been shown to cause anovulation in hamsters (Davis et al., 2001), and to show a prolongation of diestrus of up to 10 days in rats, with evidence of Hg in the ovaries (Stadnicka, 1980). A previous study in our laboratory in which both male and female rats were exposed to mercuric chloride (HgCl2) and then mated together, revealed significant differences in implantation efficiency, fertility, live births, and litter size (Atkinson et al., 2001), however, because both males and females were exposed, it is hard to differentiate what proportion the males and females contributed to the differences in fertility and reproductive variables.
The evidence of fertility problems, in females, in Hg exposed work environments, together with the animal-based studies, demonstrates that inorganic Hg has adverse effects in human and rodent female reproduction. However, a study is needed to ascertain the effects of inorganic Hg on fertility, from chronic exposure, in concentrations that do not cause any physical signs, to see if these concentrations affect reproduction adversely.
2. Materials and methods
2.1. Subjects
Seventeen pregnant Sprague–Dawley rats were purchased from Harlan Sprague-Dawley (Indianapolis, IN) at approximately gestation day (GD) 11. The animals were housed in individual polycarbonate cages on beta chip bedding (Northeastern Products, Warrensburg, NY), given access to Purina certified rodent chow 5001 (Purina Mills, St. Louis, MO) and water ad libitum, and allowed to come to term. Room conditions were controlled at 22°–23 °C ambient temperature, 50–60% relative humidity and 12 h light/dark cycle. All conditions conformed to the regulations of the Tuskegee University Animal Care and Use Committee.
At postnatal day (PD) 21 60 female offspring were separated and randomly assigned to one of three experimental groups, control, low Hg and high Hg. They were housed two animals per cage with cage mates belonging to the same experimental group. All subjects were given free access to food and water. At PD 30, subjects in the Hg groups started exposure to a solution of mercuric chloride (HgCl2) in concentrations of 1.0 (low Hg), or 2.0 mg/kg/day (high Hg). All subjects (including controls) were weighed three times per week during exposure, which lasted for 60 days.
2.2. Preparation and administration of HgCl2 solutions
Concentrations of 1.0 mg/ml and 0.5 mg/ml HgCl2 were prepared. The 1.0 mg/ml solution was prepared using 1 g of mercuric chloride (HgCl2; Sigma–Aldrich Inc, St. Louis, MO) added to a solution of 998.5 ml of de-ionized H2O plus 1.5 ml of HNO3 (nitric acid). The 0.5 mg/ml solution was similarly prepared with 0.5 g of HgCl2. The amount of HgCl2 administered each day, to each subject, was calculated based upon its weight and exposure group. The control group received an equivalent volume of de-ionized H2O (with no HNO3 added). Each subject was weighed three times per week and the dose was adjusted accordingly. The solution was administered seven days a week via gavage until the subjects reached 90 days of age.
2.3. Breeding protocol
Exposure to HgCl2 ceased after 60 days, at which time 30 subjects (10 from each group) were transferred to hanging cages where they were mated with adult unexposed males. Females were examined each morning for vaginal plugs. If a plug was found, presence of sperm was confirmed by vaginal wash and the subject was transferred to its original cage. An unmated subject was transferred to the hanging cage with the same male and the procedure repeated. If no vaginal plug was observed within 10 days of mating the male was removed and replaced with a new male that had successfully mated previously with a female. This protocol was observed in order to preclude the possibility that a male may be infertile. The exposed females were euthanized at approximately gestation day (GD) 13, calculated from the day the presence of plug/sperm was confirmed.
2.4. Procedures
Subjects were anaesthetized by CO2 inhalation, weighed and blood was collected, via cardiac puncture. Euthanasia was then completed with CO2 inhalation. Blood samples were centrifuged at 3000 rpm for 10 min at 4 °C and plasma was stored at −70 °C until processed for progesterone levels. Ovaries were removed, fixed in 1% formaldehyde and examined for number of corpora lutea. The pituitary glands were also collected and stored in the same manner for future luteinizing hormone (LH) and follicle stimulating hormones (FSH) analysis.
2.5. Luteinizing hormone and follicle stimulating hormone analysis
After collection, the weight of each pituitary gland was recorded. At analysis each gland was placed in 1 ml homogenization buffer (phosphate-buffered saline containing 2.5 M urea; pH 7.4). It was then homogenized with 20 strokes in a dounce glass homogenizer followed by a freeze–thaw (−20 °C). Samples were sonicated for 60 s, followed by centrifugation at 16,000 rpm for 15 min at 4 °C. The supernate was decanted and frozen (−20 °C) until assayed for luteinizing hormone (LH) and follicle stimulating hormone (FSH). Pituitary gland content of LH was measured by radioimmunoassay at the Animal Reproduction and Biotechnology Laboratory, Colorado State University, Fort Collins, CO (primary Ab R15, standard NIDDKrLERP-3, National Hormone and Pituitary Programs, NIDDK), following homogenization (Tena-Sempere et al., 2004). Each sample was measured in duplicate. The lower limit of detection for this assay was 0.2 ng/ml. Pituitary gland content of FSH was also measured by the same laboratory (primary Ab NIDDK-anti-rFSH-S-11 and standard NIH-RFSH-RP2). The lower limit of this assay was 12.2 ng/ml.
2.6. Progesterone analysis
Plasma progesterone levels were measured using a Coat-A-Count kit according to manufacturer’s protocol (Siemans, Tarrytown, NY). The intra-assay CV was 3% and the limit of detection was 0.1 ng/ml.
2.7. Statistical analysis
Body weight was analyzed as repeated measures ANOVA 3 (HgCl2) × 8 (length of exposure) factorial design, with duration of exposure as the repeated measure. The first weight taken and every subsequent fourth weight taken were used as the repeated measure. All other variables were analyzed using ANOVA 3(HgCl2) × quantity or number of variable. When a significant difference was observed a post hoc analysis was performed to identify the differences between the groups. Results were considered significant at α level of 0.05.
3. Results
3.1. Body weight
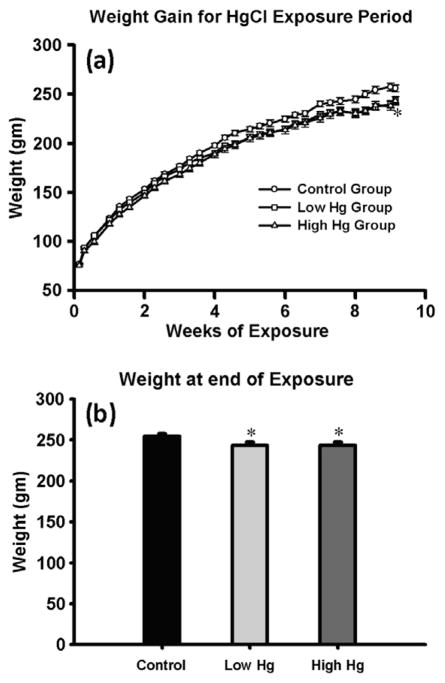
Both a main effect of HgCl2 (F(2,56) = 3.463, p = 0.038) and an interaction between exposure concentration and exposure duration (F(14,392) = 2.887, p < 0.001) revealed that the exposed groups had significantly less weight gain than controls. An analysis of weights taken on the last day of exposure also showed an overall significant difference (F(2,56) = 6.192, p = 0.004) between groups. Post hoc analysis using a Bonferroni correction revealed a significant difference between the low Hg (p = 0.015) and high Hg (p = 0.008) compared to controls. Post hoc analysis using Fisher’s Least-Significant-Difference test (FLSD) did reveal significantly lower weights in the high Hg group starting from day 9 of exposure (p = 0.015) and in the low Hg from day 30 of exposure (p = 0.044) compared to controls. However, it should be noted that FLSD is a less conservative test than Bonferroni. Table 1 gives a list of mean weights, the standard error, in grams, and whether significantly different from controls, for a particular day of exposure for all groups.
Table 1.
Mean weights of subjects across exposure period.
| Days of Hg exposure | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Group | 1 |
9 |
21 |
30 |
39 |
49 |
60 |
|||||||
| Mean | SE | Mean | SE | Mean | SE | Mean | SE | Mean | SE | Mean | SE | Mean | SE | |
| Control | 77.01 | 0.84 | 135.57 | 1.73 | 176.57 | 2.19 | 205.70 | 2.30 | 220.44 | 3.32 | 239.85 | 3.03 | 254.28 | 3.86 |
| Low Hg | 77.00 | 1.01 | 132.77 | 1.56 | 173.55 | 2.08 | 197.69* | 2.03 | 210.65* | 2.44 | 229.21* | 3.02 | 237.07* | 3.30 |
| High Hg | 75.70 | 1.34 | 127.62* | 2.55 | 168.25* | 2.87 | 194.29* | 3.56 | 210.84* | 3.63 | 225.68* | 4.18 | 238.60* | 4.24 |
Mean columns represent the mean weight in grams for an exposure group on a particular day of exposure. SE = standard error of the mean.
significant difference from controls using fisher LSD test.
3.2. Implantations
A significant effect of HgCl2 concentration was found for total implantations (F(2,56) = 3.404, p = 0.04), and non-viable implantations, (F(2,56) = 6.46, p = 0.003). Non-viable implantations were defined as sites on the uterus that showed signs of resorption or had not developed to a live fetus. Post hoc analysis (Bonferroni) indicated total implantations were significantly lower for the high Hg group (p = 0.033) compared to the control group, while the number of non-viable implantations in the high HgCl2 group were significantly higher than the low group (p = 0.047) and controls (p = 0.003). The number of corpora lutea, a measure of ovulation rate, did not differ between controls and either of the exposed groups (data not shown).
3.3. Progesterone, luteinizing and follicle stimulating hormones levels
In the low Hg group the pituitary gland was unavailable for one subject, while a plasma sample was unavailable for a separate subject. Pituitary glands were unavailable for four subjects in the high Hg group.
A significant effect for plasma progesterone levels between exposure groups (F(2,56) = 3.924, p = 0.025) was found. The mean level of progesterone in the high Hg group was 74.2 ng/ml which was significantly lower (p = 0.019) than the control group at 90.6 ng/ml (see Fig. 3).
Fig. 3.
Plasma progesterone levels. (a) Describes the mean levels of plasma progesterone by Hg exposure groups. Error bars = SE and * Indicate a significant differences from the control group. (b) Gives the distribution for the plasma progesterone levels by Hg exposure groups.
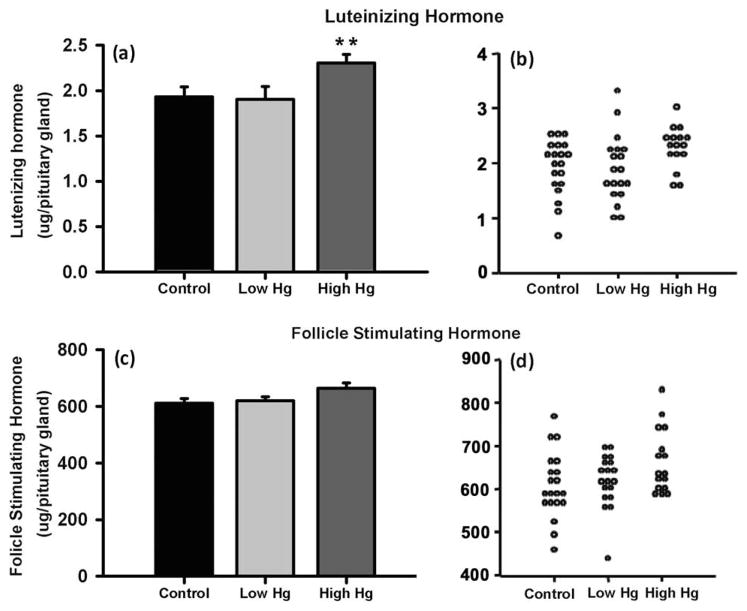
Analysis of pituitary LH showed a significant difference between groups (F(2,52) = 3.244, p = 0.047), with a mean concentration for the high Hg group at 2.3 μg/pituitary gland which was significantly higher than either the low Hg group at 1.9 μg/pituitary gland (p = 0.025), or the controls at 1.93 μg/pituitary gland (p = 0.034). In comparison, for FSH, there was no difference between the controls and the exposure groups (F(2,52) = 2.712, p = 0.076) (see Fig. 4). However, although not significant a dose response effects does appear to be present, which is evidenced by the distribution (see Fig. 4d).
Fig. 4.
(a) and (b) indicates levels of LH and the distribution according to exposure groups. (c) and (d) indicates levels of FSH and the distribution according to exposure groups. ** = significant difference from the control and the low Hg groups. Error bars represent SE.
4. Discussion
Various studies have reported reproductive effects of mercury (Hg) toxicity both in human populations (De Rosis et al., 1985; Sikorski et al., 1985; Elghany et al., 1997) and rodents (Atkinson et al., 2001; Davis et al., 2001; Stadnicka, 1980), however, for inorganic mercury the underlying mechanisms and reproductive toxic dose is still poorly understood. The present study was designed to address the effects on reproduction of inorganic mercury toxicity, in the form of mercuric chloride (HgCl2), in growing (PD 30 through 90) female rats.
The levels of mercuric chloride used in this study were selected based on a previous study (Atkinson et al., 2001) in which an exposure of 2.5 mg/kg/day in female rats showed clinical signs of lichenification, and erythema of the lips; centrifugum like erythema and alopecia. As it was desirable that we try to avoid any physical signs of HgCl2 toxicity the maximum exposure used in the present study was set at 2 mg/kg/day. This level achieved this goal as no overt physical signs of toxicity were seen. However, there was a significant decrease in weight gain in the exposed groups, (see Table 1 and Fig. 1a and b).
Fig. 1.
(a) Represents weight increase during the exposure period of mercuric chloride for the three experimental groups. Error bars represent SE. (b) Represents the mean weights of the subjects on the last day of exposure. * = A significant difference from the control group.
The main finding of the study was the lower number of implantations in the high HgCl2 compared to the other two groups. Equally important was the significantly higher number of non-viable implantations in this group compared to the controls and the low HgCl2 subjects. These data agree with, and extends previous results which showed significantly lower levels of implant efficiency, in female rats, exposed to similar concentrations of HgCl2, (Atkinson et al., 2001). However, in the Atkinson et al. (2001) study both male and females were exposed to HgCl2; therefore a direct comparison cannot be made (see Fig. 2).
Fig. 2.
(a) Represents the means of the three experimental groups for total implantations (black) and non-viable implantations (grey). Error bars represent SE, * indicates significant differences from control group. ** Indicates significant differences from control and low Hg group. (b) and (c), displays the distributions for the two variables for each of the three experimental groups.
The count of the corpora lutea showed no difference between any of the groups. This supports findings by Yang et al. (1996), who, in a similar experiment also reported no difference in the corpora lutea count between control rats and rats exposed to HgCl2. It can therefore be assumed that since the number of corpora lutea represents the number of ova released (Seethalakshmi et al., 1992), HgCl2 had no effect on ovulation rate.
Plasma progesterone was significantly lower in the high HgCl2 group compared to controls and, although the low HgCl2 group was not significantly different from the control group, levels of plasma progesterone followed a dose related pattern of lower progesterone to high HgCl2 exposure. This can be seen in the distribution chart (see Fig. 3b). In the rat, the corpora lutea are the primary source of progesterone throughout gestation, with a small amount being produced by the placenta (Numan, 1988). The luteal potential for progesterone secretion declines immediately prior to term, (Gallo et al., 1985; Strauss et al., 1975). It is possible that the lower progesterone level in the high HgCl2 group was influenced by the lower number of implantations in this group. Lower implantation rate would equate to a lower number of placentas, and thus, an overall lower amount of progesterone. However, the majority of the progesterone comes from the corpora lutea (Numan, 1988). The animals in this study were euthanized on average at day 13 of pregnancy; therefore it can be assumed that the progesterone was still being produced mainly by the corpora lutea.
Luteinizing hormone was found to be significantly higher in the high group compared to controls and the low exposure group (see Fig. 4a and b), although there was no difference between the low and control groups. A previous study found increase levels of progesterone from days 14–16 of pregnancy and decreased levels of LH during the same period, in unexposed rats (Gallo et al., 1985). Although, in the present study the levels of LH were measured in the pituitary rather than the plasma, they still show the direct opposite of levels found by Gallo et al. (1985) in unexposed animals. The risk of spontaneous abortion is slightly increased when LH levels are higher (Duijkers et al., 1993), and higher levels of induced LH have been found to influence changes in the endometrium and may interfere with the maintenance of pregnancy (Stein et al., 1993). Also, the need for progesterone in maintaining pregnancy is supported by the fact that blocking of progesterone binding sites causes abortion in human and other species (Arck et al., 2007). Therefore, both the higher levels of LH and the lower levels of progesterone may precipitate lower implantations and fetal survival.
As with LH, plasma concentrations of FSH normally stay within the range of diestrous levels during pregnancy (Taya et al., 1989), however, an inspection of the distribution of FSH levels (see Fig. 4d) indicates an upward trend in FSH levels. Although there was no significant difference found for FSH levels we see a tighter grouping within the low Hg exposure group which is toward the high end of the control group. Similarly, the high Hg exposure group is distributed across a greater range than the other group with a tendency towards higher levels of FSH.
From the above evidence it may be surmised that the higher levels of LH (and possibly FSH), together with the lower levels of progesterone in the high exposure group, (and to a lesser extent in the low exposure group) may have been influential in the lower implantation and fetal survival rate. However, the question remains where did HgCl2 have this effect?
Luteinizing hormone and FSH are produced in the anterior pituitary gland. These hormones are released in response to pulsating secretion of the gonadotropin-releasing hormone from the hypothalamus. They bind to specific G-protein coupled receptors in the ovary, and affect (among other processes) corpus luteum formation (Schirman-Hildesheim et al., 2008). The regulation of these hormones synthesis and release is an intricate interplay of positive and negative feedback loops involving the hypothalamus, pituitary and ovary (Burger et al., 2004). The higher quantities of LH (and possibly FSH) in the present study, were found in the pituitary.
In the rat, normal function of the corpus luteum is critical for the maintenance of pregnancy during its entire length, since it is the main source of circulating progesterone. This hormone, in turn, is required for implantation and for regulating pituitary gonadotropin secretion i.e., LH and FSH (Risk and Gibori, 2001). LH and FSH are instrumental in the development of the ovarian follicles and corpora lutea, which produce 17β-estradiol and progesterone, respectively (Hoyer and Devine, 2002).
The corpus luteum is formed after release of the ova partially by the influence of LH (Schirman-Hildesheim et al., 2008). After ovulation the corpora lutea produce progesterone (Channing et al., 1980). When mercuric chloride was administered to 30 day old female rats for 12 weeks, mercury was found deposited in the lutein and granulosa cells of atretic immature and mature ovarian follicles, (Stadnicka, 1980). The ovaries in the present study were not analyzed for Hg content, however, it is possible that Hg accumulating in the ovaries, although having little effect on ovulation, did however, after ovulation and the forming of the corpora lutea, disrupted the production of progesterone. This in turn may have had a twofold effect in the exposed animals. The level of progesterone failed to fully support the implantation of fertilized ova into the uterus and maintain full fetal viability. Also, the reduced progesterone levels disrupted the negative feedback loop allowing greater production of pituitary gonadotropin secretion, which in turn also had a negative effect on implantation and fetal survival. This supports our results of equality of corpora lutea between groups, but, lower implantation and fetal viability, together with higher LH and lower progesterone levels in the high HgCl2 exposure group. Additional studies are needed to help further identify the exact mechanisms that are affected by HgCl2 exposure on reproduction, especially within the mechanisms of the corpora lutea.
In summary, the purpose of this study was to examine the effects of chronic ingestion of inorganic mercury on reproduction in female rats. While not effecting ovulation, the toxic effects produced disruption of implantation of the ovum into the uterus and fetal viability. The lowering of progesterone levels, higher LH and possibly FSH levels, indicate that mercuric chloride may have a disruptive effect in the corpora lutea which manifests itself after ovulation. However, the mechanistic reasons for these effects are speculative at best due to the nature of this study. Further investigation will be needed to reveal the cause of the reproductive disruption.
Acknowledgments
The authors would like to gratefully acknowledge the help, guidance, and assistance provided by Dr. H.O. Goyal, without whose cooperation this project would not have occurred. Funding: This work was supported by the National Institutes of Health [RCMI 5 G12 RR003059].
Abbreviations
- FSH
follicle stimulating hormone
- GD
gestation day
- Hg
mercury
- Hg0
metallic
- Hg+
mercurous
- Hg2+
mercuric
- HgCl2
mercuric chloride
- LH
luteinizing hormone
- PD
postnatal day
Footnotes
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Arck P, Hansen PJ, Mulac JB, Piccinni MP, Szekeres-Bartho J. Progesterone during pregnancy: endocrine-immune cross talk in mammalian species and the role of stress. Am J Reprod Immunol. 2007;58:268–279. doi: 10.1111/j.1600-0897.2007.00512.x. [DOI] [PubMed] [Google Scholar]
- Atkinson A, Thompson SJ, Khan AT, Graham TC, Ali S, Shannon C, Clarke O, Upchurch L. Assessment of a two generation reproductive and fertility study of mercuric chloride in rats. Food Chem Toxicol. 2001;39:73–84. doi: 10.1016/s0278-6915(00)00096-x. [DOI] [PubMed] [Google Scholar]
- Burger LL, Haisenleder DJ, Dalkin AC, Marshall JC. Regulation of gonadotropin subunit gene transcription. J Mol Endocrinol. 2004;33 (3):559–584. doi: 10.1677/jme.1.01600. [DOI] [PubMed] [Google Scholar]
- Channing CP, Schaerf FW, Anderson LD, Tsafriri A. Ovarian follicular and luteal physiology. Int Rev Physiol. 1980;22:117–201. [PubMed] [Google Scholar]
- Daintith J. A Dictionary of Chemistry. 3. Oxford University Press; New York: 1996. [Google Scholar]
- Davis B, Price H, O’Connor R, Fernando R, Rowland A, Morgan D. Mercury vapor and female reproductive toxicity. Toxicol Sci. 2001;59:291–296. doi: 10.1093/toxsci/59.2.291. [DOI] [PubMed] [Google Scholar]
- De Rosis F, Anastasio SP, Selvaggi L, Beltrame A, Moriani G. Female reproductive health in two lamp factories: effects of exposure to inorganic mercury vapor and stress factors. Br J Ind Med. 1985;42 (7):488–494. doi: 10.1136/oem.42.7.488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duijkers IJ, Vemer HM, Hollanders JM, Willemsen WN, Bastiaans LA, Hamilton CJ, et al. Different follicle stimulating hormone/luteinizing hormone ratios for ovarian stimulation. Hum Reprod. 1993;8 (9):1387–1391. doi: 10.1093/oxfordjournals.humrep.a138266. [DOI] [PubMed] [Google Scholar]
- Elghany NA, Stopford W, Bunn WB, Fleming LE. Occupational exposure to inorganic mercury vapor and reproductive outcomes. Occup Med (Lond) 1997;47 (6):333–336. doi: 10.1093/occmed/47.6.333. [DOI] [PubMed] [Google Scholar]
- Gallo RV, Devorshak-Harvey E, Bona-Gallo A. Pulsatile luteinizing hormone release during pregnancy in the rat. Endocrinology. 1985;116 (6):2637–2642. doi: 10.1210/endo-116-6-2637. [DOI] [PubMed] [Google Scholar]
- Grandjean P, Weihe P, Jorgensen PJ, Clarkson T, Cernichiari E, Videro T. Impact of maternal seafood diet on fetal exposure to mercury, selenium, and lead. Arch Environ Health. 1992;47:185–195. doi: 10.1080/00039896.1992.9938348. [DOI] [PubMed] [Google Scholar]
- Hoyer PB, Devine PJ. Endocrinology and toxicology: the female reproductive system. In: Derelank MJ, Hollinger MA, editors. Handbook of Toxicology. 2. CRC Press; Boca Raton: 2002. pp. 573–596. [Google Scholar]
- Hoyer PB. Damage to ovarian development and function. Cell Tissue Res. 2005;322 (1):99–106. doi: 10.1007/s00441-005-1083-y. [DOI] [PubMed] [Google Scholar]
- Numan M. Maternal behavior. In: Knobil E, Neill JD, editors. The Physiology of Reproduction. 2. Vol. 2. Raven Press; New York: 1988. pp. 1569–1645. [Google Scholar]
- Risk M, Gibori G. Mechanisms of luteal cell regulation by prolactin. In: Horseman ND, editor. Prolactin. Kluwer Academic Publishers; Boston: 2001. pp. 265–295. [Google Scholar]
- Schirman-Hildesheim TD, Gershon E, Litichever N, Galiani D, Ben-Aroya N, Dekel N, et al. Local production of the gonadotropic hormones in the rat ovary. Mol Cell Endocrinol. 2008;282 (1–2):32–38. doi: 10.1016/j.mce.2007.11.014. [DOI] [PubMed] [Google Scholar]
- Seethalakshmi L, Flores C, Kinkead T, Carboni AA, Malhotra RK, Menon M. Effects of subchronic treatment with cis-platinum on testicular function, fertility, pregnancy outcome, and progeny. J Androl. 1992;13 (1):65–74. [PubMed] [Google Scholar]
- Sikorski R, Juszkiewicz T, Paszkowski T, Szprengier-Juszkiewicz T. Women in dental surgeries: reproductive hazards in occupational exposure to metallic mercury. Int Arch Occup Environ Health. 1985;59 (6):227–233. doi: 10.1007/BF00377918. [DOI] [PubMed] [Google Scholar]
- Stadnicka A. Localization of mercury in the rat ovary after oral administration of mercuric chloride. Acta Histochem. 1980;67:227–233. doi: 10.1016/S0065-1281(80)80026-2. [DOI] [PubMed] [Google Scholar]
- Stein BA, Kramer B, de Wet G, van der Walt LA. Dose-dependent effects of exogenous gonadotrophins on the endometrium of the rat. S Afr Med J. 1993;83 (2):122–125. [PubMed] [Google Scholar]
- Strauss JF, 3rd, Sokoloski J, Caploe P, Duffy P, Mintz G, Stambaugh RL. On the role of prostaglandins in parturition in the rat. Endocrinology. 1975;96 (4):1040–1043. doi: 10.1210/endo-96-4-1040. [DOI] [PubMed] [Google Scholar]
- Taya K, Komura H, Watanabe G, Sasamoto S. Peripheral blood levels of immunoreactive inhibin during pseudopregnancy, pregnancy and lactation in the rat. J Endocrinol. 1989;121 (3):545–552. doi: 10.1677/joe.0.1210545. [DOI] [PubMed] [Google Scholar]
- Tena-Sempere M, Barreiro ML, Aguilar E, Pinilla L. Mechanisms for altered reproductive function in female rats following neonatal administration of raloxifene. Eur J Endocrinol. 2004;150 (30):397–403. doi: 10.1530/eje.0.1500397. [DOI] [PubMed] [Google Scholar]
- Yang JM, Jiang XZ, Chen QY, Li PJ, Zhou YF, Wang YL. The distribution of HgCl2 in rat body and its effects on fetus. Biomed Environ Sci. 1996;9 (4):437–442. [PubMed] [Google Scholar]