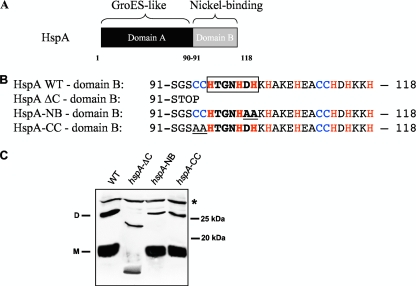
FIG. 1.
(A) Representation of the HspA protein of H. pylori with the GroES-like domain A and the nickel-binding domain B. (B) Amino acid sequence of domain B of wild-type HspA and of three mutants: HspA-ΔC, with a complete deletion of this domain, and HspA-NB and -CC, each carrying two substitutions that are underlined. Cysteine and histidine residues are in blue and red, respectively. The HX4DH motif, which in the nickel-cobalt (NiCoT) transporter family is considered to be a nickel-binding signature sequence, is boxed. (C) Immunoblot experiment with whole-cell lysates from the H. pylori wild-type strain and from the three hspA mutants after denaturing SDS-PAGE and using the monoclonal antibody P1-1, which specifically recognizes a conserved epitope of HspA domain A. The predicted molecular mass of the wild-type HspA monomer is 13 kDa, and that of HspA-ΔC is 9.8 kDa. The monomeric (M) and dimeric (D) forms of the HspA wild type (WT) are indicated on the left side of the blot. A cross-reacting unspecific protein band is marked with a star (*) and served as a loading control. Molecular mass standards are indicated at right.