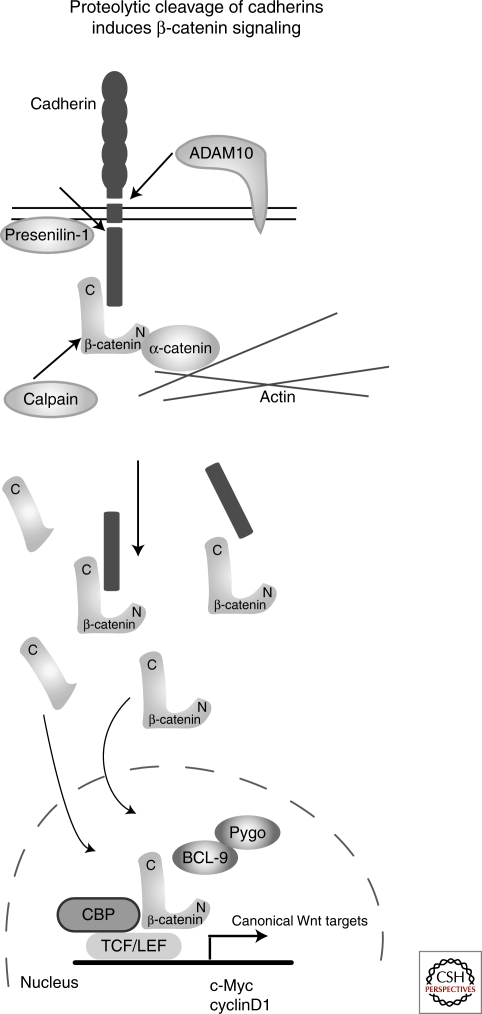
Figure 3.
Proteolytic cleavage of cadherin can release β-catenin and induce canonical Wnt signaling. Under certain cellular conditions, cadherins can be cleaved by proteases like ADAM10 and Presenilin-1. This cleavage leads to loosening of cell adhesion, but also to the release of β-catenin, which is translocated into the nucleus and as a result, activates Wnt/β-catenin target genes. Another protease, calpain, is interfering with the adhesion complex by the amino-terminal cleavage of β-catenin. β-Catenin cleaved by calpain is stabilized and can enter the nucleus and activate target genes.