Abstract
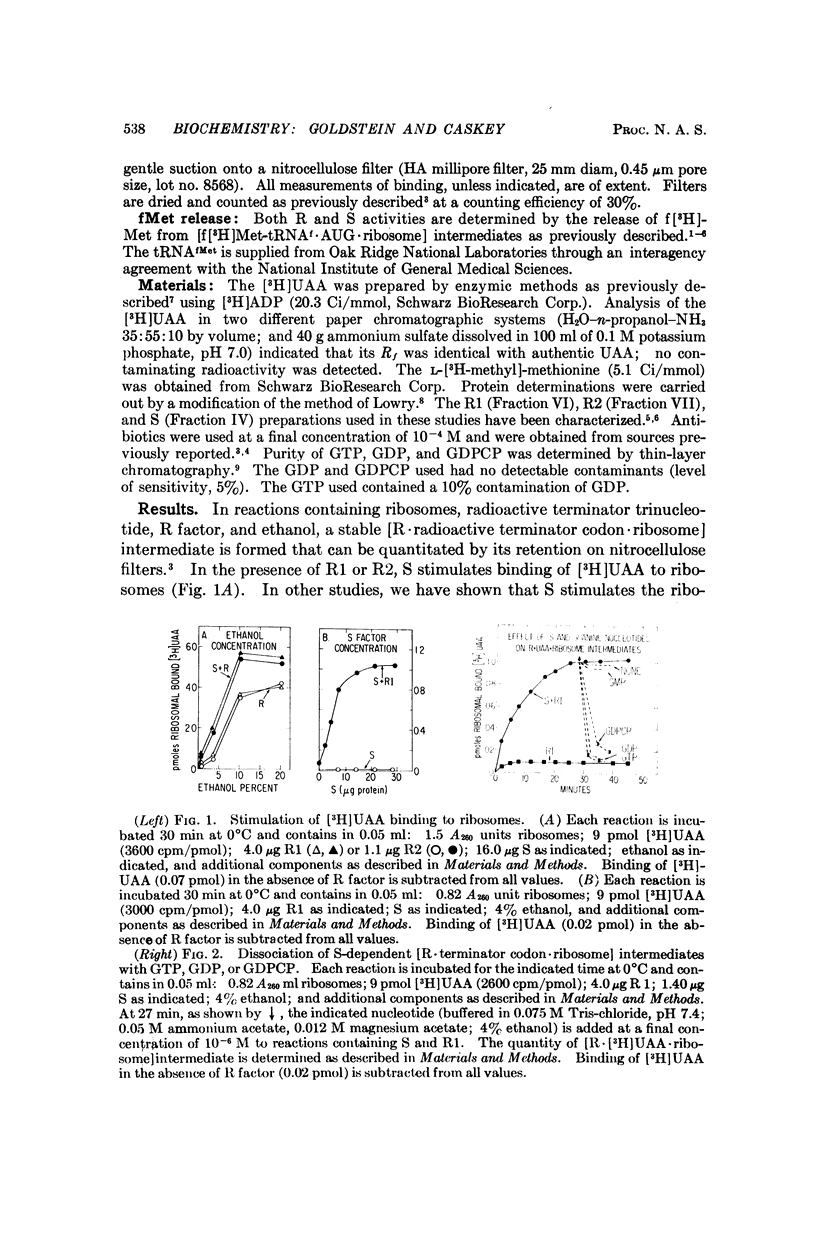
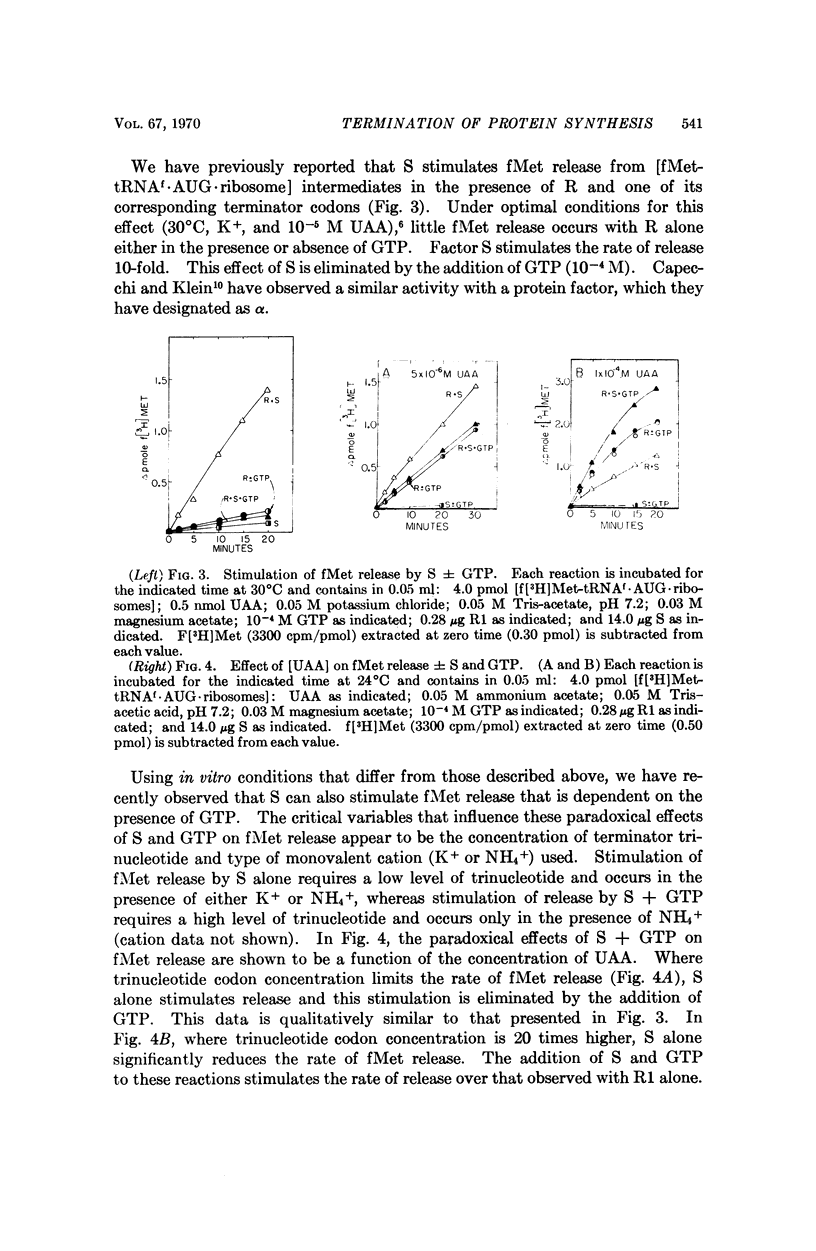
The protein factor S, previously shown to stimulate polypeptide chain termination in bacterial extracts, has two effects upon the complex formed between ribosomes, release factor, and terminator (trinucleotide) codon: (1) in the absence of GTP or GDP, S stimulates formation of an [R·UAA·ribosome] intermediate, and (2) in the presence of GTP or GDP, S participates in dissociation of this intermediate. Factor S can stimulate fMet release from [fMet-tRNAf·AUG·ribosome] intermediates in either the presence or absence of GTP or GDP. A model is proposed which relates the in vitro effects of S ± GTP (or GDP) on fMet release to the effects of S ± GTP (or GDP) on the binding and dissociation of R factor from ribosomes.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Cashel M., Lazzarini R. A., Kalbacher B. An improved method for thin-layer chromatography of nucleotide mixtures containing 32P-labelled orthophosphate. J Chromatogr. 1969 Mar 11;40(1):103–109. doi: 10.1016/s0021-9673(01)96624-5. [DOI] [PubMed] [Google Scholar]
- Caskey C. T., Tompkins R., Scolnick E., Caryk T., Nirenberg M. Sequential translation of trinucleotide codons for the initiation and termination of protein synthesis. Science. 1968 Oct 4;162(3849):135–138. doi: 10.1126/science.162.3849.135. [DOI] [PubMed] [Google Scholar]
- Caskey T., Scolnick E., Tompkins R., Goldstein J., Milman G. Peptide chain termination, codon, protein factor, and ribosomal requirements. Cold Spring Harb Symp Quant Biol. 1969;34:479–488. doi: 10.1101/sqb.1969.034.01.054. [DOI] [PubMed] [Google Scholar]
- Caskey T., Scolnick E., Tompkins R., Goldstein J., Milman G. Peptide chain termination, codon, protein factor, and ribosomal requirements. Cold Spring Harb Symp Quant Biol. 1969;34:479–488. doi: 10.1101/sqb.1969.034.01.054. [DOI] [PubMed] [Google Scholar]
- Gallant J., Harada B. The control of ribonucleic acid synthesis in Escherichia coli. 3. The functional relationship between purine ribonucleoside triphosphate pool sizes and the rate of ribonucleic acid accumulation. J Biol Chem. 1969 Jun 25;244(12):3125–3132. [PubMed] [Google Scholar]
- Goldstein J. L., Beaudet A. L., Caskey C. T. Peptide chain termination with mammalian release factor. Proc Natl Acad Sci U S A. 1970 Sep;67(1):99–106. doi: 10.1073/pnas.67.1.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldstein J., Milman G., Scolnick E., Caskey T. Peptide chain termination. VI. Purification and site of action of S. Proc Natl Acad Sci U S A. 1970 Feb;65(2):430–437. doi: 10.1073/pnas.65.2.430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Leder P., Singer M. F., Brimacombe R. L. Synthesis of trinucleoside diphosphates with polynucleotide phosphorylase. Biochemistry. 1965 Aug;4(8):1561–1567. doi: 10.1021/bi00884a015. [DOI] [PubMed] [Google Scholar]
- Milman G., Goldstein J., Scolnick E., Caskey T. Peptide chain termination. 3. Stimulation of in vitro termination. Proc Natl Acad Sci U S A. 1969 May;63(1):183–190. doi: 10.1073/pnas.63.1.183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scolnick E. M., Caskey C. T. Peptide chain termination. V. The role of release factors in mRNA terminator codon recognition. Proc Natl Acad Sci U S A. 1969 Dec;64(4):1235–1241. doi: 10.1073/pnas.64.4.1235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tompkins R. K., Scolnick E. M., Caskey C. T. Peptide chain termination. VII. The ribosomal and release factor requirements for peptide release. Proc Natl Acad Sci U S A. 1970 Mar;65(3):702–708. doi: 10.1073/pnas.65.3.702. [DOI] [PMC free article] [PubMed] [Google Scholar]


