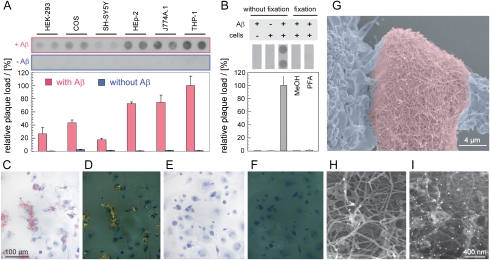
Fig. 1.
Cell-dependent formation of Aβ amyloid plaques. (A) Amyloid load obtained with different cell lines after incubation for 3 days in the presence of freshly dissolved 60 μg/mL Aβ(1–40). On day zero, the medium was additionally supplemented with 100 ng/mL seeds, corresponding to 0.2% of the total amount of Aβ peptide added on day zero. Representative raw data images from filter retention assay and densitometric quantifications shown (n = 30; 0%: background; 100%: amyloid load with THP-1 cells). (B) Amyloid load in wells containing living THP-1 cells (without fixation), Aβ(1–40) or dead THP-1 cell bodies as indicated (n = 12). Dead THP-1 cell bodies were obtained by fixation of THP-1 cells using methanol (MeOH) or PFA. MeOH and PFA were removed before addition of Aβ(1–40) peptide or seeds. Medium supplements as in panel (A). (C–F) Congo red stained THP-1 cell culture after 4 d incubation with freshly dissolved Aβ(1–40) and seeds (C, D) or after 8 d incubation without any Aβ (E, F). Bright field (C, E) or dark field (D, F) polarizing microscopy images shown. (G–I) SEM image of an amyloid plaque from THP-1 cell culture after 6 d incubation, showing a fibrillar ultrastructure. Medium supplements as in panel (A). (G) Color coding: blue, cell bodies; red, amyloid plaque. (H, I) Secondary electron SEM image (H) or mixed secondary: backscatter electron SEM image (20∶80 ratio) (I) of an Aβ immunogold labeled plaque. White points show immunogold labeled Aβ.