Abstract
In male song sparrows (Melospiza melodia), territorial challenges during the breeding season can rapidly increase circulating levels of testosterone (T). During the non-breeding season, male song sparrows are highly aggressive, but the gonads are regressed and plasma T levels are non-detectable and unaffected by territorial challenges. The pro-hormone dehydroepiandrosterone (DHEA) is elevated in song sparrow plasma and brain during the non-breeding season and may be locally converted to sex steroids in the brain to regulate aggression. The enzyme 3β-hydroxysteroid dehydrogenase/Δ5-Δ4 isomerase (3β-HSD) converts DHEA to androstenedione (AE) using the cofactor NAD+, and this is a critical rate-limiting step. We predicted that brain 3β-HSD activity varies seasonally and is rapidly modulated by aggressive challenges. In the first study, brain 3β-HSD activity was highest in the non-breeding season in specific regions. In the second study, a simulated territorial challenge rapidly increased aggressive behavior in non-breeding song sparrows. Brain 3β-HSD activity, when measured without exogenous NAD+, increased by ∼250 to 500% in telencephalic regions of challenged subjects. When brain 3β-HSD activity was measured with exogenous NAD+, these effects of territorial challenges were not observed. These data suggest that territorial challenges rapidly increase endogenous NAD+levels or increase 3β-HSD activity specifically within a NAD-rich subcellular compartment. Together, these two studies suggest a shift from systemic to local sex steroid signaling in the non-breeding season. Local steroid signaling produces high spatial and temporal specificity of steroid signals and avoids the costs of high systemic T levels during the non-breeding season.
Keywords: 3beta-HSD, aggression, androstenedione, aromatase, coenzyme, cofactor, DHEA, estrogens, intracrine, NAD+, neurosteroid, song sparrow, songbird, testosterone, Challenge Hypothesis, estradiol
Introduction
Conspecific social interactions profoundly affect behavior, hormones and the brain (Francis et al., 1993; Insel and Fernald, 2004; Adkins-Regan, 2005; Robinson et al., 2008). For example, during periods of social instability, aggressive interactions can rapidly increase circulating levels of testosterone (T) in some species, including humans (“Challenge Hypothesis”) (Wingfield et al., 1990; Archer, 2006; Goymann, 2009). Studies of song sparrows, Melospiza melodia, were critical in the formulation of the Challenge Hypothesis. In male song sparrows, plasma T levels are high during the breeding season (spring) and rapidly increase even higher during territorial disputes (Wingfield et al., 1990).
In sedentary song sparrow populations, males are also highly aggressive towards territorial intruders during the non-breeding season (autumn and winter) (Wingfield and Hahn, 1994). Intriguingly, during the non-breeding season, the gonads are completely regressed, circulating T levels are very low, and castration does not affect aggression (Wingfield and Hahn, 1994). Importantly, aggressive interactions do not increase systemic T levels in the plasma of non-breeding song sparrows (Wingfield and Hahn, 1994; Soma and Wingfield, 2001). Androgens and estrogens synthesized locally in the brain may regulate non-breeding aggression (Schmidt et al., 2008). Local steroid signaling can circumvent the ‘costs’ of high systemic T levels during the non-breeding season in song sparrows (Wingfield et al., 2001; Soma, 2006; Schmidt et al., 2008) as well as in other species that display territorial aggression outside the breeding season (Caldwell et al., 1984; Demas et al., 2007; Soma et al., 2008).
Circulating levels of the pro-hormone dehydroepiandrosterone (DHEA) are high in non-breeding song sparrows (Soma and Wingfield, 2001; Newman et al., 2008), but aggressive interactions do not increase systemic DHEA levels (Soma and Wingfield, 2001). However DHEA treatment increases territorial singing and the size of a song control nucleus (HVC) during the non-breeding season (Soma et al., 2002). DHEA does not bind to a classical intracellular steroid receptor (Mellon and Griffin, 2002; Widstrom and Dillon, 2004; Labrie et al., 2005), but can be locally converted into active sex steroids such as T and estradiol (E2) (Soma et al., 2004; Hajszan et al., 2004). Inhibition of E2 synthesis in wintering male song sparrows reduces aggressive behavior (Soma et al., 1999a; Soma et al., 2000a; Soma et al., 2000b). These data suggest that DHEA is converted to androgens and estrogens within the brain to promote non-breeding aggression.
The steroidogenic enzymes that metabolize DHEA to active sex steroids are expressed in the avian brain in a region-specific manner (Vanson et al., 1996; London et al., 2006). 3β-hydroxysteroid dehydrogenase/Δ5-Δ4 isomerase (3β-HSD) catalyzes the conversion of DHEA to androstenedione (AE), an active and aromatizable androgen (Schlinger et al., 2008). Importantly, 3β-HSD and other HSDs interconvert hydroxysteroids and ketosteroids using nicotinamide cofactors as electron donors or acceptors (Agarwal and Auchus, 2005). In particular, for the conversion of DHEA to AE, 3β-HSD requires the cofactor nicotinamide adenine dinucleotide (NAD+) to serve as an electron acceptor. 3β-HSD has been primarily studied in the gonads, adrenal glands, liver, and placenta, and less is known about 3β-HSD in the brain (Vanson et al., 1996; Payne and Hales, 2004). The majority of studies on 3β-HSD activity have focused on parameters such as substrate affinity and specificity, maximum velocity, subcellular localization, and regulation by end products (Yates and Deshpande, 1974; Gower and Cooke, 1983). Far less is known about how environmental and social cues regulate 3β-HSD. For example, recent studies in songbirds demonstrate that brain 3β-HSD activity is rapidly regulated by restraint stress (Soma et al., 2004). 3β-HSD may play an important role in the regulation of aggressive behavior (Goodson et al., 2005; Soma et al., 2008), but its regulation by relevant social cues has not been examined.
Here, in Experiment 1, we examined chronic seasonal regulation of brain 3β-HSD activity in wild male song sparrows. In Experiment 2, we examined acute social regulation of brain 3β-HSD activity (with and without exogenous NAD+) in the non-breeding season.
Materials and Methods
All protocols complied with institutional (University of Washington and University of British Columbia), NIH and the Canadian Council for Animal Care guidelines.
Experiment 1: Seasonal regulation of brain 3β-HSD activity
Field protocol
Wild male song sparrows were captured near Seattle, WA using mist nets and song playback during the breeding (May 4-5), molt (Aug 30-31), and non-breeding (Nov 14-16) seasons (n=6 per season). The amount of playback used to capture subjects did not differ among seasons (F2,15=1.98, p=0.17; 3.58 ± 0.37 min). Subjects were euthanized by rapid decapitation (3.68 ± 0.26 min after capture), and trunk blood was collected. In the field, blood was kept on wet ice, and the brain was dissected as per Soma and colleagues (2004), and immediately frozen with dry ice. We collected several regions, including the central medial telencephalon (contains septum), caudal telencephalon (contains RA), ventromedial telencephalon (contains taenial amygdala), rostral and caudal diencephalon, cerebellum, and optic lobes. In the lab, blood was centrifuged and plasma stored at -20°C, and tissues were stored at -80°C.
Steroid assays
DHEA and T were extracted from plasma using dichloromethane, separated using Celite chromatography columns, and measured via radioimmunoassays as in (Soma and Wingfield, 2001). DHEA was measured using a specific antibody (Endocrine Sciences, Calabasas Hills, CA). The DHEA antibody has a low cross-reactivity with DHEA-S (0.05%), T (0.08%), 5α-DHT (0.07%), AE (2.5%), pregnenolone (0.09%), and progesterone (0.07%). The recovery for DHEA was 67.3 ± 1.7 % and the intra-assay variability was 3.25%. The T antibody (Wien Lab, Succasunna, NJ) shows 60% cross-reactivity with 5α-DHT (Soma et al., 1999b), but T and 5α-DHT are separated during chromatography. The recovery for T was 53.8 ± 1.6 %, and the intra-assay variability was 7.1% for T.
3β-HSD activity assay with exogenous NAD+
We measured the in vitro conversion of [3H]DHEA to [3H]AE and [3H]5β-androstanedione (5β-A) in whole homogenates, as in (Soma et al., 2004). Each sample was homogenized on ice in 200 μL ice-cold sucrose phosphate (SPO4) buffer. Whole homogenate (180 μL) was incubated with [1,2,6,7-3H]DHEA (specific activity = 74 Ci/mmol, Perkin-Elmer). [3H]DHEA was repurified before use. The [3H]DHEA concentration was 200 nM, similar to previous studies (Vanson et al., 1996; Soma et al., 2004; Pradhan et al., 2008). This substrate concentration was sub-saturating, but over 80% of [3H]DHEA remained at the end of incubations (unpublished results). This substrate concentration yielded low background values and provided the best signal to noise ratio. Radioinert AE (25 μM final concentration) was added to protect formed [3H]AE from further metabolism (“cold trap”). Incubations also included 1 mM NAD+ (20 μL). Samples were incubated at 41°C for 180 min. In a timecourse study, product formation was linear from 0 to 240 min (linear regression: r2=0.9613; data not shown). Formed steroids were extracted with diethyl ether (2×) and separated using thin layer chromatography (TLC), as in (Soma et al., 2004). The appropriate bands (AE, 5β-A) were scraped from the silica-gel plates. We did not measure formed [3H]T because [3H]T runs below the substrate ([3H]DHEA) in this system, leading to very high background values. Formed steroids were eluted from the silica with methanol, and aliquots were counted in a liquid scintillation counter. For each assay, background values were obtained from tubes containing everything but tissue. Recovery was calculated by processing tubes containing a known amount of [3H]AE in parallel (average recovery was 85.27 ± 4.32% across assays). Protein content for each sample was determined using the Bradford method. Each sample was corrected for background values and recovery, and data are presented as fmole per mg protein per min of incubation.
Formed [3H]AE was recrystallized (3×) to constant specific activity to confirm its identity as in (Soma et al., 2004). Trilostane, a specific 3β-HSD inhibitor, decreased [3H]AE formation by 98.5%, as in previous studies (Soma et al., 2004; Pradhan et al., 2008).
Experiment 2: Social regulation of brain 3β-HSD activity
Field protocol
Wild male song sparrows were studied during the non-breeding season (Nov 23-Dec 8) near Vancouver, BC. Individual territories were mapped at least 2 d before the simulated territorial intrusion (STI) or control. Immediately before each STI or control, a mist net was set up, furled, and pushed down near the ground. For the STI, a caged live conspecific decoy was placed near the center of the territory along with conspecific song playback for 30 min. Control subjects were exposed to an empty cage without playback for 30 min. An observer standing 15-20 m away recorded territorial behavior. We recorded the latency to a response of any kind (call, song, or flight), the latency to sing, the number of songs, the time spent in close proximity to the decoy (within 1 or 5 m), and the number of flights directed towards the decoy (data to presented separately). After 30 min, the mist net was unfurled, and the subject was captured using a small amount of playback (Control: 1.36 ± 0.6 min, range = 0.17 to 6 min; STI: 1.64 ± 0.53 min, range = 0.3 to 5.03 min), and this did not differ between groups (t=0.332, p=0.744). Subjects were euthanized by rapid decapitation (Control: 4.19 ± 0.24 min after capture; STI: 4.13 ± 0.27 min; t=0.184, p=0.857). In the field, the brain was dissected and immediately frozen on dry ice. We collected the same regions as in Experiment 1, as well as the rostral telencephalon, dorsal telencephalon (contains HVC), caudal medial nidopallium (NCM), and hippocampus, as in (Newman and Soma, 2009). Tissue was stored at -80°C.
Experiment 2A: 3β-HSD activity assay with exogenous NAD+
We determined 3β-HSD activity as in Experiment 1, with slight modifications. We used supernatants prepared from whole homogenates because 3β-HSD activity was ca. 6× higher in supernatants (data not shown), as in (Pradhan et al., 2008). Brain tissue from the right or left side (randomized) was homogenized with a motorized homogenizer on ice (2 sec pulses, 3 to 6×) in 300 μL ice-cold SPO4 buffer, and supernatant was obtained by centrifugation at 1000 g at 4°C for 30 min. This supernatant is enriched with mitochondria and microsomes (which contain 3β-HSD), and thus allows for short incubation times (Pradhan et al., 2008). The pellet containing cell debris, unbroken cells, and cell nuclei was discarded. 180 μL of the supernatant was incubated with 200 nM [3H]DHEA and NAD+ (20 μL; 1 mM final concentration). Incubations were performed at 41°C for 5 min (based on timecourse studies, Fig. 1A). Product formation was linear from 0 to 7.5 min (linear regression: r2=0.9570). Steroids were extracted with diethyl ether, following which all the procedures were identical to (Pradhan et al., 2008). The dpm were corrected for background values for each assay (average recovery was 82.33 ± 1.8 % across all assays). All data are presented as fmole per mg protein per min of incubation.
Figure 1.
Timecourse of 3β-HSD activity using 200 nM [3H]DHEA as substrate and brain supernatants from non-breeding adult male song sparrows. (A) Formed tritiated androgens measured with the addition of 1 mM NAD+, from 2.5 to 60 min. n = 3 replicates per timepoint. (B) Formed tritiated androgens and estrogens measured without the addition of NAD+, from 10 to 360 min. n = 2 replicates per timepoint.
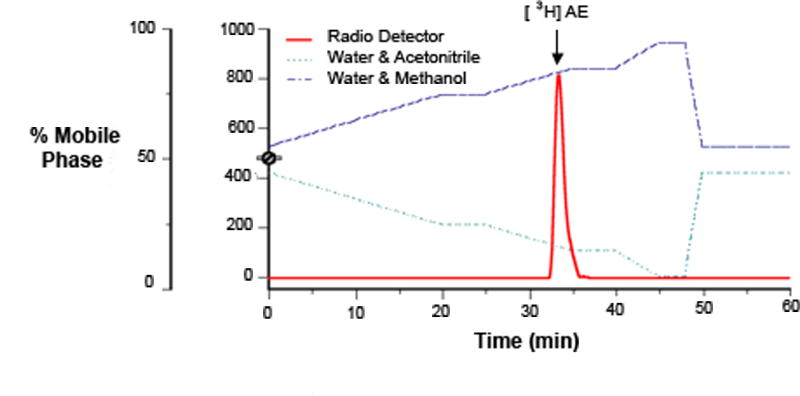
We confirmed the identity of formed [3H]AE via HPLC with continuous flow radiodetection, as in (Pradhan et al., 2008). HPLC analyses showed a single peak with a retention time identical to [3H]AE standards (Fig. 2).
Figure 2.
Representative HPLC chromatograph illustrating the peak and retention time of [3H]AE produced in vitro from brain tissue of non-breeding adult male song sparrows. Substrate was 200 nM [3H]DHEA. Steroids were separated using TLC and then injected through the HPLC coupled to a flow radiodetector.
Experiment 2B: 3β-HSD activity assay without exogenous NAD+
For some of the larger brain samples (rostral telencephalon, central medial telencephalon, caudal telencephalon), we also measured 3β-HSD activity in the absence of exogenous NAD+, allowing us to measure formation of [3H]AE and [3H]5β-A, and also [3H]5α-androstanedione ([3H]5α-A), [3H]estrone ([3H] E1), and [3H]E2. Note that high levels of exogenous NAD+ abolish 5α-reductase and aromatase activities (Soma et al., 2004). Thus, in the absence of saturating concentrations of exogenous NAD+, we were able to assess activities of 3β-HSD, 5β-reductase, 5α-reductase, and aromatase.
Brain tissues were homogenized in ice-cold SPO4 buffer and centrifuged at 1000 g at 4°C for 30 min, as in Experiment 2A. 200 μL of the supernatant was then incubated with 200 nM [3H]DHEA at 41°C for 90 min. This incubation time was chosen based on timecourse studies (Fig. 1B). Product formation was linear from 0 to 120 min (androgens: r2=0.9980; estrogens: r2=0.8889). Steroids were extracted using diethyl ether, androgens and estrogens were separated using phenolic partitioning, and androgens and estrogens were chromatographed separately as in (Soma et al., 2004). The dpm were corrected for background values and recovery for each assay. Across all assays, average recovery was 84.67 ± 0.88 % for androgens and 14.59 ± 0.59 % for estrogens, similar to previous studies using this protocol (Soma et al., 1999b Soma et al., 2004; Soma et al., 2003; Tam and Schlinger, 2007). All data are presented as fmole per mg protein per min of incubation. Fadrozole, a specific aromatase inhibitor, abolished formation of [3H]estrogens, as in previous studies (Soma et al., 2004). 3[H]AE, 3[H]5β-A and 3[H]E1+E2 were also expressed as a percentage of total 3β-HSD metabolites, to reveal the proportion of each metabolite formed during the assay.
Statistics
All data are presented as mean ± sem. Data were log-transformed, if necessary, to achieve homogeneity of variance and normality of distribution, and analyzed using SPSS 11.0 or Prism 4.0 for Mac OS X. For Experiment 1, we used one-way ANOVAs to examine the effects of season on playback time used to capture subjects, plasma hormone levels, body measures, and brain 3β-HSD activity. A significant main effect of season was broken down using post hoc Fisher's protected least significant difference (LSD) tests. For Experiment 2, we used t-tests to examine the effects of STIs on enzyme activities. Pearson correlational analysis was used to examine the correlation between the amount of playback used to capture subjects and 3β-HSD activity in specific brain regions. We also examined the correlation between 3β-HSD activity and time spent in close proximity to the decoy. All tests were two-tailed and values of p ≤ 0.05 were considered significant.
Results
Experiment 1: Seasonal regulation of brain 3β-HSD activity
There was a significant effect of season on plasma levels of DHEA and T (Table 1) (DHEA: F2,18=3.205, p=0.048; T: F2,18=24.063, p<0.0001). Plasma DHEA levels were highest in the breeding season, lowest during molt, and intermediate in the non-breeding season. Plasma T levels were highest in the breeding season and basal in the molt and non-breeding season. Testis volume and cloacal protuberance length (androgen-dependent) were largest in the breeding season (Table 1).
Table 1.
Seasonal changes in plasma dehydroepiandrosterone (DHEA), testosterone (T), testis volume, and cloacal protuberance length in free-living adult male song sparrows.
| Breeding | Molt | Non-breeding | F statistic | p | |
|---|---|---|---|---|---|
| Plasma DHEA (ng/ml) | 0.48 ± 0.06a | 0.31 ± 0.02b | 0.42 ± 0.04ab | 3.205 | 0.048 |
| Plasma T (ng/ml) | 4.82 ± 1.64a | 0.28 ± 0.06b | 0.20 ± 0.03b | 24.063 | <0.0001 |
| Testes volume (mm3) | 2913.31 ± 350.48a | 73.28 ± 58.58b | 6.91 ± 0.63c | 65.403 | <0.0001 |
| Cloacal protruberance length (mm) | 7.48 ± 0.29a | 5.07 ± 0.37b | 3.66 ± 0.22c | 40.643 | <0.0001 |
Note: Within rows, values with different superscripts differ significantly from each other. n=6 per season.
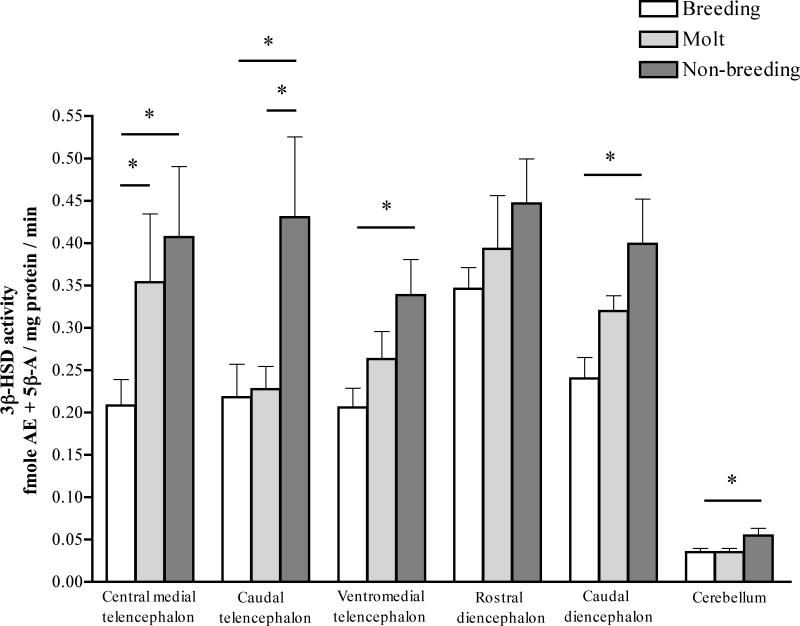
Next, we measured brain 3β-HSD activity in whole homogenates with exogenous NAD+ (Fig. 3). In the central medial telencephalon, 3β-HSD activity changed seasonally (F2,14=6.098, p=0.012), and was lower in the breeding season than molt (Fisher LSD, p=0.018) and non-breeding season (p=0.012). In the caudal telencephalon, 3β-HSD activity also changed seasonally (F2,15=3.841, p=0.045), and was higher in the non-breeding season than molt (p=0.033) and breeding season (p=0.027). In the ventromedial telencephalon, caudal diencephalon and cerebellum, 3β-HSD activity changed seasonally (F2,15=3.857, p=0.045; F2,15= 4.266, p=0.034; F2,15=4.205, p=0.036, respectively), and was higher in the non-breeding season than breeding season (p<0.05 in all 3 cases). No significant seasonal changes were observed in the rostral diencephalon (F2,14=1.037, p=0.38) or optic lobes (F2,15=0.99, p=0.39; data not shown).
Figure 3.
Seasonal changes in baseline brain 3β-HSD activity in wild male song sparrows (n=6 per season). [3H]DHEA was converted to [3H]AE (major product) and [3H]5β-A (minor product). *p<0.05.
Next, we focused on two telencephalic regions in which enzymatic activity changed seasonally and examined whether the amount of playback used to capture subjects was correlated with 3β-HSD activity. Within each season the amount of playback used was not correlated with 3β-HSD activity in the central medial telencephalon or the caudal telencephalon (p>0.22 in all cases). Moreover, when subjects from all seasons were pooled, again the amount of playback used was not correlated with 3β-HSD activity in the central medial telencephalon or the caudal telencephalon (p>0.29 in both cases).
Experiment 2: Social regulation of brain 3β-HSD activity
During the non-breeding season, we exposed wild male song sparrows to an STI or control for 30 min. STIs elicited robust aggression, including singing, “wing waving,” and approaching the decoy closely, as in previous studies (Wingfield and Hahn, 1994). The latency to respond to the STI was 0.61 ± 0.47 min and the latency to sing was 2.78 ± 2.37 min. Response and song latencies for the control group were 16.52 ± 2.90 min and 30.0 ± 0.0 min, respectively.
Experiment 2A: 3β-HSD activity assay with exogenous NAD+
In Experiment 2A, we measured 3β-HSD activity in supernatants with exogenous NAD+. Under these assay conditions, there were no significant effects of STI on 3β-HSD activity, except in the cerebellum, in which STI decreased 3β-HSD activity (Table 2). To assess 5β-reductase activity, we expressed [3H]5β-A as a percentage of total 3β-HSD metabolites ([3H]AE + [3H]5β-A) (as in (Pradhan et al., 2008)). STI significantly increased 5β-reductase activity in the dorsal telencephalon and decreased 5β-reductase activity in the rostral diencephalon (Table 2).
Table 2.
Effect of simulated territorial intrusions on brain 3β-HSD activity in wild non-breeding song sparrows. [3H]DHEA was used as the substrate and exogenous NAD+ was added as the cofactor.
| Total metabolites fmole AE + 5β-A / mg protein / min | 5β-A/Total % | |||||
|---|---|---|---|---|---|---|
| Control | STI | p | Control | STI | p | |
| Rostral telencephalon | 158.62 ± 52.87 (9) | 127.35 ± 18.52 (9) | 0.235 | 1.06 ± 0.69 | 1.78 ± 1.38 | 0.649 |
| Central medial telencephalon | 107.61 ± 27.86 (9) | 119.84 ± 23.52 (9) | 0.757 | 2.53 ± 0.98 | 3.38 ± 1.58 | 0.656 |
| Central lateral telencephalon | 20.14 ± 2.48 (9) | 31.48 ± 8.00 (9) | 0.101 | 7.96 ± 4.18 | 6.29 ± 1.56 | 0.713 |
| Caudal telencephalon | 44.53 ± 7.02 (7) | 65.66 ± 7.23 (7) | 0.519 | 0.33 ± 0.31 | 1.30 ± 0.36 | 0.067 |
| Dorsal telencephalon | 19.71 ± 5.96 (9) | 21.60 ± 5.43 (8) | 0.232 | 20.48 ± 8.65 | 53.14 ± 11.49 | 0.036* |
| Ventromedial telencephalon | 42.04 ± 11.55 (9) | 37.37 ± 21.91 (9) | 0.188 | 32.00 ± 11.30 | 10.27 ± 6.8 | 0.158 |
| NCM | 70.79 ± 11.35 (7) | 60.04 ± 12.88 (9) | 0.606 | 2.31 ± 2.00 | 5.19 ± 2.3 | 0.377 |
| Hippocampus | 22.58 ± 5.81 (8) | 11.22 ± 4.94 (9) | 0.082 | 54.18 ± 15.51 | 37.18 ± 12.25 | 0.398 |
| Rostral diencephalon | 1175.19 ± 537.20 (9) | 600.75 ± 226.25 (9) | 0.339 | 1.30 ± 0.51 | 0.18 ± 0.07 | 0.022* |
| Caudal diencephalon | 126.52 ± 27.01 (8) | 184.42 ± 75.57 (8) | 0.483 | 3.34 ± 1.78 | 1.46 ± 0.82 | 0.351 |
| Cerebellum | 164.92 ± 45.42 (9) | 70.65 ± 16.66 (9) | 0.002** | 0.67 ± 0.37 | 11.74 ± 11.04 | 0.331 |
Note: The sum of the metabolites is an index of 3β-HSD activity and the percentage of 5β-A is an index of 5β-reductase activity. Numbers in parentheses denote sample sizes. Values in boldface indicate a significant difference between groups.
p<0.05,
p<0.01.
Experiment 2B: 3β-HSD activity assay without exogenous NAD+
In Experiment 2B, we focused on telencephalic regions and measured 3β-HSD activity in supernatants without exogenous NAD+. In the central medial telencephalon, STI significantly increased 3β-HSD activity by ∼ 515% (t15=2.524, p=0.023; Fig. 4). Similarly, in the caudal telencephalon, STI significantly increased 3β-HSD activity by ∼242% (t13=3.656, p=0.003; Fig. 4). In contrast, there was no effect of STI on 3β-HSD activity in the rostral telencephalon (Control: 0.97 ± 0.72 fmole/mg protein/min; STI: 0.88 ± 0.34 fmole/mg protein/min; t16=0.119, p=0.907).
Figure 4.
Effect of a simulated territorial intrusion (30 min) on brain 3β-HSD activity in wild male song sparrows during the non-breeding season. NAD+ was not added in this experiment (Expt. 2B). The aggressive challenge rapidly increased 3β-HSD activity in the central medial telencephalon (n=9 control and 8 STI) and caudal telencephalon (n=7 control and 8 STI). *p<0.05, **p<0.01
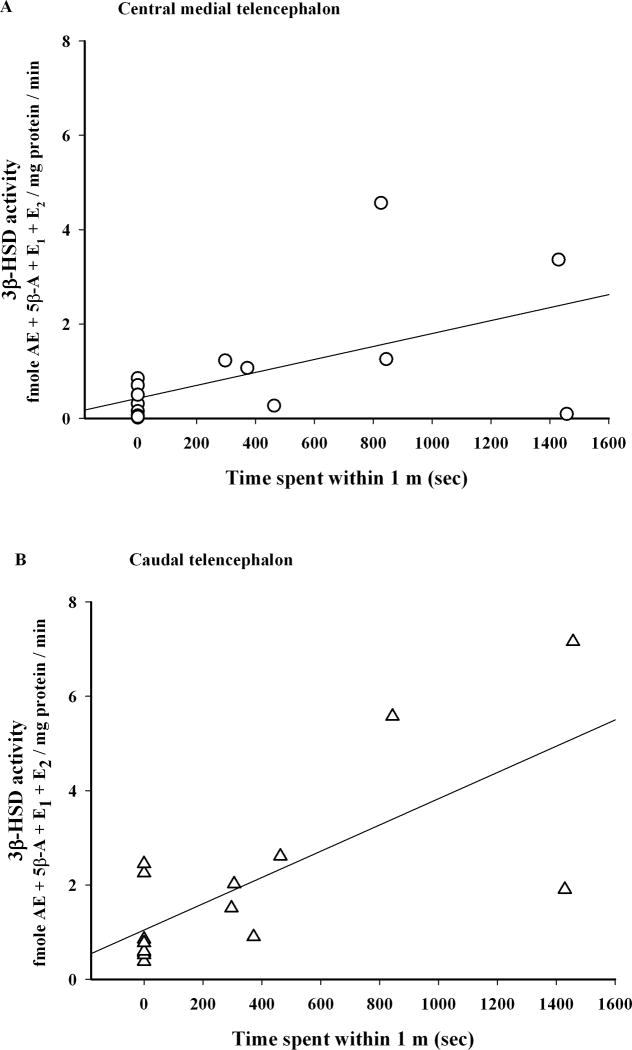
Further, 3β-HSD activity was positively correlated with the time the subject spent within 1 m of the decoy (Fig. 5; Pearson correlation; central medial telencephalon: n=17, r=0.56, p=0.020; caudal telencephalon: n=15, r=0.73, p= 0.002) and also within 5 m of the decoy (Pearson correlation; central medial telencephalon: n=17, r=0.61, p=0.009; caudal telencephalon: n=15, r=0.58, p=0.024).
Figure 5.
Correlation between time spent by the resident within 1 m of the intruder and brain 3β-HSD activity. NAD+ was not added in this experiment (Expt. 2B). Time spent within 1m was positively correlated with 3β-HSD activity in (A) central medial telencephalon (n=9 control and 8 STI) and (B) caudal telencephalon (n=7 control and 8 STI).
Next, we focused on the above two telencephalic regions and examined whether the amount of playback used to capture subjects was correlated with 3β-HSD activity. The amount of playback used to capture control or STI subjects was not correlated with 3β-HSD activity in the central medial telencephalon or the caudal telencephalon (p>0.27 in all cases).
Lastly, we expressed each metabolite of [3H]DHEA as a percentage of total metabolites, to calculate indices of 5β-reductase, 5α-reductase, and aromatase activities (Table 3) (as in (Soma et al., 2004; Pradhan et al., 2008)). STI increased the percentage of [3H]AE in the central medial telencephalon and caudal telencephalon. STI did not affect the percentage of [3H]5β-A in any region. [3H]5α-A was not detected, similar to previous studies (Soma et al., 2004). Surprisingly, STI decreased the percentage of [3H]estrogens (an index of aromatase activity) in the central medial telencephalon and caudal telencephalon. However, the absolute values of [3H]E1+[3H]E2/mg protein/min were not affected by STI in these regions (central medial telencephalon, control: 0.027 ± 0.006 fmole/mg protein/min, STI: 0.026 ± 0.007 fmole/mg protein/min, p=0.98; caudal telencephalon, control: 0.040 ± 0.011 fmole/mg protein/min, STI: 0.026 ± 0.007 fmole/mg/min, p=0.29).
Table 3.
Effect of simulated territorial intrusions (30 min) in brains of wild non-breeding song sparrows. [3H]DHEA was used as a substrate, and no exogenous NAD+ was added. Data are expressed as a percentage of total 3β-HSD metabolites.
| Control | STI | p | |
|---|---|---|---|
| Rostral telencephalon | |||
| AE/Total (%) | 16.6 ± 10.98 | 62.84 ± 15.88 | 0.077 |
| 5β-A/Total (%) | 12.05 ± 10.93 | 6.45 ± 3.48 | 0.889 |
| E1+E2/Total (%) | 60.23 ± 15.93 | 30.71 ± 15.22 | 0.189 |
| Central medial telencephalon | |||
| AE/Total (%) | 48.59 ± 15.56 | 89.23 ± 8.55 | 0.038* |
| 5β-A/Total (%) | 14.16 ± 8.45 | 0.68 ± 0.68 | 0.108 |
| E1+E2/Total (%) | 37.24 ± 13.12 | 10.08 ± 8.62 | 0.018* |
| Caudal telencephalon | |||
| AE/Total (%) | 92.76 ± 1.45 | 96.11 ± 0.56 | 0.043* |
| 5β-A/Total (%) | 1.35 ± 0.76 | 2.32 ± 0.54 | 0.130 |
| E1+E2/Total (%) | 5.90 ± 1.82 | 1.37 ± 0.40 | 0.008** |
Note: Formed [3H]AE was converted to [3H]5β-A, [3H]E1 and [3H]E2. p values are from t tests. Boldface indicates p ≤ 0.05.
Discussion
These results suggest a novel mechanism for regulation of aggression during the non-breeding season, when gonadal androgen synthesis and plasma androgen levels are low. First, brain 3β-HSD activity is chronically upregulated during the non-breeding season. Second, during the non-breeding season, brain 3β-HSD activity is acutely upregulated even further by territorial challenges in a region-specific manner. The conversion of the circulating pro-hormone DHEA to active sex steroids within the brain is critical during the non-breeding season, when local sex steroid signaling is predominant (Schmidt et al., 2008). Lastly, these data suggest that acute social regulation of brain 3β-HSD activity may involve changes in cellular NAD+ concentrations or changes in 3β-HSD specifically within NAD-rich subcellular compartments.
Seasonal regulation of brain 3β-HSD activity
Brain 3β-HSD activity varied seasonally in a region-specific manner (Fig. 3). In the central medial telencephalon (contains septum and BNST), caudal telencephalon (contains song nucleus RA), ventromedial telencephalon (contains taenial amygdala), and caudal diencephalon, 3β-HSD activity was higher in the non-breeding season than the breeding season. Levels during molt were, in general, intermediate. All of the above samples contain sex steroid-sensitive regions; for example, the BNST contains intracellular androgen receptors and estrogen receptors (Adkins-Regan, 2005; Demas et al., 2007). This seasonal pattern of brain 3β-HSD activity is different from that of brain aromatase activity in song sparrows. Soma et al. (2003) found that aromatase activity in the ventromedial telencephalon is lowest in the molt and similarly high in the breeding and non-breeding seasons. Aromatase activity in the diencephalon is higher in the breeding season than molt and the non-breeding season. Taken together, these data suggest that neural steroidogenic enzymes are differentially regulated across seasons. 3β-HSD, the first step in the local conversion of circulating DHEA to active sex steroids, may be a critical rate-limiting step in this multi-stage process. During the non-breeding season, the combination of elevated plasma DHEA, brain 3β-HSD activity, and brain aromatase activity in specific regions could result in high local E2 levels, which promote male aggression (Soma et al., 2000b). Brain steroidogenic enzymes also change seasonally in other species, although these other studies have not looked at 3β-HSD (Pasmanik and Callard, 1988; Foidart et al., 1998; Riters et al., 2001).
Seasonal changes in brain 3β-HSD activity might be regulated by physiological indicators of photoperiod, such as melatonin. Chronic melatonin treatment (mimicking short-day profiles) increases aggression in Siberian hamsters (Demas et al., 2004), and recent data suggest that acute increases in melatonin at night increase the conversion of DHEA to T during aggressive encounters in hamsters (Scotti et al., 2009). Future studies will directly examine the effects of melatonin on brain 3β-HSD activity in song sparrows.
An important question is whether the seasonal changes in brain 3β-HSD activity are the consequence, at least in part, of our method of capture of the wild subjects (which required a minimal amount of playback, but no decoy). This does not appear to be the case. First, there were no seasonal differences in the amount of playback used to capture subjects (∼3.5 min in all 3 seasons, see Methods). Second, the amount of playback used to capture subjects was not correlated with brain 3β-HSD activity within the breeding, molt, or non-breeding seasons (see Results). Similarly, in Experiment 2B, the amount of playback used to capture control or STI individuals was not correlated with 3β-HSD activity. However, it is still possible that the playback used capture to control individuals was enough to regulate steroidogenic enzymes rapidly. In the future, this question can be further addressed in a laboratory study.
Social regulation of brain 3β-HSD, 5β-reductase, and aromatase activities
Simulated territorial intrusions (STI) rapidly elicited aggressive behavior in non-breeding subjects. When exogenous NAD+ was added, there were different effects on 3β-HSD and 5β-reductase activities. While STI had no effect on 3β-HSD activity (except in the cerebellum), STI increased 5β-reductase activity in the dorsal telencephalon (contains song nucleus HVC) and decreased 5β-reductase activity in the rostral diencephalon after 30 min (Table 2). 5β-reductase is considered an “inactivation shunt” that deactivates androgens (Massa et al., 1983). The changes in 5β-reductase activity might regulate the escalation of aggressive behavior during the STI. As the STI continues, territorial owners switch from low intensity aggression (singing) to high intensity aggression (approaching the decoy closely and physical attacks) (Searcy et al., 2006). It is possible that increased 5β-reductase activity in or near HVC reduces singing and that decreased 5β-reductase activity in the rostral diencephalon increases high intensity aggression.
In the absence of exogenous NAD+, the STI rapidly increased brain 3β-HSD activity in behaviorally-relevant portions of the telencephalon (Fig. 4). Moreover, in these telencephalic regions, 3β-HSD activity was positively correlated with the time the subject spent near the decoy. More time spent in close proximity to the intruder reflects a more aggressive response (Nice, 1943; Soma et al., 2000). These rapid changes in brain 3β-HSD activity (200 to 500% increases) are greater than previously reported rapid changes in brain aromatase activity (20 to 90%) (Black et al., 2005; Cornil et al., 2005). These data suggest that rapid changes in brain 3β-HSD activity, in addition to or in the absence of changes in brain aromatase activity, could quickly modulate local E2 levels. Such mechanisms might allow rapid and transient increases in E2 at specific sites, perhaps leading to rapid modulation of behavior (Balthazart and Ball, 2006; Remage-Healey et al., 2008; Cornil, 2009). For example, in mice, E2 rapidly increases aggression under a short photoperiod but not a long photoperiod (Trainor et al., 2007).
Surprisingly, in these telencephalic regions, aggressive encounters rapidly increased the percentage of [3H]AE (relative to total 3β-HSD metabolites) and decreased the percentage of [3H]estrogens (Table 3). However, STI had no effect on the absolute amounts of [3H]E1 + [3H]E2 formed. These data raise the question of whether aromatase was 1) inhibited or 2) functioning at its Vmax in controls because substrates were present at saturating concentrations. While we do not know the brain concentrations of endogenous AE or T in these subjects, the concentrations of formed [3H]AE in the test tubes (0.06 to 0.6 nM) are well below the measured Km of aromatase in songbird brain using thin layer chromatography (13.4 to 29 nM) (Vockel et al., 1988; Schlinger et al., 1994; Soma et al., 1999b). Taken together, the data suggest that aromatase was inhibited after a 30 min STI. Note that social interactions are dynamic, and it is possible that aggressive encounters rapidly increased and then decreased aromatase activity. Future studies should use shorter STI exposure (e.g., 5 min). Rapid and dynamic changes in brain aromatase activity following social interactions have been documented in other species. When male Japanese quail were allowed to interact with a female for 1, 5, or 15 min, preoptic aromatase activity was reduced at 5 min, but similar to control subjects at 1 and 15 min (Cornil et al., 2005). In a sex-changing fish, Lythrypnus dalli, sampled after male removal, the dominant female increases aggressive behavior by 30 min (M. Black, pers. comm.), but brain aromatase activity decreases by 3 hr (Black et al., 2005). In these species, studying androgen-synthesizing enzymes may provide some insight into the regulation of social behavior.
Another interesting point is that after the STI is terminated, aggressive behavior subsides more quickly in the non-breeding season than in the breeding season (Wingfield and Hahn, 1994). Thus, after winter territorial challenges end, brain 3β-HSD or aromatase activities might rapidly return to baseline, resulting in transient pulses of sex steroids in particular regions.
Possible mechanisms for rapid regulation of brain 3β-HSD activity
We detected large changes in 3β-HSD activity within only 30 min, a time frame that strongly suggests non-genomic mechanisms (Cornil et al., 2006). Moreover, these changes were observed only when NAD+ was not added. Thus 3β-HSD activity may be regulated by rapid changes in endogenous NAD+ concentrations, and this hypothesis will be tested in future studies. Cofactor bioavailability regulates the activities of other enzymes in the nervous system, including tyrosine hydroxylase and tryptophan hydroxylase, which are regulated by the availability of the cofactor tetrahydrobiopterin (Thony et al., 2000). In most in vitro enzyme activity assays, saturating levels of cofactors are added to maximize reaction rate, but this may obscure changes in endogenous cofactors. The role of cofactors in neural steroidogenesis is poorly understood and invites further investigation (Agarwal and Auchus, 2005).
Alternatively, the absence of exogenous NAD+ may have allowed us to inadvertently measure 3β-HSD activity specifically within a NAD-rich subcellular compartment, such as mitochondria. 3β-HSD is a membrane-bound enzyme present in both mitochondria and microsomes (endoplasmic reticula) (Schlinger et al., 2008). Mitochondria contain high levels of endogenous NAD+ (regenerated via the electron transport chain). Thus, in the absence of exogenous NAD+, we may have measured 3β-HSD activity specifically in mitochondria. In preliminary studies, we measured brain 3β-HSD activity in several subcellular compartments. Without exogenous NAD+, 3β-HSD activity was detectable in a pellet containing mitochondria and synaptosomes (P2), but not in a pellet containing microsomes (P3) (L. Lau, D. Pradhan, K. Soma, unpublished results), similar to previous studies (Chapman and Sauer, 1979; Cherradi et al., 1994). In Experiment 2A, exogenous NAD+ may have maximized both mitochondrial and microsomal 3β-HSD activities, obscuring a specific change in mitochondrial 3β-HSD activity. Future work will examine whether aggressive interactions stimulate brain 3β-HSD activity within specific subcellular compartments. Interestingly, mitochondria are abundant at presynaptic terminals, and synaptosomes contain high levels of aromatase in songbirds (Rohmann et al., 2007). Also, synaptosomal and microsomal aromatase can be regulated differentially in songbirds (Remage-Healey et al., 2009).
AE and aggressive behavior
Many studies have focused on the importance of T for regulation of aggressive behavior. AE is a precursor to T, and like T, can be aromatized to an estrogen and can bind to the intracellular androgen receptor with high affinity. Thus, AE as well as T might be important for aggressive behavior as well as other behaviors. Interestingly, plasma AE levels are elevated in female spotted hyenas (Glickman et al., 1992) and female ring-tailed lemurs (Drea, 2007), and male guinea pigs (Wichmann et al., 1984). Plasma AE levels are, however, non-detectable in non-breeding song sparrows (K. Soma, unpublished results). Here, we measured the neural conversion of [3H]DHEA to [3H]AE but we could not measure formed [3H]T. In our system, [3H]T runs below the substrate ([3H]DHEA), resulting in very high background values. In the future, it will be important to determine which pathway is more important: DHEA → AE → T → E2 or alternatively DHEA → AE → E1 → E2.
Conclusions
Many studies have examined the regulation and functions of brain aromatase (Trainor et al., 2006; Cornil, 2009). In contrast, brain 3β-HSD has been identified in several species, but little is known about its regulation, and studies linking it to a biological function are lacking. Together, the present results suggest that brain 3β-HSD activity is high during the non-breeding season and rapidly increases even higher during territorial disputes (Fig. 6). Neural androgen synthesis might be elevated and more important during the non-breeding season, when gonadal androgen synthesis is basal. During the breeding season, rapid fluctuations in neural androgen synthesis could still occur; this will be tested in future experiments. Overall, the relative importance of neural and gonadal androgen synthesis appears to be dynamic and fluctuates seasonally. Thus, the Challenge Hypothesis may operate at a local level during the non-breeding season, which represents a large percentage of the lifetimes of most animals. Local steroid signaling may avoid many of the costs of high systemic T levels (Wingfield et al., 2001) and may involve rapid changes at multiple levels, such as steroidogenic enzymes, substrates, cofactors, and receptors. Coordination across these levels is likely to be important for regulating rapid and transient changes in behavior. Such mechanisms produce spatial and temporal specificity of steroid signals and support the emerging concept that locally-synthesized steroids may function more like neuromodulators or neurotransmitters than classical blood-borne hormones (Balthazart and Ball, 2006).
Figure 6.

Hypothesized patterns of gonadal androgen synthesis (solid line) and neural androgen synthesis (broken line) in male song sparrows. Level A, constitutive levels of androgen synthesis for homeostatic functions; Level B, seasonal increase in androgen synthesis (regulated by predictable environmental cues); Level C, facultative and transient increase in androgen synthesis (regulated by unpredictable social cues).

Acknowledgments
We thank Kim Schmidt and Lani Sheldon for help in the field, Loretta Lau for help with assays, and Drs. Colin Brauner and Jeff Richards for comments on the manuscript. This work was supported by grants from NSERC and Brains & Behavior Program at Georgia State University to DSP; NSERC and the Michael Smith Foundation for Health Research (MSFHR) to AEMN; NSF (IBN-0342242) to JCW; and NIH (MH61994) to BAS; and MSFHR and CIHR to KKS.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Adkins-Regan E. Hormones and Animal Social Behavior. Princeton University Press; Princeton and Oxford: 2005. [Google Scholar]
- Agarwal AK, Auchus RJ. Minireview: cellular redox state regulates hydroxysteroid dehydrogenase activity and intracellular hormone potency. Endocrinology. 2005;146:2531–2538. doi: 10.1210/en.2005-0061. [DOI] [PubMed] [Google Scholar]
- Archer J. Testosterone and human aggression: an evaluation of the challenge hypothesis. Neurosci Biobehav Rev. 2006;30:319–345. doi: 10.1016/j.neubiorev.2004.12.007. [DOI] [PubMed] [Google Scholar]
- Balthazart J, Ball GF. Is brain estradiol a hormone or a neurotransmitter? Trends Neurosci. 2006;29:241–249. doi: 10.1016/j.tins.2006.03.004. [DOI] [PubMed] [Google Scholar]
- Black MP, Balthazart J, Baillien M, Grober MS. Socially induced and rapid increases in aggression are inversely related to brain aromatase activity in a sex-changing fish, Lythrypnus dalli. Proc R Soc B. 2005;272:2435–2440. doi: 10.1098/rspb.2005.3210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caldwell GS, Glickman SE, Smith ER. Seasonal Aggression Independent of Seasonal Testosterone in Wood Rats. Proc Natl Acad Sci USA. 1984;81:5255–5257. doi: 10.1073/pnas.81.16.5255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chapman JC, Sauer LA. Intracellular localization and properties of 3β-hydroxysteroid dehydrogenase/isomerase in the adrenal cortex. J Biol Chem. 1979;254:6624–6630. [PubMed] [Google Scholar]
- Cherradi N, Defaye G, Chambaz EM. Characterization of the 3β-hydroxysteroid dehydrogenase activity associated with bovine adrenocortical mitochondria. Endocrinology. 1994;134:1358–1364. doi: 10.1210/endo.134.3.8119176. [DOI] [PubMed] [Google Scholar]
- Cornil CA. Rapid Regulation of Brain Oestrogen Synthesis: The Behavioural Roles of Oestrogens and their. Fates J Neuroendocrinol. 2009;21:217–226. doi: 10.1111/j.1365-2826.2009.01822.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cornil CA, Ball GF, Balthazart J. Functional significance of the rapid regulation of brain estrogen action: Where do the estrogens come from? Brain Res. 2006;1126:2–26. doi: 10.1016/j.brainres.2006.07.098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cornil CA, Dalla C, Papadopoulou-Daifoti Z, Baillien M, Dejace C, Ball GF, Balthazart J. Rapid decreases in preoptic aromatase activity and brain monoamine concentrations after engaging in male sexual behavior. Endocrinology. 2005;146:3809–3820. doi: 10.1210/en.2005-0441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Demas GE, Cooper MA, Albers HE, Soma KK. Novel Mechanisms underlying neuroendocrine regulation of aggression: A synthesis of rodent, avian, and primate studies. In: Blaustein JD, editor. Neurochemistry and Neuroendocrinology. Springer Science+Business Media, LLC; New York: 2007. pp. 337–372. [Google Scholar]
- Demas GE, Polacek KM, Durazzo A, Jasnow AM. Adrenal hormones mediate melatonin-induced increases in aggression in male Siberian hamsters Phodopus sungorus. Horm Behav. 2004;46:582–591. doi: 10.1016/j.yhbeh.2004.07.001. [DOI] [PubMed] [Google Scholar]
- Drea CM. Sex and seasonal differences in aggression and steroid secretion in Lemur catta: are socially dominant females hormonally ‘masculinized’? Horm Behav. 2007;51:555–567. doi: 10.1016/j.yhbeh.2007.02.006. [DOI] [PubMed] [Google Scholar]
- Foidart A, Silverin B, Baillien M, Harada N, Balthazart J. Neuroanatomical Distribution and Variations across the Reproductive Cycle of Aromatase Activity and Aromatase-Immunoreactive Cells in the Pied Flycatcher (Ficedula hypoleuca) Horm Behav. 1998;33:180–196. doi: 10.1006/hbeh.1998.1448. [DOI] [PubMed] [Google Scholar]
- Francis RC, Soma K, Fernald RD. Social regulation of the brain-pituitary-gonadal axis. Proc Natl Acad Sci USA. 1993;90:7794–7798. doi: 10.1073/pnas.90.16.7794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glickman SE, Frank LG, Licht P, Yalcinkaya T, Siiteri PK, Davidson J. Sexual differentiation of the female spotted hyena One of nature's experiments. Ann NY Acad Sci. 1992;662:135–159. doi: 10.1111/j.1749-6632.1992.tb22858.x. [DOI] [PubMed] [Google Scholar]
- Goodson JL, Saldanha CJ, Hahn TP, Soma KK. Recent advances in behavioral neuroendocrinology: insights from studies on birds. Horm Behav. 2005;48:461–473. doi: 10.1016/j.yhbeh.2005.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gower DB, Cooke GM. Regulation of steroid-transforming enzymes by endogenous steroids. J Steroid Biochem. 1983;19:1527–1556. doi: 10.1016/0022-4731(83)91130-5. [DOI] [PubMed] [Google Scholar]
- Goymann W. Social modulation of androgens in male birds. Gen Comp Endocrinol. 2009;163:149–157. doi: 10.1016/j.ygcen.2008.11.027. [DOI] [PubMed] [Google Scholar]
- Hajszan T, MacLusky NJ, Leranth C. Dehydroepiandrosterone increases hippocampal spine synapse density in ovariectomized female rats. Endocrinology. 2004;145:1042–1045. doi: 10.1210/en.2003-1252. [DOI] [PubMed] [Google Scholar]
- Insel TR, Fernald RD. How the brain processes social information: searching for the social brain. Annu Rev Neurosci. 2004;27:697–722. doi: 10.1146/annurev.neuro.27.070203.144148. [DOI] [PubMed] [Google Scholar]
- Labrie F, Luu-The V, Belanger A, Lin SX, Simard J, Pelletier G, Labrie C. Is dehydroepiandrosterone a hormone? J Endocrinol. 2005;187:169–196. doi: 10.1677/joe.1.06264. [DOI] [PubMed] [Google Scholar]
- London SE, Monks DA, Wade J, Schlinger BA. Widespread capacity for steroid synthesis in the avian brain and song system. Endocrinology. 2006;147:5975–5987. doi: 10.1210/en.2006-0154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Massa R, Bottoni L, Lucini V. Brain testosterone metabolism and sexual behaviour in birds. In: Balthazart B, Prove E, Gilles R, editors. Hormones and Behaviour in Higher Vertebrates. Springer-Verlag; Berlin: 1983. pp. 230–236. [Google Scholar]
- Mellon SH, Griffin LD. Neurosteroids: biochemistry and clinical significance. Trend Endocrinol Metabol. 2002;13:35–43. doi: 10.1016/s1043-2760(01)00503-3. [DOI] [PubMed] [Google Scholar]
- Newman AE, Pradhan DS, Soma KK. Dehydroepiandrosterone and corticosterone are regulated by season and acute stress in a wild songbird: jugular versus brachial plasma. Endocrinology. 2008;149:2537–2545. doi: 10.1210/en.2007-1363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newman AE, Soma KK. Corticosterone and dehydroepiandrosterone in songbird plasma and brain: effects of season and acute stress. Eur J Neurosci. 2009;29:1905–1914. doi: 10.1111/j.1460-9568.2009.06748.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nice MM. Studies in the life history of the Song Sparrow. Vol. 6. New York: Transactions of the Linnaean Society of New York; 1943. pp. 1–328. [Google Scholar]
- Pasmanik M, Callard GV. Changes in brain aromatase and 5α-reductase activities correlate significantly with seasonal reproductive cycles in goldfish Carassius auratus. Endocrinology. 1988;122:1349–1356. doi: 10.1210/endo-122-4-1349. [DOI] [PubMed] [Google Scholar]
- Payne AH, Hales DB. Overview of steroidogenic enzymes in the pathway from cholesterol to active steroid hormones. Endocr Rev. 2004;25:947–970. doi: 10.1210/er.2003-0030. [DOI] [PubMed] [Google Scholar]
- Pradhan DS, Yu Y, Soma KK. Rapid estrogen regulation of DHEA metabolism in the male and female songbird brain. J Neurochem. 2008;104:244–253. doi: 10.1111/j.1471-4159.2007.04953.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Remage-Healey L, Maidment NT, Schlinger BA. Forebrain steroid levels fluctuate rapidly during social interactions. Nat Neurosci. 2008;11:1327–1334. doi: 10.1038/nn.2200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Remage-Healey L, Oyama RK, Schlinger BA. Elevated aromatase activity in forebrain synaptic terminals during song. J Neuroendocrinol. 2009;21:191–199. doi: 10.1111/j.1365-2826.2009.01820.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riters LV, Baillien M, Eens M, Pinxten R, Foidart A, Ball GF, Balthazart J. Seasonal variation in androgen-metabolizing enzymes in the diencephalon and telencephalon of the male European starling (Sturnus vulgaris) J Neuroendocrinol. 2001;13:985–997. doi: 10.1046/j.1365-2826.2001.00723.x. [DOI] [PubMed] [Google Scholar]
- Robinson GE, Fernald RD, Clayton DF. Genes and social behavior. Science. 2008;322:896–900. doi: 10.1126/science.1159277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rohmann KN, Schlinger BA, Saldanha CJ. Subcellular compartmentalization of aromatase is sexually dimorphic in the adult zebra finch brain. Dev Neurobiol. 2007;67:1–9. doi: 10.1002/dneu.20303. [DOI] [PubMed] [Google Scholar]
- Schlinger BA, Pradhan DS, Soma KK. 3β-HSD activates DHEA in the songbird brain. Neurochem Int. 2008;52:611–620. doi: 10.1016/j.neuint.2007.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlinger BA, Amur-Umarjee S, Shen P, Campagnoni AT, Arnold AP. Neuronal and non-neuronal aromatase in primary cultures of developing zebra finch telencephalon. J Neurosci. 1994;14:7541–7552. doi: 10.1523/JNEUROSCI.14-12-07541.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt KL, Pradhan DS, Shah AH, Charlier TD, Chin EH, Soma KK. Neurosteroids, immunosteroids, and the Balkanization of endocrinology. Gen Comp Endocrinol. 2008;157:266–274. doi: 10.1016/j.ygcen.2008.03.025. [DOI] [PubMed] [Google Scholar]
- Scotti MA, Schmidt KL, Newman AE, Bonu T, Soma KK, Demas GE. Aggressive encounters differentially affect serum dehydroepiandrosterone and testosterone concentrations in male Siberian hamsters (Phodopus sungorus) Horm Behav. 2009 doi: 10.1016/j.yhbeh.2009.07.004. in press. [DOI] [PubMed] [Google Scholar]
- Searcy WA, Anderson RC, Nowicki S. Bird song as a signal of aggressive intent. Behav Ecol Sociobiol. 2006;60:234–241. [Google Scholar]
- Soma KK, Scotti ML, Newman AE, Charlier TD, Demas GE. Novel mechanisms for neuroendocrine regulation of aggression. Front Neuroendocrinol. 2008;29:476–489. doi: 10.1016/j.yfrne.2007.12.003. [DOI] [PubMed] [Google Scholar]
- Soma KK. Testosterone and aggression: Berthold, birds and beyond. J Neuroendocrinol. 2006;18:543–551. doi: 10.1111/j.1365-2826.2006.01440.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soma KK, Alday NA, Hau M, Schlinger BA. Dehydroepiandrosterone metabolism by 3β-hydroxysteroid dehydrogenase/Δ5-Δ4 isomerase in adult zebra finch brain: sex difference and rapid effect of stress. Endocrinol. 2004;145:1668–1677. doi: 10.1210/en.2003-0883. [DOI] [PubMed] [Google Scholar]
- Soma KK, Schlinger BA, Wingfield JC, Saldanha CJ. Brain aromatase, 5α-reductase, and 5β-reductase change seasonally in wild male song sparrows: Relationship to aggressive and sexual behavior. J Neurobiol. 2003;56:209–221. doi: 10.1002/neu.10225. [DOI] [PubMed] [Google Scholar]
- Soma KK, Wissman AM, Brenowitz EA, Wingfield JC. Dehydroepiandrosterone (DHEA) increases territorial song and the size of an associated brain region in a male songbird. Horm Behav. 2002;41:203–212. doi: 10.1006/hbeh.2001.1750. [DOI] [PubMed] [Google Scholar]
- Soma KK, Wingfield JC. Dehydroepiandrosterone in songbird plasma: seasonal regulation and relationship to territorial aggression. Gen Comp Endocrinol. 2001;123:144–155. doi: 10.1006/gcen.2001.7657. [DOI] [PubMed] [Google Scholar]
- Soma KK, Sullivan KA, Tramontin AD, Saldanha CJ, Schlinger BA, Wingfield JC. Acute and chronic effects of an aromatase inhibitor on territorial aggression in breeding and nonbreeding male song sparrows. J Comp Physiol [A] 2000a;186:759–769. doi: 10.1007/s003590000129. [DOI] [PubMed] [Google Scholar]
- Soma KK, Tramontin AD, Wingfield JC. Oestrogen regulates male aggression in the non-breeding season. Proc Biol Sci B. 2000b;267:1089–1096. doi: 10.1098/rspb.2000.1113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soma KK, Sullivan K, Wingfield JC. Combined aromatase inhibitor and antiandrogen treatment decreases territorial aggression in a wild songbird during the nonbreeding season. Gen Comp Endocrinol. 1999a;115:442–453. doi: 10.1006/gcen.1999.7334. [DOI] [PubMed] [Google Scholar]
- Soma KK, Bindra RK, Gee J, Wingfield JC, Schlinger BA. Androgen-metabolizing enzymes show region-specific changes across the breeding season in the brain of a wild songbird. J Neurobiol. 1999b;41:176–188. [PubMed] [Google Scholar]
- Tam H, Schlinger BA. Activities of 3β-HSD and aromatase in slices of developing and adult zebra finch brain. Gen Comp Endocrinol. 2007;150:26–33. doi: 10.1016/j.ygcen.2006.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thony B, Auerbach G, Blau N. Tetrahydrobiopterin biosynthesis, regeneration and functions. Biochem J. 2000;347(Pt 1):1–16. [PMC free article] [PubMed] [Google Scholar]
- Trainor BC, Kyomen HH, Marler CA. Estrogenic encounters: how interactions between aromatase and the environment modulate aggression. Front Neuroendocrinol. 2006;27:170–179. doi: 10.1016/j.yfrne.2005.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trainor BC, Lin S, Finy MS, Rowland MR, Nelson RJ. Photoperiod reverses the effects of estrogens on male aggression via genomic and nongenomic pathways. Proc Natl Acad Sci USA. 2007;104:9840–9845. doi: 10.1073/pnas.0701819104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vanson A, Arnold AP, Schlinger BA. 3β-hydroxysteroid dehydrogenase/isomerase and aromatase activity in primary cultures of developing zebra finch telencephalon: dehydroepiandrosterone as substrate for synthesis of androstenedione and estrogens. Gen Comp Endocrinol. 1996;102:342–350. doi: 10.1006/gcen.1996.0077. [DOI] [PubMed] [Google Scholar]
- Vockel A, Prove E, Balthazart J. Changes in the activity of testosterone-metabolizing enzymes in the brain of male and female zebra finches during the post-hatching period. Brain Res. 1988;463:330–340. doi: 10.1016/0006-8993(88)90406-4. [DOI] [PubMed] [Google Scholar]
- Wichmann U, Wichmann G, Krause W. Serum levels of testosterone precursors, testosterone and estradiol in 10 animal species. Exp Clin Endocrinol. 1984;83:283–290. doi: 10.1055/s-0029-1210342. [DOI] [PubMed] [Google Scholar]
- Widstrom RL, Dillon JS. Is there a receptor for dehydroepiandrosterone or dehydroepiandrosterone sulfate? Semin Reprod Med. 2004;22:289–298. doi: 10.1055/s-2004-861546. [DOI] [PubMed] [Google Scholar]
- Wingfield JC, Hahn TP. Testosterone and territorial behavior in sedentary and migratory sparrows. Anim Behav. 1994;47:77–89. [Google Scholar]
- Wingfield JC, Hegner RE, Dufty AM, Ball GF. The challenge hypothesis -theoretical implications for patterns of testosterone secretion, mating systems, and breeding. Strategies Am Nat. 1990;136:829–846. [Google Scholar]
- Wingfield JC, Lynn S, Soma KK. Avoiding the ‘costs’ of testosterone: ecological bases of hormone-behavior interactions. Brain Behav Evol. 2001;57:239–251. doi: 10.1159/000047243. [DOI] [PubMed] [Google Scholar]
- Yates J, Deshpande N. Kinetic studies on the enzymes catalysing the conversion of 17α-hydroxyprogesterone and dehydroepiandrosterone to androstenedione in the human adrenal gland in vitro. J Endocrinol. 1974;60:27–35. doi: 10.1677/joe.0.0600027. [DOI] [PubMed] [Google Scholar]