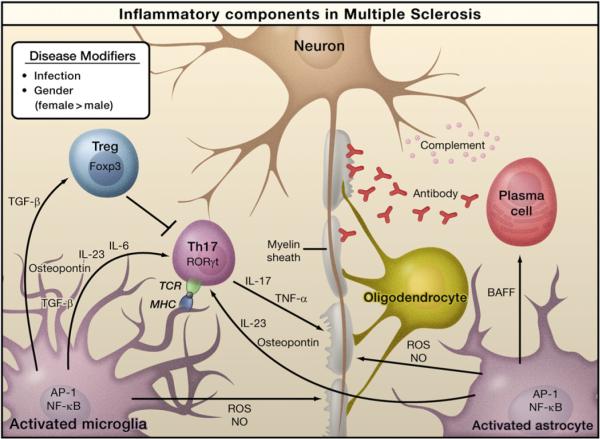
Figure 4. Inflammation in Multiple Sclerosis.
Infection by bacteria or viruses or other environmental stimuli trigger the activation of microglia and astrocytes in multiple sclerosis (MS), leading to the production of proinflammatory cytokines through activation of the transcription factors NF-κB and AP-1. Naive T cells recognize myelin-derived antigen presented in the context of MHC molecules by antigen-presenting cells. In the presence of IL-6 and TGF-β, the naïve T cells are induced to express retinoic acid receptor-related orphan receptor γt (RORγt) and differentiate into Th17 cells. Activated microglia and astrocytes secrete IL-23 and osteopontin, which induce Th17 cells to secrete IL-17 and TNF-α resulting in damage to the myelin sheath that protects nerve axons. Activated astrocytes produce BAFF, a survival factor for autoreactive B cells, which differentiate into plasma cells and produce anti-myelin antibodies. Activated microglia and astrocytes are also sources of reactive oxygen species (ROS) and nitric oxide (NO), which contribute to the destruction of the myelin sheath and of the neurons themselves. Regulatory T cells (Treg) that express Foxp3 suppress the activity of Th17 cells and thus help to suppress inflammation.