Abstract
Background
The molecular mechanisms underlying the pathophysiology of irritable bowel syndrome (IBS) are poorly understood. One mechanism may involve increased intestinal permeability that is reversed with glutamine supplementation. Our goal was to evaluate the expression of glutamine synthetase and its complementary miRNA in blood microvesicles and gut tissues of IBS patients with increased intestinal membrane permeability.
Methods
We evaluated 19 diarrhea-predominant IBS patients and 10 controls for intestinal membrane permeability using the lactulose/mannitol method. miRNA expression was evaluated in blood microvesicles and gut tissue. To further confirm the relationship between miRNA and glutamine synthetase expression, cell culture experiments were conducted. Glutamine synthetase was also evaluated in the gut tissues of patients.
Results
A subset of IBS patients (8/19, 42%) had increased intestinal membrane permeability and decreased glutamine synthetase expression compared to IBS patients with normal membrane permeability and to controls. Expression of miR-29a was increased in blood microvesicles, small bowel and colon tissues of IBS patients with increased intestinal membrane permeability. Increased intestinal permeability was modulated by miR-29a which has a complementary site in the 3'-UTR of the GLUL gene.
Conclusions
The results support the conclusion that GLUL regulates intestinal membrane permeability and miR-29a regulates both GLUL and intestinal membrane permeability. The data suggests that miR-29a effects on intestinal membrane permeability may be due to its regulation of GLUL. Targeting this signaling pathway could lead to a new therapeutic approach to the treatment of patients with IBS, especially because small molecules that mimic or inhibit miRNA-based mechanisms are readily available.
Keywords: Irritable bowel syndrome (IBS), miRNA, miR-29a, blood microvesicles, glutamine synthetase gene (GLUL), glutamine synthetase, intestinal membrane permeability
INTRODUCTION
Irritable bowel syndrome (IBS) is one of the most frequent functional gastrointestinal disorders seen by physicians. It affects up to 20% of the United States population.1 However, the pathophysiological mechanisms underlying abdominal pain and chronic symptoms in IBS are not well understood.2, 3 Several studies have recently shown that patients with diarrhea-predominant IBS have increased intestinal membrane permeability.4–7 Low-grade inflammation has also been reported in mucosal biopsies of some patients with increased intestinal membrane permeability.5 Indeed, patients with both inflammatory and non-inflammatory GI conditions such as inflammatory bowel disease, irritable bowel syndrome, celiac sprue, and acute alcoholic gastroenteritis have increased gut permeability.7–10 Acute symptoms in these disorders – abdominal pain, diarrhea, urgency and bloating – usually coincide with acute inflammation (as is seen in patients with IBS). Transient inflammation of the gut may cause sensitization of enteric neurons which persists long after resolution of the inflammation (post-infectious IBS)6. Several recent animal models have also been reported in which there is alteration of motility and/or sensory function following a transient inflammatory event. 11–13
Glutamine synthetase (GS) catalyzes the conversion of ammonia and glutamate to glutamine. It plays a major role in cell signaling, ammonia detoxification, inter-organ nitrogen flux, and acid-base homeostasis. Congenital glutamine deficiencies have been reported in children with glutamine synthetase gene (GLUL) mutations.14 Because of the multiple functions and importance of glutamine synthetase in cellular metabolism, both catalytic activities and synthesis are highly regulated. Previously, it has been well established that deficiencies in glutamine may lead to increased membrane permeability and supplementation with glutamine can restore intestinal membrane permeability.15–17 Thus, conditions in which decreased levels of intestinal glutamine synthetase are present may lead to low levels of available glutamine and increased intestinal membrane permeability.
Blood microvesicles are circular membrane fragments that are shed from the cell surface, and act as cell-to-cell and cell-to-organ communicators. Virtually all cells are able to release small-size vesicles (microvesicles) derived from the plasma membrane (microparticles) or from multivesicular endocytic compartments (exosomes). Several recent studies have also shown that shedding of membrane-derived microvesicles accompanies cell activation.18–25 Activation of microvesicles may serve as a physiological vehicle to deliver diverse molecules to cells, and ultimately to functional organs. Changes in molecular signature and number of microvesicles may be used to characterize certain disease processes and allow the development of new diagnostic strategies.26, 27
MicroRNAs (miRNAs) are a class of 21–23 nucleotide long non-coding RNA molecules. They are cleaved from 70- to 100-nucleotide hairpin pre-miRNA precursors. Single-stranded miRNAs bind through partial sequence complementarity to the 3'-UTR of target mRNAs and cause a block of translation as well as some degree of mRNA degradation. miRNAs are involved in the regulation of gene expression in critical biological processes, including development, differentiation, apoptosis and proliferation, through imperfect pairing with target mRNAs of protein coding genes.28, 29 The miRNA (miR)-29 family (29a, 29b, 29c) has significant complementarities to the 3`-UTRs of DNA methyltransferase 3A and -3B (de novo methyltransferases), two key enzymes involved in DNA methylation.30 Although a role for aberrant miRNA expression in gastrointestinal disorders has been postulated, the pathophysiologic role and relevance of the expression of specific miRNAs in patients with IBS has not been reported. The objective of our study was to evaluate the role of aberrantly expressed miRNA and its direct target genes in blood microvesicles and gut tissues of IBS patients who exhibit increased intestinal membrane permeability.
METHODS
Human subjects
This study was approved by the University and Veterans Administration Institutional Review Boards. Diarrhea-predominate IBS patients and healthy controls were recruited via posted advertisements. All subjects provided written informed consent during an introductory session prior to the start of the study. During the same session, participants underwent (1) a complete physical exam and (2) a standardized lactulose breath test as previously described.31,32 Also, blood was drawn for analysis of blood microvesicles and to determine an anti-endomysial antibody titer. None of the control subjects had any evidence of acute or chronic abdominal pain or IBS. The diagnosis or exclusion of IBS was made by the same gastroenterologist who examined patients based on the ROME III criteria and exclusion of organic disease.33 All patients had IBS symptoms for at least 5 years. None of the participants were allowed to ingest nonsteroidal anti-inflammatory drugs or alcohol for at least 3 weeks prior to the study.
Seven days later, all subjects that qualified for the study were brought back and underwent membrane permeability testing using lactulose/mannitol kits (Genova Diagnostics®, Asheville, NC). Seven days following membrane permeability testing, all subjects underwent endoscopy during which biopsies of the duodenum and colon were taken. For each patient, multiple random biopsies were taken from the duodenum and throughout the colon. Inflammation of the duodenum and/or colon was not an exclusion criterion for IBS patients. Specimens were stained with hematoxylin and eosin for light microscopy. The same blinded pathologist performed histological analyses on the tissues and used standard pathologic criteria to determine the presence of any inflammation in the duodenum and/or colon. In addition, biopsied tissues were prepared for total RNA extravasations for miRNA microarray profiling and real time PCR as well as western blots.
Intestinal Membrane Permeability Testing
All subjects emptied their bladders and then ingested 5 g of lactulose + 2 g of mannitol dissolved in 100 ml of water. Urine was collected from 0–5 hours and was analyzed for lactulose and mannitol. Urine was also collected from 6–24 hours and was analyzed to obtain a measure of colonic permeability.34 A lactulose/mannitol excretion ratio ≥0.07 was taken to indicate the presence of an increase in permeability (permeability between adjacent intestinal epithelial cells).
Blood Microvesicle Isolation
Blood microvesicles were isolated using the combined methods of Leonards & Ohki35, Victoria et al36 and Hunter & Marsh’s lab provided protocol (see supplementary)
miRNA Analysis
Total RNA was isolated from blood microvesicles and tissue biopsies by Trizol (Invitrogen, Carlsbad, CA), according to the manufacturer's recommendations. RNA was labeled and hybridized on miRNA microarray chips as previously described.37 (see supplementary).
Quantitative Real-Time PCR
To validate the finding of increased expression of miR-29a, we collected blood microvesicles, and small bowel and colon tissue from 8 IBS patients with increased membrane permeability, 8 IBS patients with normal membrane permeability, and compared them to 8 normal controls. Quantitative real-time PCR for miR-29a was done using a TaqMan miRNA Assay kit (see supplementary).
Immunoblotting
Protein extraction was done from the same small bowel and colon tissues obtained above to detect glutamine synthetase expression (see supplementary).
Cell Culture
Human colonic epithelial cells (FHC) and human small intestinal epithelial cells (FHs74Int) were used for this study. Both FHC and FHs74Int cells were cultured as recommended by the supplier (ATCC, Manassas, VA) and we followed the original growth protocol descriptions38,39 (see supplementary).
Transfections and Reagents
Pre-miRNA precursors and specific inhibitors of miR-29a and negative controls were purchased from Ambion (Austin, TX) (see supplementary).
Luciferase Assay
The genomic region 1.8 kb 3’UTR site of GLUL was amplified using primers 5′-AGC CTG GCT TGG GAC TAA AT-3′ (forward) and 5′-TGC TTT GGG CAG TTA CAC AG-3′ (reverse). Due to the size and CG content, PCR amplification was done using the Herculase II fusion DNA polymerase (Stratagene). The resulting fragments containing intact putative miR-29a recognition sequences from the 3'-UTR of GLUL or with random mutations were cloned in the pGL3 basic vector (Promega), the pMIR-REPORT luciferase expression vector, or the pGL3-TK-luciferase vector (from Ambion). Both FHC and FHs74Int cells were cotransfected using Transit-TKO (Invitrogen) with the reporter plasmids and either the GLUL wild type or mutant vector. Luciferase assays were done 48 h post-transfection following the manufacturer's protocol (Dual-luciferase reporter assay system; Promega). Firefly luciferase activity was normalized to Renilla luciferase activity for each sample.
In Vitro Membrane Permeability
To further clarify the mechanisms between enhanced expression of miR-29a and increased intestinal permeability, we designed an In Vitro Permeability Assay to test how miR-29a may regulate membrane permeability. FHC colon and FHs74Int small bowel epithelial cells were transfected with 100 nmol/L miR-29a–specific or control miRNA precursors / inhibitors, and the permeability index was assessed after 48 hours using In Vitro Permeability Assay Kit (purchased from Millipore Inc). We also silenced GLUL to determine if it modulated permeability. FHC and FHs74Int cell lines were transfected with 200 nmol/L GLUL siRNA, control siRNA, and negative control. In vitro epithelial permeability was assessed after 48 hours. The method of detection is illustrated in supplementary Figure 1. The in vitro permeability experiments were repeated 4 times.
Confocal Fluorescence Microscopy
FHC human colon and FHs74Int small intestinal epithelial cells were transfected with miRNA precursors, as described above, plated onto fibronectin-coated chamber slides (BD Biosciences) and stained on day 3 (detail see supplementary).
Transmission Electron Microscopy (TEM)
Blood micro vesicles in suspension were fixed for TEM (detail see supplementary).
STATISTICAL ANALYSIS
Statistical analysis was done using GeneSpring GX software version 7.3 (Agilent Technologies) and Prism version 6. One way ANOVA was done followed by Tukey comparison. Analysis of variance (ANOVA) was also done followed by the Benjamini and Hochberg correction for false-positive reduction. T-tests were also used to analyze all data. Values are expressed as mean ± standard deviation (SD). Tissue and blood samples from IBS patients (and controls) were paired for comparison based on close matching for age and sex in all experiments.
Analysis of Microarray Data
Raw data were normalized and analyzed by GeneSpring GX software version 7.3 (Agilent Technologies). The GeneSpring software generated unique values for each miRNA species, determining the average of the results from four spots. Samples were normalized using the on-chip median normalization feature of GeneSpring software; the result for each cell line time course experiment was normalized to that at time zero.
miRNAs showing an altered expression across the time course were identified using the filter of the n-fold-change tool. To this end, a GeneSpring GX filter with the n-fold-change tool was used to point out the miRNAs that, for at least two cell lines, were 1.5-fold upregulated at 24 h compared to expression levels at time zero. The 1.5-fold-change threshold was chosen on the basis of previously published studies employing these particular types of microarrays.31
Analysis of variance (ANOVA) with the Benjamini and Hochberg correction for false-positive reduction was used to compare the average values obtained with miRNA probes in all cell lines at 24 and 48 h with values at time zero. Hierarchical cluster analysis was performed using average linkage and Pearson correlation as a measure of similarity.
RESULTS
A total of 19 diarrhea-predominant IBS patients (19 white females, mean age 27 + 3.3 years) and 10 healthy controls (10 white females, white, mean age 29 + 5.2 years) participated in the current study. All participants had a negative hydrogen breath test for bacterial overgrowth and a negative antiendomysial antibody titer. Biopsies of the duodenum and colon from all participants were stained with hematoxylin and eosin for light microscopy and did not reveal any evidence of intestinal inflammation.
Intestinal Membrane Permeability
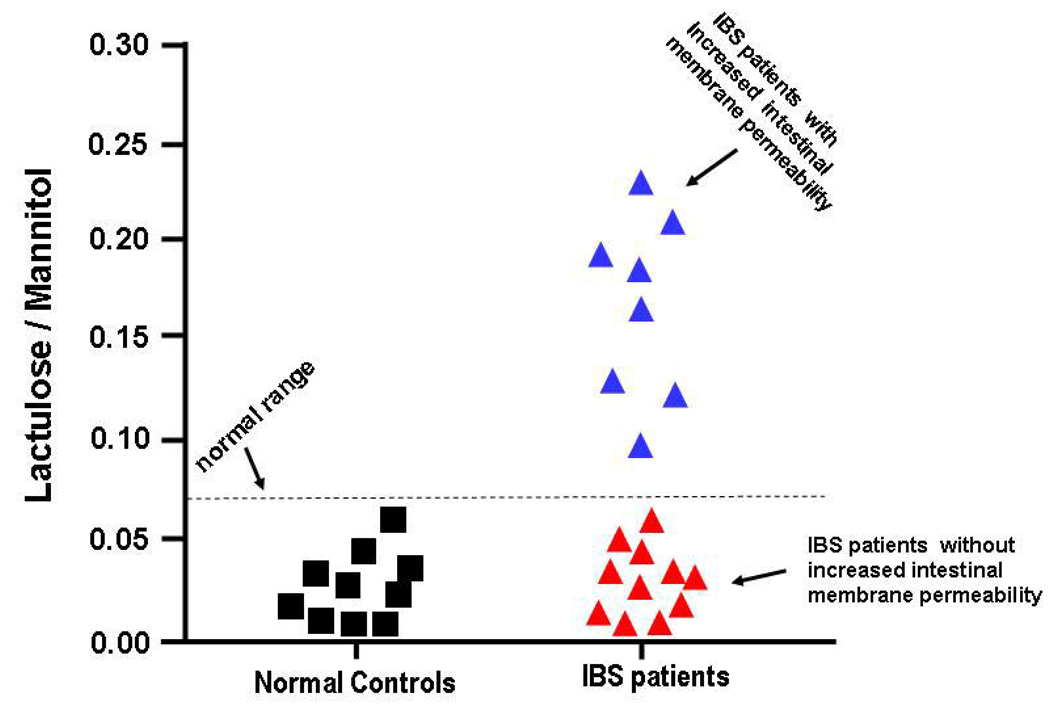
A subset of IBS patients, 8/19 (42%) had increased intestinal membrane permeability with an elevated lactulose/mannitol ratio of ≥0.07, whereas, 11/19(58%) of the IBS patients and all of the controls had a lactulose/mannitol ratio of <0.07 (See Figure 1). The results for lactulose/mannitol excretion were consistent with increased intestinal membrane permeability of both the small intestine and colon for urine collected during hours 0–5 hours and for urine collected during hours 6–24.
Figure 1.
This figure depicts 0–5 hour collection for lactulose/mannitol excretion ratios. A lactulose/mannitol excretion ratio ≥0.07 indicated increased intestinal permeability.
miRNA Array of Blood Microvesicles
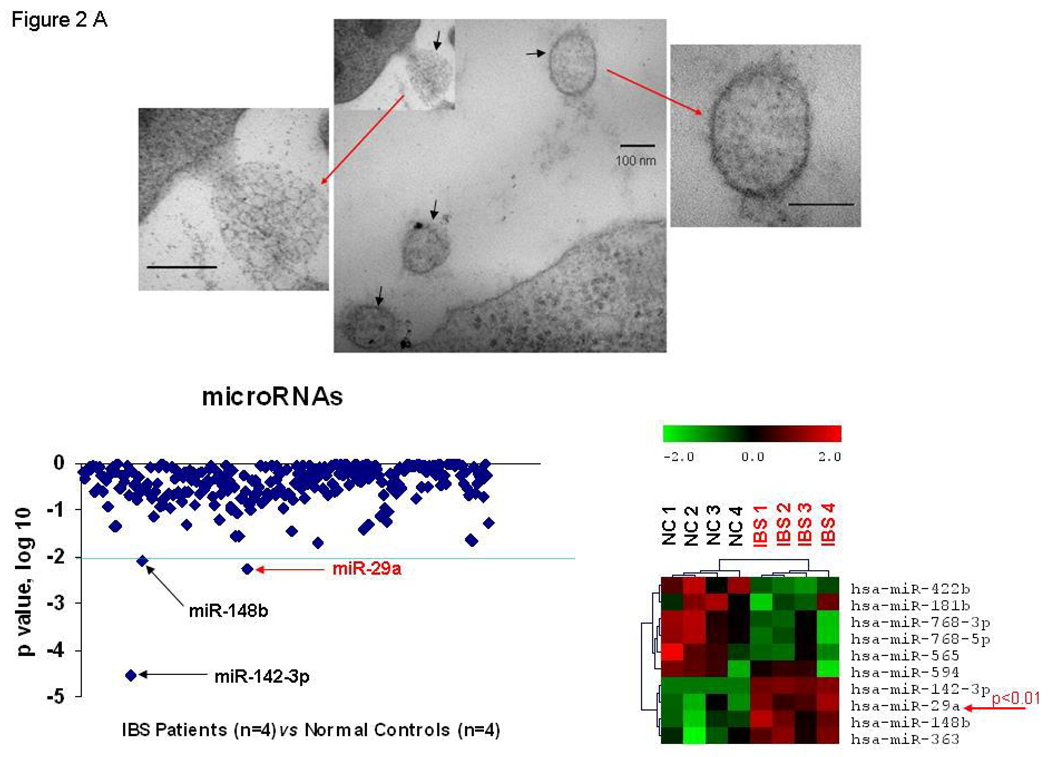
We took advantage of hybridization based miRNA microarray techniques to profile and compare blood-microvesicles from IBS patients with increased membrane permeability and normal controls as described above. Upper panel of Figure 2A illustrates electron microscope picture of blood-microvesicle from one of the participants. We found several miRNAs whose expression was consistently up-regulated in blood microvesicles samples from IBS patients with increased membrane permeability compared to controls (Figure 2A-lower, left panel). Three up-regulated miRNAs with a p<0.001 were identified: miR-142-3p, miRNA-29a and miR-148b (Figure 2A-lower, left panel). The blood microvesicle miRNA array data exhibited a unique miRNA expression profile for IBS patients with increased membrane permeability compared to controls (Figure 2A-lower, right panel). This cluster analysis distinguished expression of specific miRNAs that were either significantly decreased or significantly increased in the microvesicles of these patients (Figure 2A-lower, right panel).
Figure 2.
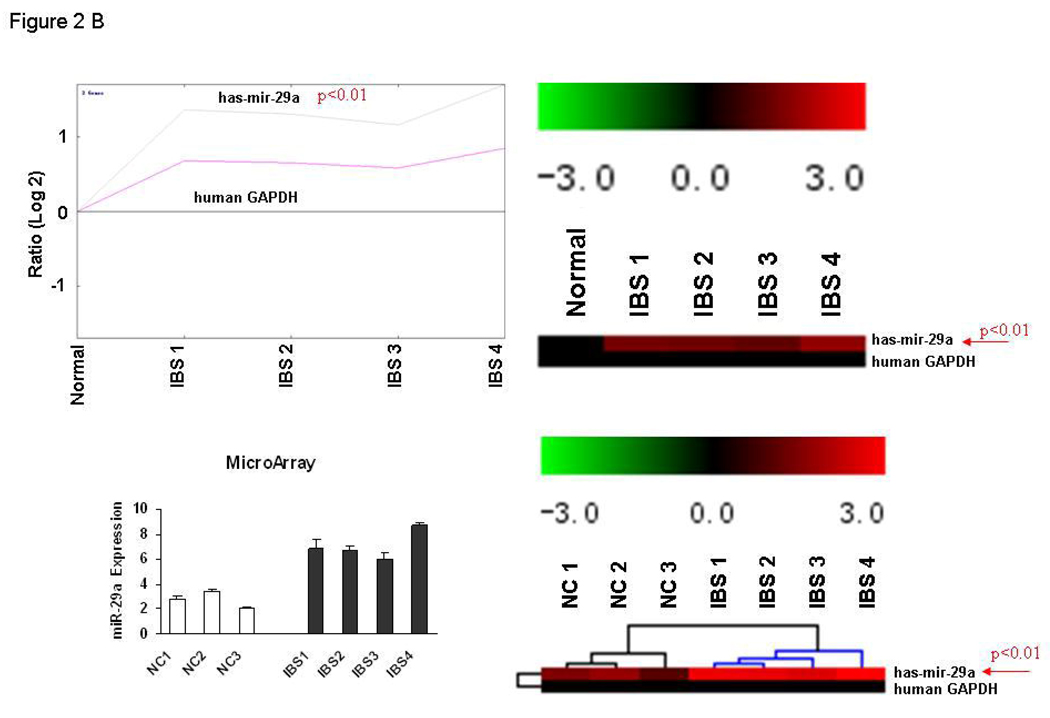
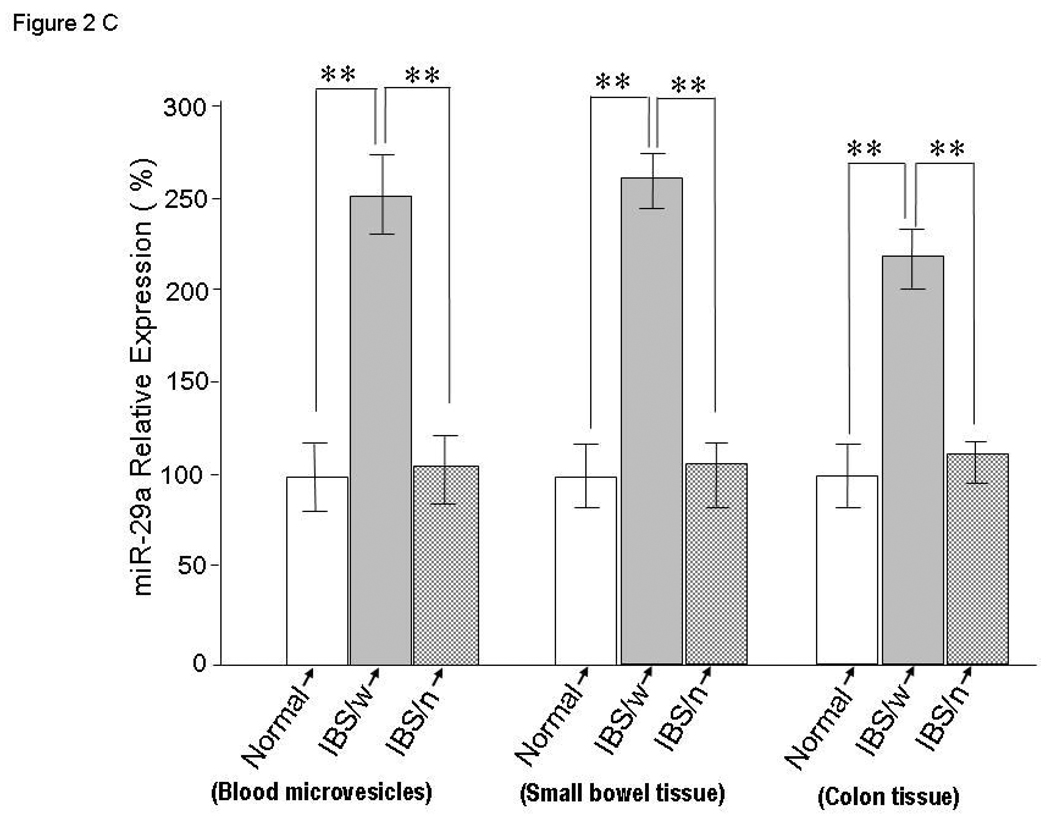
Alterations in miRNA expression in blood microvesicles and colon tissues from IBS Patients. Panel A: Upper panel – Illustrates electron microscope picture of blood-microvesicle; Left lower panel – We randomly selected blood-microvesicles from 4 of the 8 IBS patients with increased membrane permeability and 4 normal controls. Total RNA and miRNA were isolated from blood microvesicles and miRNA profiling was done. The log (base 10) ratio of P values for relative miRNA expression was plotted for each miRNA in descending order of formal name. Representative p-values for spots as calculated by unpaired tests for selected miRNAs from whole blood microvesicles of IBS patients vs. controls. The lower right graph shows the generated heat maps that were row normalized between samples based on the minimum and maximum values for each miRNA and thus provided a non-quantitative visual representation of relative differences between each miRNA. The heat map of the most up-regulated and down-regulated miRNAs is illustrated in the lower right graph. miR-29a was increased in blood microvesicle samples from all four IBS patients when compared to controls (p < 0.01). Panel B: Differential expression of miR-29a in colon tissues from IBS patients. Top two panels represent fold-change in miR-29a expression in IBS patients compared with controls. Bottom two panels illustrate the miR-29a/GAPDH expression ratios in 4 IBS patients and 3 controls. Panel C: miRNA expression analysis for miR-29a was done using TaqMan microRNA qRT-PCR Assay kits. Quantitative data from 8 IBS patients with increased intestinal membrane permeability, 8 IBS patient without increased intestinal membrane permeability, and 8 normal controls representing the mean and standard error are presented in the bar graph. (**p<0.001 compared with expression in controls, there is no differences between normal control and IBS with normal permeability). IBS/w = IBS with increased intestinal permeability; IBS/n = IBS without increased intestinal permeability.
miRNA Microarray of Colon Tissue
We obtained colonic biopsies from 4 of the 8 IBS patients with increased membrane permeability and from 3 normal controls above and used miRNA microarrays to analyze the tissue samples. The colon tissue miRNA expression patterns in IBS patients and normal controls were analyzed using a self-organizing tree algorithm. A dendrogram showing 3 clusters was generated. Cluster 1 defined a group of 291 miRNA isoforms that showed a marked increase in expression in IBS patients. Cluster 2 included 337 miRNA isoforms that were minimally increased or unchanged in expression. Cluster 3 included a group of 305 miRNA isoforms which were unchanged or decreased in IBS patients compared with normal controls. The miRNA data from the colon biopsies in these IBS patients revealed that, as seen for blood microvesicles, several miRNAs were up-regulated, including miR-29a. Thus, miR-29a was up-regulated in both blood microvesicles and colon tissue of IBS patients with increased membrane permeability. Figure 2B illustrates the increased expression of miR-29a in colon tissues from these IBS patients. miR-29a expression was significantly increased by more than 3-fold in all 4 IBS patients with increased intestinal membrane permeability (filled bars) when compared with normal controls (unfilled bars).
Quantitative Real-Time PCR
Interestingly, miR-29a was consistently over-expressed in blood microvesicles, small bowel and colon in IBS patients with increased intestinal membrane permeability compared to IBS patients with normal intestinal permeability and controls (Figure 2C).
Association of miR-29a and in Vitro Membrane Permeability
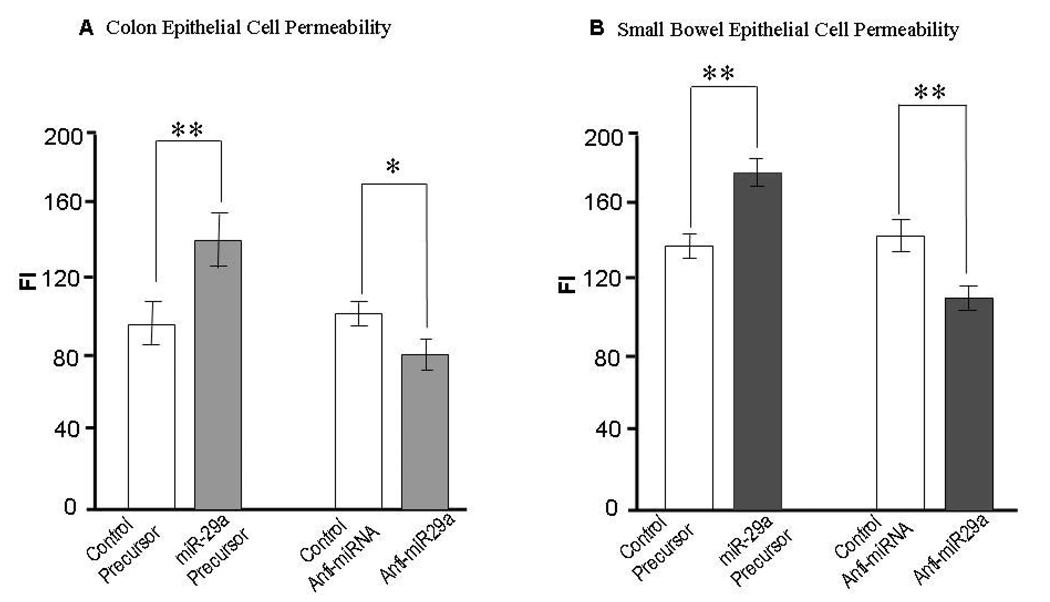
Over-expression of miR-29a significantly enhanced epithelial permeability in both FHC and FHs74Int cells whereas inhibiting miR-29a had the opposite effect (Figure 3). These data indicates that miR-29a alters colon and small bowel epithelial cell permeability in vitro, and suggests a role for miR-29a in the regulation of intestinal membrane permeability in vivo.
Figure 3.
Modulation of colon and small bowel epithelial cell permeability by miR-29a. FHC colonic cells and FHs74Int small intestinal epithelial cells were transfected with either100 nmol/L miR-29a–specific miRNA precursors / inhibitors or control miRNA precursors / inhibitors. Permeability was assessed after 48 hours using In Vitro Permeability Assay Kits. Over-expression of miR-29a significantly enhanced epithelial permeability in FHC (Panel A) and FHs74Int (Panel B) cells whereas silencing miR-29a had the opposite effect (*p < 0.05, ** p< 0.01 when compared with controls).
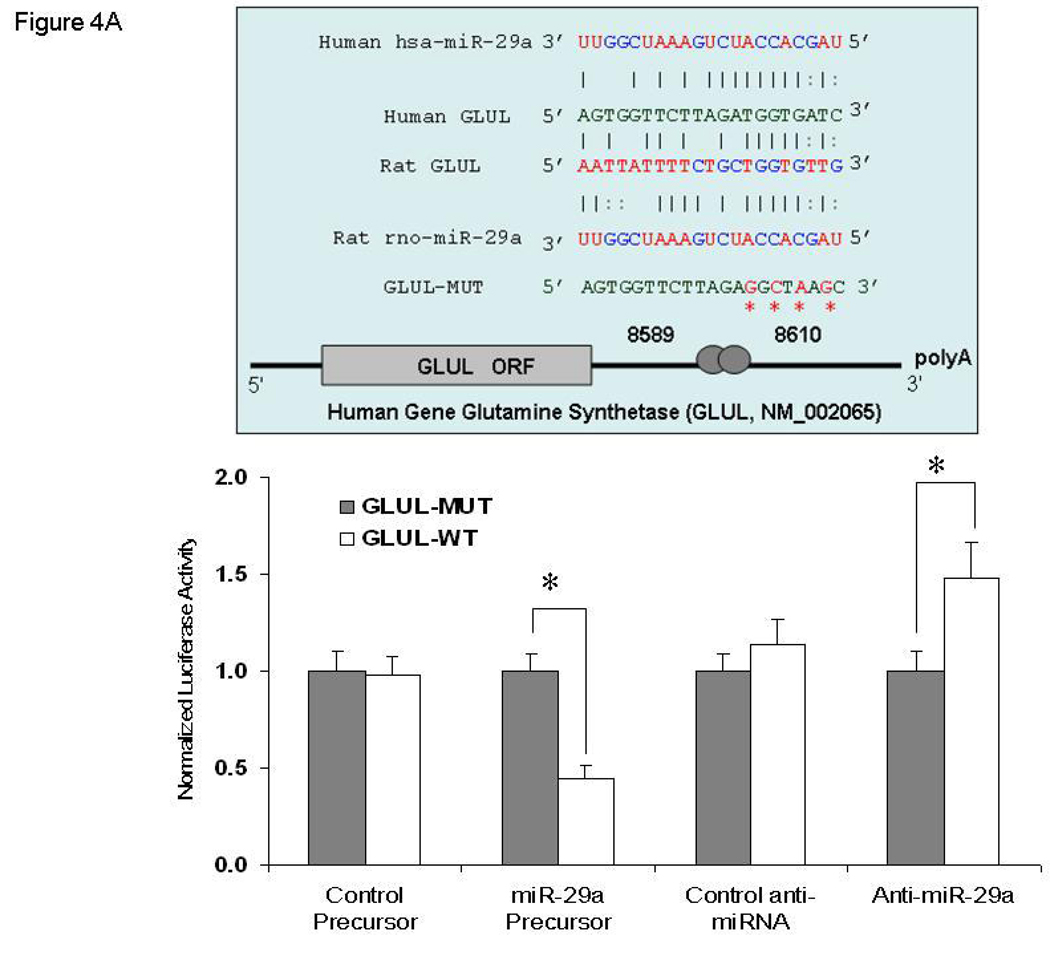
Identification of GLUL as a miR-29a Target
Further investigation of miR-29a revealed that GLUL carries a putative miR-29a binding site, as predicted by the Sanger miRNA data base target search program (miRBase http://microrna.sanger.ac.uk/sequences/). There was perfect complementarity between miR-29a and the 3’UTR of GLUL over the first 9 nucleotides (including the 2−7 seed), which is consistent with it being a bona fide mir-29a binding site, as shown in the upper panel of Figure 4A. To confirm target specificity between miR-29a and GLUL, we did a dual-luciferase reporter assay with the vector containing the putative (mutant) GLUL 3’ UTR target site downstream of a luciferase reporter gene. Relative luciferase reporter activity of FHC cells, co-transfected miR-29a precursor with GLUL-WT (wild type), was significantly lower than FHC cells transfected with GLUL-MUT (mutant) (p = 0.003). In contrast, FHC cells transfected with anti-miR-29a inhibitor and GLUL-WT displayed increased luciferase activity when compared to GLUL-MUT (p = 0.002; Figure 4A, lower panel). Among the eleven gastrointestinal associated miRNAs normalized to RNU6B, there was higher expression of miR-29a in colonic and small intestinal epithelial cells when compared to expression of several other miRNAs (supplementary Figure 4). This data further supports that miR-29a has a physiologic role in gastrointestinal tract.
Figure 4.
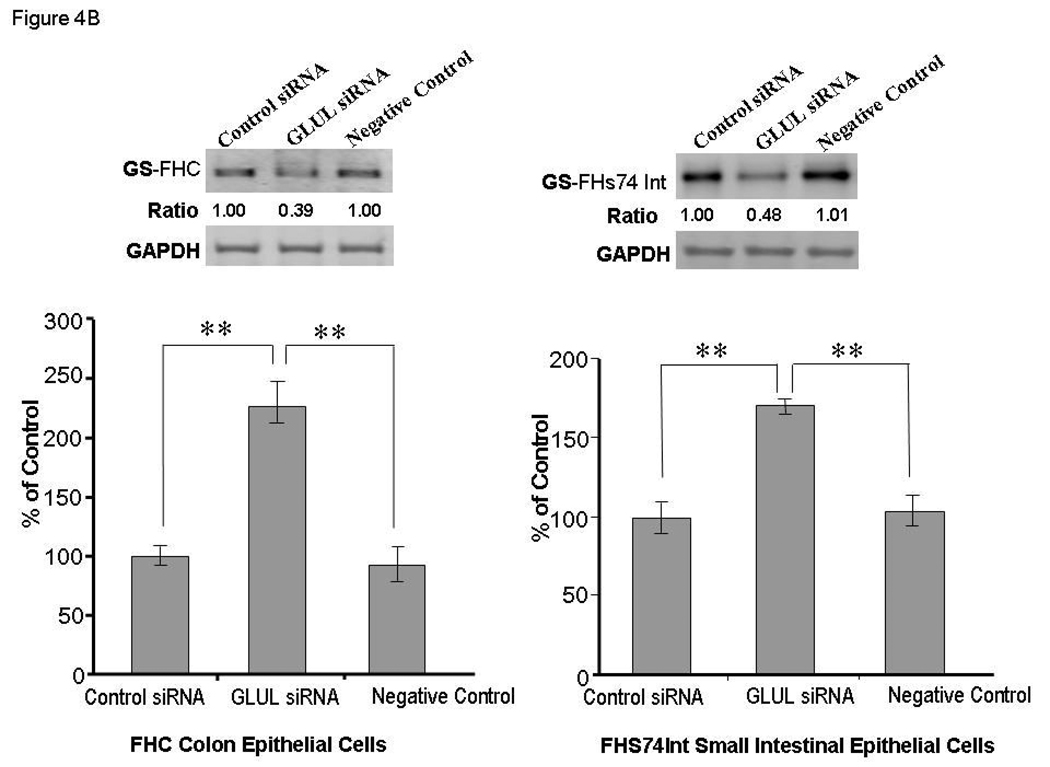
Panel A: Identification of GLUL as a target of miR-29a. Top Graph: The location of the putative miR-29a target site in the GLUL 3'-UTR is shown. The sequence of the mutated target site with mutations to disrupt base pairing between miR-29a binding sites and GLUL is also displayed. The mutation sites are labeled with red symbol “*”. Bottom Graph: FHC human colon epithelial cells were transfected with 1 µg of the GLUL-wt or GLUL-mut firefly luciferase expression construct, along with either miR-29a or control precursors (or inhibitors). Luciferase assays were done after 48 h using a dual luciferase reporter assay system. All the groups are normalized to the average of GLUL MUT group co-transfected either with control precursor or with control anti-miRNA (average value = 1). A significant decrease (or increase) in relative firefly luciferase activity in the presence of wild type miR-29a with miR-29a precursors (or anti-miR-29a inhibitors) indicates the presence of a miR-29a modulated target sequence in the 3'-UTR of GLUL. Bars show mean ± SD from eight separate experiments done in triplicate (*p<0.05). Panel B: Lower panel indicate down-regulation of GLUL increased epithelial cell permeability. All the groups are normalized to the percentage of control siRNA group in both cell lines tested (average value of control siRNA group = 100%). The mean and standard deviation from 4 separate experiments are shown when compared with control siRNA-transfected cells (**p<0.001). Upper panel show immunoblot analysis: Cell lysates were obtained after in-vitro epithelial permeability for immunoblot analysis of glutamine synthetase expression. Down-regulation of GLUL silenced glutamine synthetase expression in FHC and FHs74Int cells. (GS= glutamine synthetase).
These data suggest that GLUL is one of the targets of miR-29a and that miR-29a can down-regulate GLUL by translational repression. Since there is conservation of human miR-29a and GLUL, we next evaluated the role of GLUL in the small bowel and colon of IBS patients as well as in human colon / small bowel epithelial cells.
Silencing GLUL enhances epithelial permeability
Enteral glutamine maintains the intestinal barrier and reduces bacterial translocation. Loss of GLUL could be associated with altered epithelial barrier function. Changes in GLUL expression may reflect aberrant expression of miR-29a. GLUL silencing by RNA interference in FHC and FHs74Int cells results in an increase in epithelial cell permeability, similar to those increases observed with enforced expression of miR-29a by transfection with miR-29a precursor (Figure 4B, lower panel), suggesting the direct regulation of colon and intestinal epithelial barrier function by glutamine synthetase (Supplementary Figure 3).
miR-29a Regulation of Glutamine Synthetase Expression
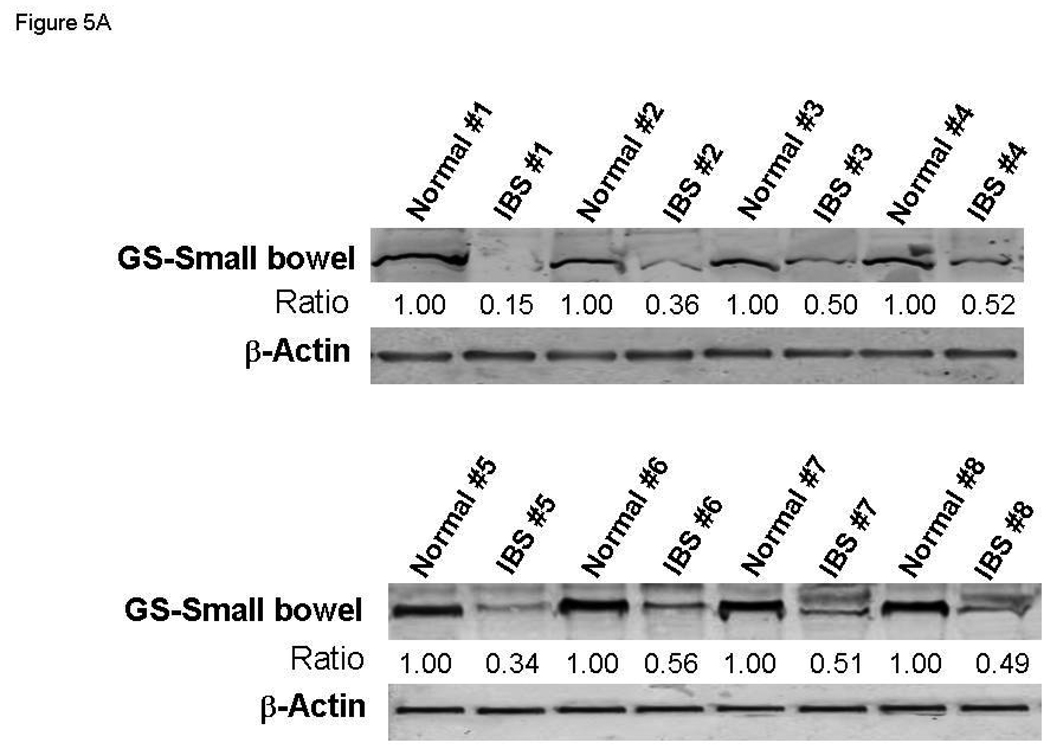
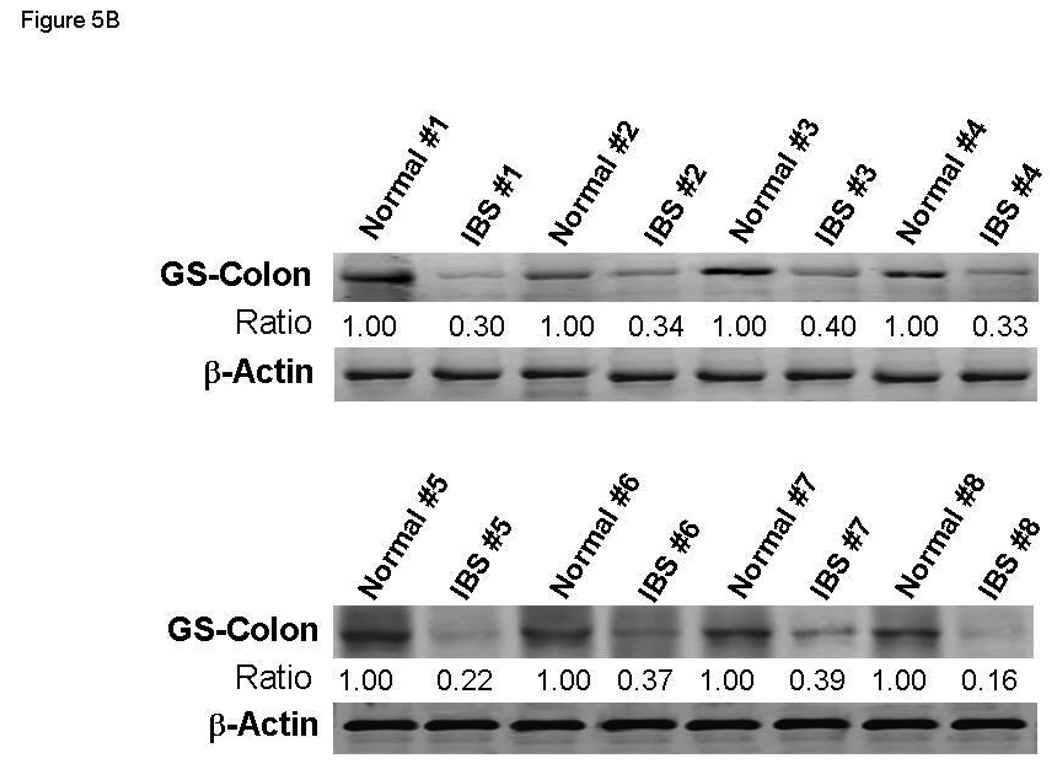
Based on the above observations, we hypothesized that a function of miR-29a is to repress the expression / activation of glutamine synthetase in small bowel / colonic epithelial cells, which may be an important mechanism driving increased intestinal membrane permeability by reducing glutamine synthesis. To test this hypothesis, we examined the expression of glutamine synthetase in small bowel and colon in the subset group of IBS patients with increased miR-29a. Human small bowel (n=8) and colon (n=8) biopsy tissues were obtained from IBS patients with increased intestinal membrane permeability and upregulated miR-29a expression. Small bowel (n=8) and colon biopsy tissues (n=8) were also obtained from normal controls. Glutamine synthetase expression in small bowel was significantly reduced in these IBS patients (Figure 5A). Our data also showed decreased glutamine synthetase expression in the colon and small bowel of these IBS patients (Figure 5B).
Figure 5.
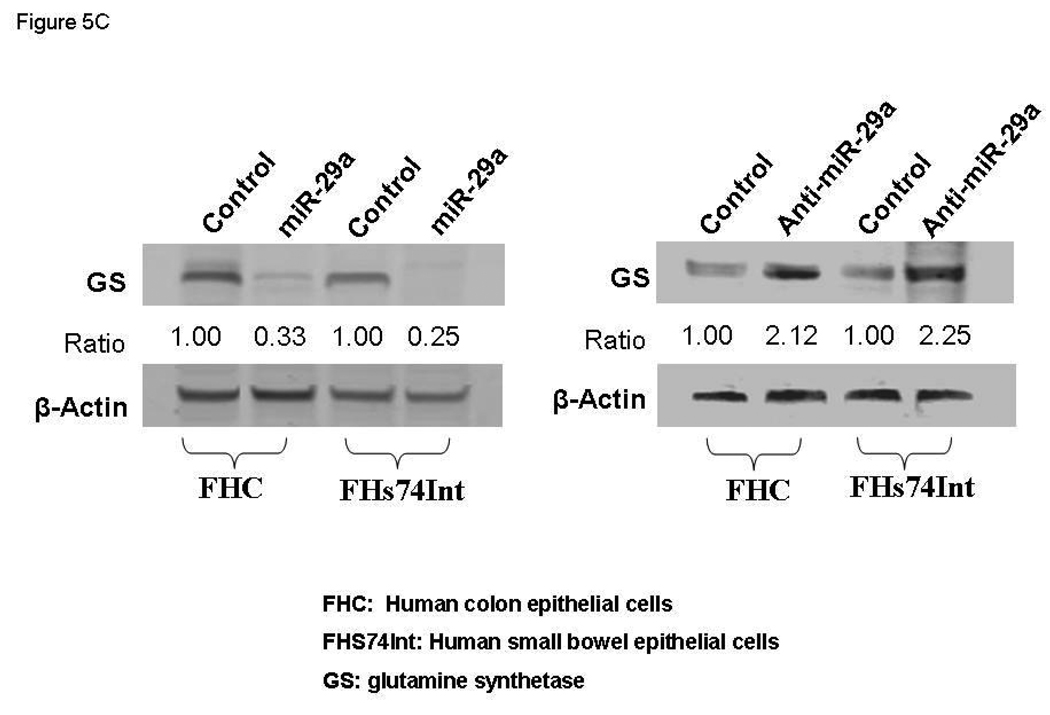
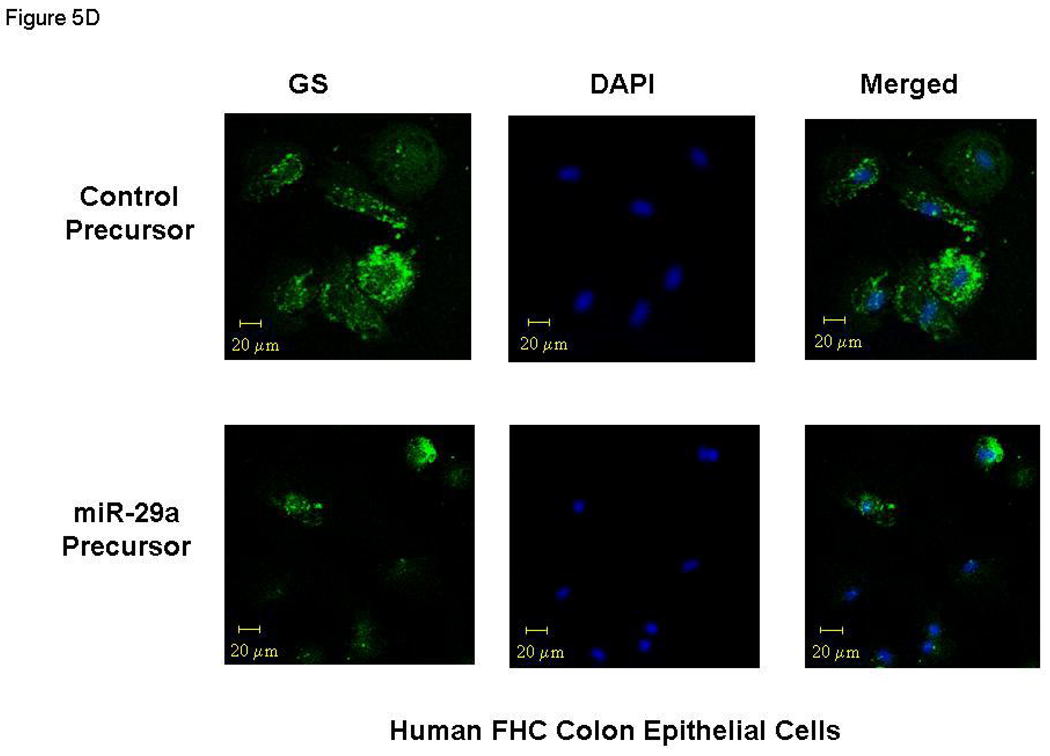
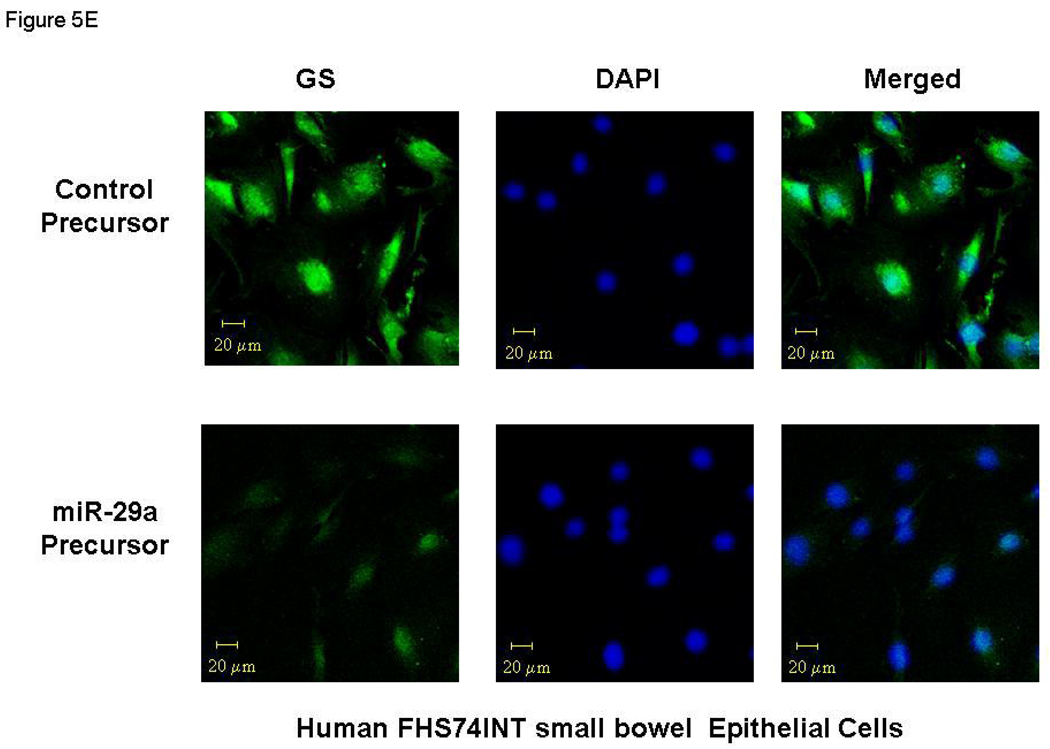
miR-29a modulates expression of glutamine synthetase (GS). Small bowel tissue (Panel A) and colon tissue (Panel B) homogenates were obtained from 8 IBS patients with increased intestinal membrane permeability as well as 8 normal controls. Glutamine synthetase expression was significantly reduced in all 8 IBS patients compared with normal controls. Panel C: FHC human colon and FHs74Int small bowel epithelial cells were transfected with 100 nmol/L miR-29a control or precursors. Cell lysates were obtained after 72 hours for immunoblot analysis of glutamine synthetase expression. Up-regulation of miR-29a significantly silenced glutamine synthetase expression in human colon and intestinal epithelial cells. Panels D & E: Human FHC colon and human FHs74Int small bowel epithelial cells were transfected with 100 nmol/L miR-29a precursor or control construct. Immunocyto-chemistry for glutamine synthetase (GS) was done after 72 hours. A decrease in glutamine synthetase expression was observed in both types of epithelial cells transfected with miR-29a precursor (lower panels) compared with control precursor (upper panels).
We then examined the effects of miR-29a modulation on epithelial cell phenotype. Transfection of miR-29a precursors / inhibitors for 72 hours significantly down-regulated / up-regulated glutamine synthetase protein levels respectively in both FHC and FHs74Int epithelial cells (Figure 5C). Confocal immunofluorescent staining of human FHC colon and human FHS-74 small bowel epithelial cells for GLUL also revealed that expression was reduced after miR-29a over-expression (Figure 5D lower panel and 5E lower panel). Thus, the extent of reduction of glutamine synthetase was directly correlated with miR-29a expression and led to increased membrane intestinal permeability.
DISCUSSION
Our study demonstrates that a subset of patients with diarrhea-predominant IBS have increased intestinal permeability that is associated with increased miR-29a expression in blood microvesicles, small bowel and colon tissue. It also delivered the first evidence that altered gastrointestinal miR-29a expression may mediate changes in intestinal membrane permeability in IBS patients through glutamine-dependent signaling pathways. Furthermore, altered miR-29a in blood microvesicles could serve as a unique molecular marker for IBS patients with increased intestinal membrane permeability.
Two major functions of the gastrointestinal tract are to act as an absorptive organ and to serve as a barrier to bacteria, macromolecules, and toxic compounds.8,40 Disruption of this barrier can lead to local gastrointestinal dysfunction as well as systemic abnormalities such as bacterial translocation and sepsis. Moreover, abnormalities of the immune or mechanical barriers lead to enhanced uptake of inflammatory luminal macromolecules and pathogenic bacteria. Increased permeability of the mucosal barrier in the intestines appears to correlate with a number of clinical disorders including inflammatory bowel disease, food allergies, allergic disorders, rheumatoid arthritis, celiac disease, and several chronic dermatological conditions.41 Several recent studies have shown that increased intestinal permeability may be a potential etiologic factor in diarrhea-predominant IBS patients.4–7, 42
In patients with post-infectious IBS, it has been hypothesized that chronic inflammation with increases in intestinal permeability, mast cells, proinflammatory cytokines, serotonin-containing enterochromatin cells, and T lymphocytes play a role.4, 43, 44, 45 The activation of these inflammatory factors may lead to increased intestinal permeability, especially in a subset of patients prone to or with diarrhea-predominant IBS.4, 36 Increased intestinal permeability allows the passage of bacteria and antigens through the mucosal layer of the gut. This could lead to activation of mucosal immune responses and, subsequently, the chronic diarrhea and abdominal pain seen in IBS patients.43
The human gastrointestinal tract is the major site of glutamine utilization in the body. Glutamine is a major energy source for rapidly dividing intestinal mucosal cells of the digestive tract. Its depletion results in epithelial atrophy and a subsequent increase in permeability of the intestinal barrier. Glutamine supplementation has been shown to decrease bacterial translocation and intestinal permeability after intestinal injury.15–17 Glutamine supplementation has also been shown to decrease intestinal permeability and improve gastrointestinal function in Crohn’s disease and in patients with advanced esophageal cancer undergoing radiochemotherapy.41,42 It also provides clinical benefits to critically ill patients who develop increased intestinal permeability that leads to septicemia of enteric origin.15–17
Glutamine synthetase is an important enzyme that helps maintain adequate levels of intestinal glutamine. Glutamine synthetase has been shown to be important for cell proliferation in rat intestinal crypt cells.48 Mucosal epithelial cells lining the gastrointestinal tract have a relatively small free glutamine pool and rely on glutamine synthetase. Inhibition of glutamine synthetase decreased proliferation of cultured rat intestinal cells.48 Thus, conditions in which decreased levels of intestinal glutamine synthetase are present may lead to low levels of available glutamine and a shortage of the energy supply for metabolism in epithelial cells, resulting in increased permeability of the intestinal barrier. Interestingly, our study evaluated IBS patients with increased membrane permeability who also had reduced levels of intestinal glutamine synthetase activity associated with increased miR-29a expression. Therefore, it is possible that miR-29a plays a key role in directly modulating intestinal permeability through glutamine dependent signaling pathways which directly down regulate glutamine synthetase expression and lead to increased intestinal permeability.
Our study has confirmed that glutamine synthetase is a target of miR-29a. These data indicate that decreased levels of glutamine synthetase may lead to insufficient intestinal glutamine levels and result in increased intestinal permeability. Our data also suggest that miR-29a modulates colon and small bowel epithelial cell permeability as confirmed in our in vitro cell culture study. Meanwhile, miR-29a is also predicted to target Claudin-1, an important mucosal tight junction protein that is significantly reduced in D-IBS patients. Claudin-1 has been shown to be a critical component in the regulation of paracellular permeability to ions and pathogens. Since regulation by miRNAs is obviously not as simple as one miRNA–one target gene; miR-29a could be a valid translational molecule that may be involved in the regulation of intestinal permeability in some IBS patients through multiple target genes.
Interestingly, miR-29a expression was also systemically up-regulated as indicated by its increased expression in microvesicles in the blood of these IBS patients. Microvesicles are released by many cell types and differ in composition depending on cell origin and status.49–51 Release of intact vesicles is increasingly recognized as a major avenue for the delivery of extracellular signals or bioactive factors.18–25 Furthermore, changes in circulating microvesicles may reflect alteration of localized physiological processes such as those seen in the gastrointestinal tract of IBS patients with increased intestinal permeability. Therefore, microvesicles have great potential to serve as diagnostic markers of disease processes. Microvesicles may also allow the development of new diagnostic strategies based on the analysis of molecular signature profiling, such as miRNA arrays.
MicroRNAs influence a number of chronic disease processes and may also be involved in regulating disease activity in gastrointestinal disorders. Recently, one study evaluated patients with both active and inactive ulcerative colitis.52 They found differential expression of 11 miRNAs present in patients with active ulcerative colitis. Aside from these findings, they also evaluated a few patients with Crohn’s disease, microscopic colitis, infectious colitis, and irritable bowel syndrome. miR-29a was increased (but not significantly) in the IBS patients that they evaluated. Several factors may account for their findings which differ from ours including a small sample size and the inclusion of fewer IBS patients that were not clinically phenotyped. We have carefully phenotyped a specific subset of IBS patients that were diarrhea-predominant and who had increased intestinal permeability. In our study, multiple random biopsies were taken from the duodenum and throughout the colon in 19 IBS patients instead of using tissue from the sigmoid colon only. Another recent study has been carried out evaluating miR-510 in diarrhea-predominant IBS patients.53 They chose miR-510 directly from miRBase without microRNA profiling since it was predicted to target the serotonin receptor gene, HTR3E. Thus, the functional role of microRNAs in IBS has not been extensively evaluated by other investigators. In our study, we did microRNA profiling of all available miRNAs in order to identify all possible miRNAs that might have potential functional regulation properties in IBS patients.
In summary, our current study, which includes microvesicle-miRNA profiling data from IBS patients, revealed up-regulation of miR-29a. This supports a potential role for miR-29a in regulating intestinal membrane permeability in diarrhea-predominant IBS patients. Thus, profiling of systemic microvesicles may be useful as a molecular marker for IBS patients with increased intestinal permeability and could lead to new therapeutic interventions. Our study is unique in that it suggests that some patients with diarrhea-predominant IBS have increased intestinal permeability associated with increased miR-29a expression in blood microvesicles and in small intestinal and colonic tissue. Thus, altered gastrointestinal miR-29a expression may mediate intestinal membrane permeability in IBS patients through glutamine dependent mechanisms as our study confirms the functional interaction between miR29a and the GLUL gene. Since our preliminary study addresses the molecular basis of gut permeability and the role of miRNAs in IBS, additional larger studies are needed in order to validate our hypothesis that miRNA-29a expression in blood microvesicles may serve as a biomarker with which to identify IBS patients with increased intestinal permeability. Future studies are now needed to evaluate miR-29a as a potential therapeutic target in the treatment of IBS. Other functional targets of miR-29a will also need to be further in IBS patients.
What is already known about this subject
The molecular mechanisms underlying the pathophysiology of irritable bowel syndrome (IBS) are poorly understood
Patients with diarrhea-predominant IBS have increased intestinal membrane permeability
Deficiencies in glutamine may lead to increased intestinal membrane permeability
What are the new findings
Blood microvesicles from IBS patients may be involved in gastrointestinal signaling pathways via a horizontal transfer of miRNA
Increased expression of miR-29a leads to decreased glutamine synthetase levels and resulting increased intestinal membrane permeability in IBS patients through glutamine dependent mechanisms
miRNA profiling of systemic microvesicles may be useful as a molecular marker for IBS patients with increased intestinal permeability
How might it impact clinical practice in the foreseeable future
Characterization of the molecular basis of IBS will provide new molecular prognostic markers and targets for therapy
Glutamine may serve as potential therapeutic agent to treat patients with IBS and increased intestinal membrane permeability
The signaling pathway of miRNA modulated targets could lead to a new therapeutic approach for IBS patients
Supplementary Material
Acknowledgements
The authors thank Drs. Hunter & Marsh for portions of the microvesicle preparation protocol for support.
Funding:
This study was supported by an NIH grant (NIH RO1-NS053090) to GN Verne and by a VA Merit Review Award to GN Verne from the Medical Research Service of the Department of Veterans Affairs.
Footnotes
Advance online articles have been peer reviewed and accepted for publication but have not yet appeared in the paper journal (edited, typeset versions may be posted when available prior to final publication). Advance online articles are citable and establish publication priority; they are indexed by PubMed from initial publication. Citations to Advance online articles must include the digital object identifier (DOIs) and date of initial publication.
To order reprints of this article go to: http://gut.bmj.com/cgi/reprintform
Competing Interests:
None of the authors have any competing interests to declare.
Copyright Licence Statement:
The corresponding author has the right on behalf of all authors and does grant on behalf of all authors, an exclusive licence (or non exclusive for government employees) on a worldwide basis to the BMJ Publishing Group LTd and its Licensees to permit this article (if accepted) to be published in Gut editions and any other BMJPGL products to exploit all subsidiary rights, as set out in our license (http://gut.bmjjournals.com/ifora/licence.dtl).
REFERENCES
- 1.Verne GN, Cerda JJ. Irritable bowel syndrome. Streamlining the diagnosis. Postgrad Med. 1997;102:197–208. doi: 10.3810/pgm.1997.09.322. [DOI] [PubMed] [Google Scholar]
- 2.Verne GN, Himes NC, Robinson ME, et al. Central representation of visceral and cutaneous hypersensitivity in the irritable bowel syndrome. Pain. 2003;103(1–2):99–110. doi: 10.1016/s0304-3959(02)00416-5. [DOI] [PubMed] [Google Scholar]
- 3.Verne GN, Robinson ME, Price DD. Hypersensitivity to visceral and cutaneous pain in the irritable bowel syndrome. Pain. 2001;93(1):7–14. doi: 10.1016/S0304-3959(01)00285-8. [DOI] [PubMed] [Google Scholar]
- 4.Dunlop SP, Hebden J, Campbell E, et al. Abnormal intestinal permeability in subgroups of diarrhea-predominant irritable bowel syndromes. Am J Gastroenterol. 2006;101:1288–1294. doi: 10.1111/j.1572-0241.2006.00672.x. [DOI] [PubMed] [Google Scholar]
- 5.Spiller RC, Jenkins D, Thornley JP, et al. Increased rectal mucosal enteroendocrine cells, T lymphocytes, and increased gut permeability following acute Campylobacter enteritis and in post-dysenteric irritable bowel syndrome. Gut. 2000;47:804–811. doi: 10.1136/gut.47.6.804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Spiller RC. Role of infection in irritable bowel syndrome. J Gastroenterol. 2007;42 Suppl XVII:41–47. doi: 10.1007/s00535-006-1925-8. [DOI] [PubMed] [Google Scholar]
- 7.Zhou Q, Zhang B, Verne GN. Intestinal membrane permeability and hyper-sensitivity in the irritable bowel syndrome. Pain. 2009;146:41–46. doi: 10.1016/j.pain.2009.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bjarnason I, MacPherson A, Hollander D. Intestinal permeability: an overview. Gastroenterology. 1995;108:1566–1581. doi: 10.1016/0016-5085(95)90708-4. [DOI] [PubMed] [Google Scholar]
- 9.Keshavarzian A, Holmes EW, Patel M, Iber F, Fields JZ, Pethkar S. Leaky gut in alcoholic cirrhosis: A possible mechanism for alcohol-induced liver damage. Am J Gastroenterol. 1999;94(1):200–207. doi: 10.1111/j.1572-0241.1999.00797.x. [DOI] [PubMed] [Google Scholar]
- 10.Keshavarzian A, Fields JZ, Vaeth J, Holmes EW. The differing effects of acute and chronic alcohol on gastric and intestinal permeability. Am J Gastroenterol. 1994;89(12):2205–2211. [PubMed] [Google Scholar]
- 11.Al Chaer ED, Kawasaki M, Pasricha PJ. A new model of chronic visceral hypersensitivity in adult rats induced by colon irritation during postnatal development. Gastroenterology. 2000;119(5):1276–1285. doi: 10.1053/gast.2000.19576. [DOI] [PubMed] [Google Scholar]
- 12.Mayer EA, Collins SM. Evolving pathophysiologic models of functional gastrointestinal disorders. Gastroenterology. 2002;122(7):2032–2048. doi: 10.1053/gast.2002.33584. [DOI] [PubMed] [Google Scholar]
- 13.Zhou Q, Caudle RM, Price DD, Verne GN. Visceral and somatic hypersensitivity in a subset of rats following TNBS-Induced colitis. Pain. 2008;134(1–2):9–15. doi: 10.1016/j.pain.2007.03.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Häberle J, Görg B, Rutsch F, et al. Congenital glutamine deficiency with glutamine synthetase mutations. N Engl J Med. 2005;353:1926–1933. doi: 10.1056/NEJMoa050456. [DOI] [PubMed] [Google Scholar]
- 15.Souba WW, Klimberg VA, Plumley DA, Salloum RM, Flynn TC, Bland KI, Copeland EM., 3rd The role of glutamine in maintaning a healthy gut and supporting the metabolic response to injury and infection. J Surg Res. 1990;48(4):383–391. doi: 10.1016/0022-4804(90)90080-l. [DOI] [PubMed] [Google Scholar]
- 16.Klimberg VA, Souba WW. The importance of intestinal glutamine metabolism in maintaining a healthy gastrointestinal tract and supporting the body’s response to injury and illness. Surg Annu. 1990;22:61–76. [PubMed] [Google Scholar]
- 17.Labow BI, Souba WW. Glutamine. World J Surg. 2000;24(12):1503–1513. doi: 10.1007/s002680010269. [DOI] [PubMed] [Google Scholar]
- 18.Valadi H, Ekstrom K, Bossios A, Sjostrand M, Lee JJ, Lotvall JO. Exosome-mediated transfer of mRNA and microRNAs is a novel mechanism of genetic exchange between cells. Nature Cell Biology. 2007;9(6):654–659. doi: 10.1038/ncb1596. [DOI] [PubMed] [Google Scholar]
- 19.Fevrier B, Raposo G. Exosomes: endosomal-derived vesicles shipping extracellular messages. Curr Opin Cell Biol. 2004;16:415–421. doi: 10.1016/j.ceb.2004.06.003. [DOI] [PubMed] [Google Scholar]
- 20.Greenwalt TJ. The how and why of exocytic vesicles. Transfusion. 2006;46:143–152. doi: 10.1111/j.1537-2995.2006.00692.x. [DOI] [PubMed] [Google Scholar]
- 21.Hugel B, Martinez MC, Kunzelmann C, et al. Membrane microparticles: two sides of the coin. Physiology (Bethesda) 2005;20:22–27. doi: 10.1152/physiol.00029.2004. [DOI] [PubMed] [Google Scholar]
- 22.Pap E, Pallinger E, Paszoi M, Falus A. Highlights of a new type of intercellular communication microvesicle-based information transfer. Inflamm Res. 2009;58(1):1–8. doi: 10.1007/s00011-008-8210-7. Review. [DOI] [PubMed] [Google Scholar]
- 23.Geijnen HF, Schiel AE, Fijnheer R, gauze HJ, Sixma JJ. Activated platelets release two types of membrane vesicles: micrivesicles by surface shedding and exosomes derived from exocytosis of multivesicular bodies and alpha-granules. Blood. 1999;94(11):3791–3799. [PubMed] [Google Scholar]
- 24.VanWijk MJ, VanBavel E, Sturk A, et al. Microparticles in cardiovascular diseases. Cardiovas Res. 2003;59:277–287. doi: 10.1016/s0008-6363(03)00367-5. [DOI] [PubMed] [Google Scholar]
- 25.Horstmann LL, Jy W, Jimenez JJ, et al. New horizons in the analysis of circulating cell-derived microparticles. Keio J Med. 2004;53:210–230. doi: 10.2302/kjm.53.210. [DOI] [PubMed] [Google Scholar]
- 26.Ratajczak J, Wysoczynski M, Hayek F, et al. Membrane-derived microvesicles: important and underappreciated mediators of cell-to-cell communication. Leukemia. 2006;20:1487–1495. doi: 10.1038/sj.leu.2404296. [DOI] [PubMed] [Google Scholar]
- 27.Sloand EM, Mainwaring L, Keyvanfar L, et al. Transfer of glycosylphosphatidylinositol-anchored proteins to deficient cells after erythrocyte transfusion in paroxysmal nocturnal hemoglobinuria. Blood. 2004;104:3782–3788. doi: 10.1182/blood-2004-02-0645. [DOI] [PubMed] [Google Scholar]
- 28.Kim J, Krichevsky A, Grad Y, et al. Identification of many microRNAs that copurify with polyribomes in mammalian neurons. Proc Natl Acad Sci USA. 2004;101:360–365. doi: 10.1073/pnas.2333854100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Farh KK, Grimson A, Jan C, et al. The widespread impact of mammalian microRNA on mRNA repression and evolution. Science. 2005;310:1817–1821. doi: 10.1126/science.1121158. [DOI] [PubMed] [Google Scholar]
- 30.Fabbri M, Garzon R, Cimmino A, et al. MicroRNA-29 family reverts aberrant methylation in lung cancer by targeting DNA methyltranferases 3A and 3B. Proc Natl Acad Sci USA. 2007;104:15805–15810. doi: 10.1073/pnas.0707628104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Vanner S. The lactulose breath test for diagnosing SIBO in IBS patients: Another nail in the coffin. Am J Gastroenterol. 2008;103:964–965. doi: 10.1111/j.1572-0241.2008.01798.x. [DOI] [PubMed] [Google Scholar]
- 32.Bratten JR, Spainer J, Jones MP. Lactulose breath testing does not discriminate patients with irritable bowel syndrome from healthy controls. Am J Gastroenterol. 2008;103:958–963. doi: 10.1111/j.1572-0241.2008.01785.x. [DOI] [PubMed] [Google Scholar]
- 33.Longstreth GF, Thompson WG, Chey WD, et al. Functional bowel disorders. Gastroenterology. 2006;130:1480–1491. doi: 10.1053/j.gastro.2005.11.061. [DOI] [PubMed] [Google Scholar]
- 34.Camilleri M, Nadeau A, Lamsam J, Nord SL, Ryks M, Burton D, Sweetser S, Zinsmeister AR, Singh R. Understanding measurements of intestinal permeability in healthy humans with urine lactulose and mannitol exretion. Neurogastroenterol Motil. 2009 doi: 10.1111/j.1365-2982.2009.01361.x. (In Press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Leonards KS, Ohki S. Isolation and characterization of large (0.5–1.0 µm) cytoskeleton-free vesicles from human and rabbit erythrocytes. Biochim Biophys Acta. 1983;728:383–393. doi: 10.1016/0005-2736(83)90510-2. [DOI] [PubMed] [Google Scholar]
- 36.Victoria EJ, Branks MJ, Masouredis SP. Immunoreactivity of the Rho(D) antigen in cytoskeleton-free vesicles. Transfusion. 1987:2732–2735. doi: 10.1046/j.1537-2995.1987.27187121469.x. [DOI] [PubMed] [Google Scholar]
- 37.Liu C, Calin GA, Volinia S, Croce CM. MicroRNA expression profiling using microarrays. Nature Protocols. 2008;3(4):563–578. doi: 10.1038/nprot.2008.14. [DOI] [PubMed] [Google Scholar]
- 38.Siddiqui KM, Chopra DP. Primary and long term epithelial cell cultures from human fetal normal colonic mucosa. In Vitro. 1984;20:859–868. doi: 10.1007/BF02619632. Siddiqui and Chopra, 1984. [DOI] [PubMed] [Google Scholar]
- 39.Smith HS. In vitro properties of epithelial cell lines established from human carcinomas and nonmalignant tissue. J Natl Cancer Inst. 1979 Feb;62(2):225–230. [PubMed] [Google Scholar]
- 40.Macdonald TT, Montelenone G. Immunity, inflammation, and allergy in the gut. Science. 2005;307:1920–1925. doi: 10.1126/science.1106442. [DOI] [PubMed] [Google Scholar]
- 41.Porras M, Martín MT, Yang PC, et al. Correlation between cyclical epithelial barrier dysfunction and bacterial translocation in the relapses of intestinal inflammation. Inflamm Bowel Dis. 2006;12:843–852. doi: 10.1097/01.mib.0000231571.88806.62. [DOI] [PubMed] [Google Scholar]
- 42.Camilleri M, Gorman H. Intestinal permeability and irritable bowel syndrome. Neurogastroenterol Motil. 2007;19:545–552. doi: 10.1111/j.1365-2982.2007.00925.x. [DOI] [PubMed] [Google Scholar]
- 43.Spiller RC. Postinfectious irritable bowel syndrome. Gastroenterology. 2003;124:1662–1671. doi: 10.1016/s0016-5085(03)00324-x. [DOI] [PubMed] [Google Scholar]
- 44.Dupont AW. Post-infectious irritable bowel syndrome. Curr Gastroenterol Rep. 2007;9:378–384. doi: 10.1007/s11894-007-0046-8. [DOI] [PubMed] [Google Scholar]
- 45.Wang LH, Fang XC, Pan GZ. Bacillary dysentery as a causative factor of irritable bowel syndrome and its pathogenesis. Gut. 2004;53:1096–2011. doi: 10.1136/gut.2003.021154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Yoshida S, Matsui M, Shirouzu Y, et al. Effects of glutamine supplements and radiochemotherapy on the systemic immune and gut barrier function in patients with advanced esophageal cancer. Ann Surg. 1998;227:485–491. doi: 10.1097/00000658-199804000-00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Sido B, Seel C, Hochlehnert A, et al. Low intestinal glutamine level and low glutaminase activity in Crohn’s Disease: a rational for glutamine supplementation? Dig Dis Sci. 2006;51:2170–2179. doi: 10.1007/s10620-006-9473-x. [DOI] [PubMed] [Google Scholar]
- 48.DeMarco V, Dyess K, Strauss D, et al. Inhibition of glutamine synthetase decreases proliferation of cultured rat intestinal epithelial cells. J Nutr. 1999;129:57–62. doi: 10.1093/jn/129.1.57. [DOI] [PubMed] [Google Scholar]
- 49.Halicka HD, Bedner E, Darzynkiewicz Z. Segregation of RNA and separate packaging of DNA and RNA in apoptotic bodies during apoptosis. Exp Cell Res. 2000;260:248–256. doi: 10.1006/excr.2000.5027. [DOI] [PubMed] [Google Scholar]
- 50.Levine SJ. Mechanisms of soluble cytokine receptor generation. J Immunol. 2004;173:5343–5348. doi: 10.4049/jimmunol.173.9.5343. [DOI] [PubMed] [Google Scholar]
- 51.Taback B, Hoon DS. Circulating nucleic acids and proteomics of plasma/serum: clinical utility. Ann NY Acad Sci. 2004;1022:1–8. doi: 10.1196/annals.1318.002. [DOI] [PubMed] [Google Scholar]
- 52.Wu F, Zikusoka M, Trindade A, Dassopoulus T, Harris ML, Bayless TM, Brant SR, Chakravarti S, Kwon JH. MicroRNAs are differentially expressed in ulcerative colitis and alter expression of macrophage inflammatory peptide-2α. Gastroenterology. 2008;135:1624–1635. doi: 10.1053/j.gastro.2008.07.068. [DOI] [PubMed] [Google Scholar]
- 53.Kapeller J, Houghton LA, Monnikes H, Walstab J, Moller D, Bonisch H, Burwinkle B, Autschbach F, Funke B, Lasitschka F, Buchner KJ, Schmidtmann M, van der Voort I, Wisser AS, Berg T, Rappold G, Niesler B. First evidence for an association of a functional variant in the microRNA-510 target site of the serotonin receptor-type 3E gene with dirrhea predominant irritable bowel syndrome. Human Molecular Genetics. 2008;17(19):2967–2977. doi: 10.1093/hmg/ddn195. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.