Table 5.
Physicochemical properties of the benzoate- or the salicylate-based urea compounds
| Compound | 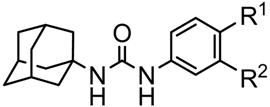 |
Solubility in watera (mg/mL) | Mp (°C) | Experimental parameters and logP | ||||
|---|---|---|---|---|---|---|---|---|
| R1 (para) | R2 (meta) | Concentrationb (μM) | logPc | clogPd | ||||
| Octanol | Buffer | |||||||
| 13 | COOCH3 | H | 0.4 | 219–220 | 2445 | <0.3 | >3.9 | 4.3 |
| 17 | COOH | H | 10 | >300 | 2720 | 17 | 2.2 | 3.8/−0.5e |
| 27 | COOH | OH | 199 | 205–206 | 1641 | 39 | 1.6 | 3.9/−1.1e |
| 28 | COOCH3 | OH | 0.2 | 212–213 | 3886 | <0.3 | >4.1 | 4.1 |
Solubility in 0.1 M sodium phosphate buffer (pH 7.4) at 23 ± 1.5 °C.
Buffer saturated 1-octanol and 0.1 M sodium phosphate buffer (pH 7.4) were used. See Supplementary information in detail.
logP value was calculated following the equation; logP = log10 ([conc. of compound]octanol/[conc. of compound]buffer).
clogPwas calculated using ChemDraw® software (CambridgeSoft, Cambridge, MA).
The clogP of both the acid and base forms of the carboxylic acid are given.
