Abstract
There is no consensus regarding the definition of cardiac syndrome X (CSX). We systematically reviewed recent literature using a standardized search strategy. We included 57 articles. A total of 47 studies mentioned a male/female distribution. A meta-analysis yielded a pooled proportion of females of 0.56 (n = 1,934 patients, with 95% confidence interval: 0.54–0.59). As much as 9 inclusion criteria and 43 exclusion criteria were found in the 57 articles. Applying these criteria to a population with normal coronary angiograms and treated in 1 year at a general hospital, the attributable CSX incidence varied between 3 and 11%. The many inclusion and exclusion criteria result in a wide range of definitions of CSX and these have large effects on the incidence. This shows the need for a generally accepted definition of CSX.
Keywords: Cardiac syndrome X, Microvascular angina, Angina pectoris and normal coronary arteries
Introduction
The syndrome of angina pectoris with a normal coronary arteriogram, often termed cardiac syndrome X (CSX), is an important clinical entity [65]. About 20% of patients with anginal chest pain have normal coronary angiograms (CAG). Some physicians regard this as sufficient to diagnose CSX, the so-called broad diagnosis of CSX [65, 66]. However, the cause of CSX is not conclusively established and its definition is controversial. For example, a subgroup of these patients have objective signs of ischaemia, such as the classic downsloping ST-segment depression on exercise testing and/or a reversible defect detected by myocardial single-photon emission computed tomography (SPECT). Consequently, other authorities consider that CSX is discounted by a positive result from any diagnostic test for ischaemia, especially SPECT; and there is also the view that CSX might best be relabelled as one of several causes of microvascular dysfunction [37]. Furthermore, there are various exclusion criteria, such as hypertension and diabetes, that may or may not be used to discount the diagnosis of CSX. Finally, in overviews and textbooks it is often stated that (pre-menopausal) women are especially prone to developing cardiac syndrome X. In fact, some authors exclusively study female patient populations, but there is considerable variation in the proportion of women in other studies of CSX patients.
The present paper is a review to determine the gender and main definitions for CSX in the recent literature and also the effects of different definitions upon the attributable CSX incidence. To study this latter aspect we examined and analysed the CAG data for a population of patients treated in a general hospital in the Netherlands.
Methods
CSX definitions
The PubMed database was used to identify papers in which criteria and definitions for CSX are described. Our search consisted of ‘(cardiac syndrome x[tiab]) NOT (case reports[pt] OR comment[pt] OR review[pt])’, with limits set to English language, humans, and publication date between June 2003 and July 2008. We limited our search strategy to cardiac syndrome X and excluded metabolic syndrome X and tako-tsubo cardiomyopathy [6, 24, 42].
This search yielded 112 references. Conference abstracts, pediatric papers, and ‘epublications ahead of print’ were excluded. Of 83 articles the full text was retrieved for further analysis. Articles were independently surveyed by two authors (IV and GT) and reviewed with regard to their used definitions of CSX. The survey left 57 studies for this review.
Impact of CSX definition upon CSX incidence
All the CAGs made in 2003 in a general hospital (Spaarne Hospital Hoofddorp, The Netherlands) were collected. An extensive search was made to obtain the clinical history, physical examination, routine laboratory tests, and echocardiography results for all patients with CAGs stated to be completely normal. All CAGs were independently evaluated by two experienced observers (AK and DO).
Only patients with original angiograms and sufficient clinical data were included in the present study.
Results
CSX definitions
The 57 papers selected for determining the main definitions of CSX covered a total of 2,375 patients. A normal coronary angiogram was used in the majority of the studies. However, in a few studies minimal coronary artery disease, like atheromatous plaque without critical obstructions, is included as “normal coronary angiogram” [18, 22, 56]. A few studies state minimal coronary artery disease in the exclusion criteria [1]. But in the majority of the studies there was no statement regarding minimal artery disease. Only few studies specified the interpretation of the normal coronary angiograms, like revision of the CAG films, blinded observation, or interpretation by more reviewers.
Table 1 lists the nine CSX definitions, and hence inclusion criteria, obtained from our review of the 57 studies.
Table 1.
Inclusion criteria for CSX
| Inclusion criteria for CSX | Number of studies |
|---|---|
| Angina pectoris | 18 |
| Positive exercise stress test | |
| Normal coronary arteries | |
| Effort induced angina pectoris | 17 |
| Positive exercise stress test | |
| Normal coronary arteries | |
| Angina pectoris | 11 |
| Positive exercise stress test OR positive SPECT | |
| Normal coronary arteries | |
| Angina pectoris | 4 |
| Normal coronary arteries | |
| Angina pectoris | 4 |
| Positive exercise stress test AND positive SPECT | |
| Normal coronary arteries | |
| Angina pectoris | 1 |
| Positive exercise stress test | |
| No significant CAD on CAG | |
| Effort induced AP | 1 |
| Positive exercise stress test | |
| Normal coronary arteries AND prolonged coronary flow on CAG | |
| Angina pectoris | 1 |
| Positive exercise stress test | |
| Normal coronary arteries AND reduced LVEF on echo | |
| Angina pectoris | 1 |
| Positive SPECT | |
| Normal coronary arteries |
CSX cardiac syndrome X; AP angina pectoris; CAG coronary angiogram; SPECT single photon emission tomography; LVEF left ventricular ejection fraction
In more detail:
Out of the 57 studies 42 required positive exercise electrocardiograms for the diagnosis of CSX. The general criterion, when given, for a positive exercise stress test result was uniform ST depression ≥1 mm. Only one study considered stress-induced angina without significant ST depression to be positive for ischaemia [15].
As much as 11 studies regarded positive myocardial perfusion images to be good alternatives to a positive result from exercise testing.
As much as 18 studies considered effort-induced angina pectoris as an inclusion criterion.
Four studies defined CSX simply as angina pectoris and normal CAG, the so-called broad diagnosis of CSX.
Table 2 lists no less than 43 exclusion criteria for CSX. The most frequently used exclusion criteria are valvular heart disease, diabetes mellitus, left ventricular hypertrophy, hypertension, and cardiomyopathy.
Table 2.
Exclusion criteria included studies
| Exclusion criteria used in the studies for present review | N studies of 57 total |
|---|---|
| Valvular heart disease | 33 |
| Diabetes mellitus | 32 |
| Left ventricular hypertrophy | 26 |
| Hypertension | 24 |
| Cardiomyopathy (non specified) | 23 |
| Renal failure | 22 |
| History of myocardial infarction | 15 |
| LV dysfunction | 14 |
| Coronary spasm | 13 |
| Hepatic dysfunction | 12 |
| Arrhytmias | 9 |
| Inflammatory disease | 9 |
| Dyslipidaemia | 8 |
| Left bundel branch block | 8 |
| Cardiac disease (non specified) | 7 |
| Gastro Intestinal disorder | 7 |
| Systemic disease (non specified) | 6 |
| Smoking | 6 |
| Thyroid dysfunction | 5 |
| Obesitas | 5 |
| Non-cardiac chest pain | 5 |
| Condunction disorder (incl LBTB) | 5 (13) |
| Hypertrophic CMP (total CMP) | 5 (23) |
| Congestive CMP (total CMP) | 5 (23) |
| Dilated CMP (CMP total) | 4 (23) |
| Alcoholism | 4 |
| Metabolic syndrome | 4 |
| Dysphagia/oesophagitis | 3 |
| Malignancy | 3 |
| LVEF < 40% | 2 |
| Psychiatric ilness | 2 |
| Auto immune disease | 2 |
| Estrogen replacement therapy | 2 |
| PTCA/CABG in history | 2 |
| Ectasia on CAG | 2 |
| Bridging on CAG | 2 |
| Claudicatio intermittens | 2 |
| Heart failure (non specfied) (total) | 1 (17) |
| Myocarditis | 1 |
| Congenital heart disease | 1 |
| Aortic wall diseases | 1 |
| Anaemia | 1 |
| Thrombocytopenia | 1 |
In overviews and textbooks it is often stated that (pre-menopausal) women are especially prone to develop cardiac syndrome X.
In Table 3 data of individual studies are presented.
Table 3.
Included studies of CSX patients
| Included study of CSX patients | Year | n | Female (%) | Mean age (Y) |
|---|---|---|---|---|
| Lee [41] | 2008 | 21 | 81 | 55 |
| Timurkaynak [61] | 2008 | 79 | 54 | 50 |
| Cemin [11] | 2008 | 11 | 100 | 59 |
| Zorc-Pleskovic [66] | 2008 | 31 | 100 | 55 |
| Lanza [38] | 2008 | 18 | 61 | 58 |
| Altun [2] | 2007 | 9 | 77 | 49 |
| Cotrim [12] | 2008 | 91 | 48 | 51 |
| Asbury [5] | 2008 | 64 | 100 | 58 |
| Demir [18] | 2008 | 17 | 41 | 57 |
| Grabczewska [23] | 2007 | 53 | 57 | 55 |
| Li [44] | 2007 | 36 | 100 | NA |
| Okyay [48] | 2007 | 32 | 66 | 53 |
| Dabek [15] | 2007 | 34 | 65 | 57 |
| Yildiz [64] | 2007 | 10 | 79 | 49 |
| Kayikcioglu [33] | 2007 | 30 | 60 | 46 |
| Mao [45] | 2007 | 51 | 78 | 21 |
| Dabek [14] | 2007 | 36 | 65 | 57 |
| Vermeltfoort [63] | 2007 | 20 | 75 | 55 |
| Galiuto [21] | 2007 | 17 | 53 | 55 |
| Alroy [1] | 2007 | 42 | 100 | 52 |
| Huang [29] | 2007 | 12 | 60 | 63 |
| Guzik [28] | 2007 | 43 | 65 | 44 |
| Gur [26] | 2007 | 23 | 70 | 49 |
| Gur [27] | 2007 | 23 | NA | NA |
| Sgueglia [59] | 2007 | 30 | 73 | 61 |
| Russell [55] | 2007 | 24 | 89 | 54 |
| Sen [56] | 2007 | 203 | 58 | 53 |
| Shmilovich [60] | 2007 | 17 | 71 | 58 |
| de Vries [17] | 2006 | 42 | 26 | 58 |
| Nam [47] | 2006 | 52 | NA | NA |
| Eskandarian [19] | 2006 | 40 | 73 | 46 |
| Cay [10] | 2006 | 126 | 62 | 53 |
| Jadhav [30] | 2006 | 52 | 100 | 56 |
| Czepczynski [13] | 2006 | 68 | 63 | 45 |
| Leu [43] | 2006 | 92 | 21 | 64 |
| Lanza [40] | 2005 | 10 | 59 | 70 |
| Cay [9] | 2005 | 80 | 52 | 51 |
| On [49] | 2005 | 36 | 53 | 36 |
| Guo [25] | 2005 | 22 | 68 | 48 |
| Masci [46] | 2005 | 41 | NA | 53 |
| Senen [57] | 2005 | 21 | 48 | 56 |
| Pasqui [51] | 2005 | 30 | 60 | 54 |
| Sestito [58] | 2005 | 30 | 73 | 60 |
| De Candia [16] | 2005 | 21 | 57 | 55 |
| Valeriani [62] | 2005 | 16 | 50 | 60 |
| Cavusoglu [8] | 2005 | 31 | 45 | 52 |
| Kolasinska-Kloch [36] | 2004 | 42 | 52 | 46 |
| Kolasilska-Kloch [35] | 2004 | 25 | 36 | 49 |
| Altun [3] | 2004 | 8 | 75 | 46 |
| Asbury [4] | 2004 | 100 | 100 | 61 |
| Fabian [20] | 2004 | 40 | 40 | 55 |
| Lanza [39] | 2004 | 55 | 50 | 57 |
| Qian [54] | 2004 | 126 | 47 | 53 |
| Osamichi [50] | 2004 | 24 | 71 | 58 |
| Gorgulu [22] | 2003 | 18 | 44 | 51 |
| Pizzi [53] | 2004 | NA | NA | 59 |
| Kidawa [34] | 2003 | 50 | 64 | 49 |
NA not available
In 53 out of the total of 57 included studies the female/male distribution of the study population was mentioned. Six studies were focussed upon female patients with CSX. These studies were excluded for the pooling of the proportion of women in the study populations. Ultimately 47 studies with a total of 1,934 patients could be used for the meta-analysis.
The proportion of women in the 47 studies of CSX patients varied considerably ranging from 0.21 to 1.0, with a pooled estimate of 0.56 (95% confidence interval: 0.54–0.59), see also Fig. 1. Hence, there is a significantly higher proportion of woman compared to males in the group of patients with CSX.
Fig. 1.
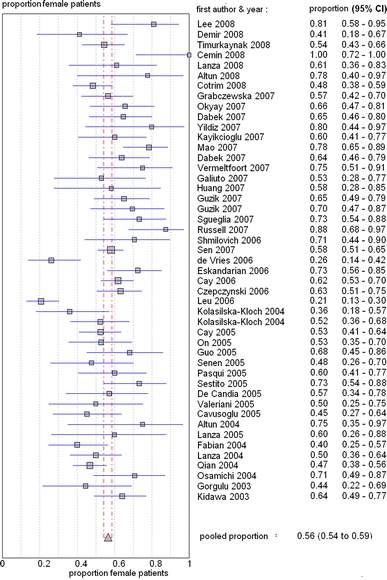
Plot of proportion of women of individual studies, including 95% confidence intervals
CSX incidence
As much as 567 CAGs were performed in 1 year at a general hospital. Figure 2 is a flowchart showing how we analysed the available information. The main stages are:
Original CAGs or clinical data could not be found for eight patients, leaving 559 cases for analysis.
Out of the available 559 CAGs 78 (14%) were considered as absolutely normal.
Excluding 13 cases with no angina pectoris as indication for CAG, and another 5 with an acute coronary syndrome, resulted in an incidence of 60 (11%) CSX patients according to the broad diagnosis of CSX (angina pectoris and normal coronary arteries) [17, 23, 54, 65, 66].
Another 15 cases were excluded owing to the lack of exercise stress tests or myocardial perfusion imaging preceding the CAG.
Of the remaining 45 cases, 28 were included owing to positive results for ischaemia from exercise tests or SPECT.
Finally, a much stricter definition of CSX was obtained by using the main exclusion criteria shown in the second-to-last stage in Fig. 2. This strict definition resulted in an attributable CSX incidence of 18 (3%) patients.
Fig. 2.
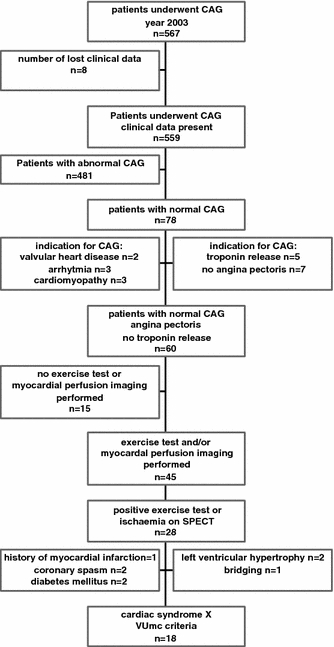
Flowchart of the available information
Discussion
CSX definitions and incidences
The present review of recent archival literature demonstrates that there is a wide range of definitions of CSX. This wide range of definitions has a large effect on the attributable incidence of CSX, as has been shown by an analysis of all available CAGs and other clinical data for a population of patients treated in 1 year at a general hospital.
Generally, it is stated that patients with chest pain and normal coronary arteriogram may represent 10–20% of those undergoing coronary arteriography because of clinical suspicion of angina [65]. This is in broad agreement with our analysis results that 11% of patients had a normal CAG. The rather low incidence of normal CAG can be the result of the use of rather strict criteria for a normal CAG including a consensus reading by two independent readers of the CAG’s.
It is generally accepted (e.g. in authoritative textbooks) that the majority of the CSX patients are women [32, 65]. Some authors have even suggested that CSX is a women’s disease. However, our review has found a pooled relative female frequency of 56% in a population of more than 1,900 CSX patients. Thus, our data do not support the assumption that CSX is a women’s disease, since 44% of the population was male. Potential pathophysiological explanations, such as estrogen depletion, which are based upon the female gender, apply only to a part of the CSX patients [31].
Inclusion and exclusion criteria
The literature survey showed that the inclusion and exclusion criteria varied. This was especially the case for the exclusion criteria.
The definition of ‘normal coronary arteries’ was particularly unclear. Most studies did not define a normal CAG, and some included patients with coronary artery disease (CAD) ranging from minimal to stenoses up to 50% of luminal diameter. Obviously, normal coronary arteries are the cornerstone of the diagnosis of CSX. Hence, there should be no doubt regarding the use of this inclusion criterion for studies of CSX patients. Future studies of CSX patients should make a clear description regarding the evaluation and results of the CAG studies of the coronary artery anatomy.
The so-called broad diagnosis for CSX, a combination of 2 inclusion criteria (angina pectoris and normal coronary arteries) was used only in four studies (7%).
Most studies used a combination of three inclusion criteria, namely (effort induced) angina pectoris, positive exercise test result, and a normal CAG. This definition was used in 46 out of 57 studies (81%). The use of this additional inclusion criterion resulted in a decrease of the incidence of CSX to 7% in our population. The definition of a positive exercise stress test appears to be more standardized than the definition of a normal CAG. Most publications used a ST depression ≥1 mm as a positive exercise stress test: only 12 out of the 57 studies did not define a positive stress test.
The use of specific exclusion criteria ranged from 2 to 58% of the selected studies of CSX patients, often depending on the main objectives of the studies, for example the use of thrombocytopenia as an exclusion criterion in a study investigating the mean platelet volume [9].
The most frequently mentioned exclusion criteria are valvular heart disease, diabetes mellitus, left ventricular hypertrophy, hypertension, and cardiomyopathy. Endothelial dysfunction has been assessed both in patients with diabetes mellitus and hypertension with normal CAG, and in patients with CSX without diabetes or hypertension. Interestingly, coronary flow reserve may be reversible in specific patient groups, e.g. in patients with hypertrophy after anti-hypertensive therapy [52]. However, we note that most international studies consider diabetes mellitus and hypertension to be exclusion criteria for CSX, thereby considering these patients as a separate group.
The existence of such a long list of exclusion criteria in the selection process of CSX patients illustrates the lack of agreement between the different research groups regarding the origins of this syndrome. Besides the use of a standard and fixed combination of inclusion criteria future studies should apply a standard combination of exclusion criteria.
In a recent editorial, Camici proposed the following exclusion criteria in order to obtain a more homogeneous set of cardiac syndrome X patients: absence of left bundle branch block; absence of even minimal irregularities on the angiogram; no evidence of diabetes mellitus, arterial hypertension, hyperlipidaemia, valve disease, epicardial arterial spasm, and cardiomyopathy [7].
In our selection of CSX patients we decided to include only patients with completely normal coronary angiograms, effort-induced angina pectoris, positive exercise stress test and/or positive SPECT study. Furthermore, we decided to add arrhythmias, left ventricular hypertrophy, myocardial infarction in the medical history, and myocardial bridging to the exclusion criteria proposed by Camici. These inclusion and exclusion criteria resulted in an attributable CSX incidence of only 3%.
Conclusions
The wide range of definitions of CSX in recent literature and the variations in inclusion and exclusion criteria, particularly the latter, make interpretation of the results of individual studies difficult. This shows the need for a generally accepted definition of CSX.
Acknowledgments
We thank Dr. R.J.H. Wanhill for his critical review and editing of the manuscript.
Conflict of interest statement
None.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
References
- 1.Alroy S, Preis M, Barzilai M, et al. Endothelial cell dysfunction in women with cardiac syndrome X and MTHFR C677T mutation. Isr Med Assoc J. 2007;9:321–325. [PubMed] [Google Scholar]
- 2.Altun A, Turgut N, Tatli E, et al. Sympathetic skin response and RR interval variation in patients with cardiac syndrome X. Angiology. 2007;58:747–750. doi: 10.1177/0003319707309117. [DOI] [PubMed] [Google Scholar]
- 3.Altun A, Ugur-Altun B, Tatli E. Decreased serum osteoprotegerin levels in patients with cardiac syndrome X. J Endocrinol Invest. 2004;27:839–843. doi: 10.1007/BF03346278. [DOI] [PubMed] [Google Scholar]
- 4.Asbury EA, Creed F, Collins P. Distinct psychosocial differences between women with coronary heart disease and cardiac syndrome X. Eur Heart J. 2004;25:1695–1701. doi: 10.1016/j.ehj.2004.07.035. [DOI] [PubMed] [Google Scholar]
- 5.Asbury EA, Slattery C, Grant A, et al. Cardiac rehabilitation for the treatment of women with chest pain and normal coronary arteries. Menopause. 2008;15:454–460. doi: 10.1097/gme.0b013e31815982eb. [DOI] [PubMed] [Google Scholar]
- 6.Athanasiadis A, Vogelsberg H, Hauer B, et al. Transient left ventricular dysfunction with apical ballooning (tako-tsubo cardiomyopathy) in Germany. Clin Res Cardiol. 2006;95:321–328. doi: 10.1007/s00392-006-0380-0. [DOI] [PubMed] [Google Scholar]
- 7.Camici PG. Is the chest pain in cardiac syndrome X due to subendocardial ischaemia? Eur Heart J. 2007;28:1539–1540. doi: 10.1093/eurheartj/ehm167. [DOI] [PubMed] [Google Scholar]
- 8.Cavusoglu Y, Entok E, Timuralp B, et al. Regional distribution and extent of perfusion abnormalities, and the lung to heart uptake ratios during exercise thallium-201 SPECT imaging in patients with cardiac syndrome X. Can J Cardiol. 2005;21:57–62. [PubMed] [Google Scholar]
- 9.Cay S, Biyikoglu F, Cihan G, et al. Mean platelet volume in the patients with cardiac syndrome X. J Thromb Thrombolysis. 2005;20:175–178. doi: 10.1007/s11239-005-4047-x. [DOI] [PubMed] [Google Scholar]
- 10.Cay S, Guray U, Korkmaz S. Increased aortic pulse and fractional pulse pressures in patients with cardiac syndrome X. Blood Press. 2006;15:179–184. doi: 10.1080/08037050600804830. [DOI] [PubMed] [Google Scholar]
- 11.Cemin R, Erlicher A, Fattor B, et al. Reduced coronary flow reserve and parasympathetic dysfunction in patients with cardiovascular syndrome X. Coron Artery Dis. 2008;19:1–7. doi: 10.1097/MCA.0b013e3282f18e8d. [DOI] [PubMed] [Google Scholar]
- 12.Cotrim C, Almeida AG, Carrageta M. Exercise-induced intra-ventricular gradients as a frequent potential cause of myocardial ischemia in cardiac syndrome X patients. Cardiovasc Ultrasound. 2008;6:3. doi: 10.1186/1476-7120-6-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Czepczynski R, Smolarek I, Rogacka D, et al. Myocardial perfusion SPECT with dipyridamole stress test in cardiac syndrome X. Nuklearmedizin. 2006;45:111–114. [PubMed] [Google Scholar]
- 14.Dabek J, Kulach A, Wilczok T, et al. Transcriptional activity of genes encoding interferon gamma (IFNgamma) and its receptor assessed in peripheral blood mononuclear cells in patients with cardiac syndrome X. Inflammation. 2007;30:125–129. doi: 10.1007/s10753-007-9028-0. [DOI] [PubMed] [Google Scholar]
- 15.Dabek J, Wilczok T, Gasior Z, et al. Gene expression of kinin receptors B1 and B2 in PBMC from patients with cardiac syndrome X. Scand Cardiovasc J. 2007;41:391–396. doi: 10.1080/14017430701499379. [DOI] [PubMed] [Google Scholar]
- 16.De Candia E, Lanza GA, Romagnoli E, et al. Abnormal pH-sensing of platelet Na+/H+ exchanger in patients with cardiac syndrome X. Int J Cardiol. 2005;100:371–376. doi: 10.1016/j.ijcard.2004.03.078. [DOI] [PubMed] [Google Scholar]
- 17.de Vries J, DeJongste MJ, Jessurun GA, et al. Myocardial perfusion quantification in patients suspected of cardiac syndrome X with positive and negative exercise testing: a [13N]ammonia positron emission tomography study. Nucl Med Commun. 2006;27:791–794. doi: 10.1097/01.mnm.0000237984.46844.42. [DOI] [PubMed] [Google Scholar]
- 18.Demir H, Kahraman G, Isgoren S, et al. Evaluation of post-stress left ventricular dysfunction and its relationship with perfusion abnormalities using gated SPECT in patients with cardiac syndrome X. Nucl Med Commun. 2008;29:208–214. doi: 10.1097/MNM.0b013e3282f52c49. [DOI] [PubMed] [Google Scholar]
- 19.Eskandarian R, Malek M, Mousavi SH, et al. Association of Helicobacter pylori infection with cardiac syndrome X. Singapore Med J. 2006;47:704–706. [PubMed] [Google Scholar]
- 20.Fabian E, Varga A, Picano E, et al. Effect of simvastatin on endothelial function in cardiac syndrome X patients. Am J Cardiol. 2004;94:652–655. doi: 10.1016/j.amjcard.2004.05.035. [DOI] [PubMed] [Google Scholar]
- 21.Galiuto L, Sestito A, Barchetta S, et al. Noninvasive evaluation of flow reserve in the left anterior descending coronary artery in patients with cardiac syndrome X. Am J Cardiol. 2007;99:1378–1383. doi: 10.1016/j.amjcard.2006.12.070. [DOI] [PubMed] [Google Scholar]
- 22.Gorgulu S, Uslu N, Eren M, et al. Aortic stiffness in patients with cardiac syndrome X. Acta Cardiol. 2003;58:507–511. doi: 10.2143/AC.58.6.2005314. [DOI] [PubMed] [Google Scholar]
- 23.Grabczewska Z, Thews M, Goralczyk K, et al. Endothelial function in patients with chest pain and normal coronary angiograms. Kardiol Pol. 2007;65:1199–1206. [PubMed] [Google Scholar]
- 24.Grawe H, Katoh M, Kuhl HP. Stress cardiomyopathy mimicking acute coronary syndrome: case presentation and review of the literature. Clin Res Cardiol. 2006;95:179–185. doi: 10.1007/s00392-006-0346-2. [DOI] [PubMed] [Google Scholar]
- 25.Guo H, Lee JD, Guo M, et al. Status of intracellular and extracellular magnesium concentration in patients with cardiac syndrome X. Acta Cardiol. 2005;60:259–263. doi: 10.2143/AC.60.3.2005001. [DOI] [PubMed] [Google Scholar]
- 26.Gur M, Yildiz A, Demirbag R, et al. Paraoxonase and arylesterase activities in patients with cardiac syndrome X, and their relationship with oxidative stress markers. Coron Artery Dis. 2007;18:89–95. doi: 10.1097/MCA.0b013e32801104e8. [DOI] [PubMed] [Google Scholar]
- 27.Gur M, Yildiz A, Demirbag R, et al. Increased lymphocyte deoxyribonucleic acid damage in patients with cardiac syndrome X. Mutat Res. 2007;617:8–15. doi: 10.1016/j.mrfmmm.2006.08.012. [DOI] [PubMed] [Google Scholar]
- 28.Guzik P, Rogacka D, Trachalski J, et al. Comparison of the exercise treadmill test and 24-hour ECG Holter monitoring in patients with syndrome X or coronary atherosclerosis. Kardiol Pol. 2007;65:262–269. [PubMed] [Google Scholar]
- 29.Huang PH, Chen YH, Chen YL, et al. Vascular endothelial function and circulating endothelial progenitor cells in patients with cardiac syndrome X. Heart. 2007;93:1064–1070. doi: 10.1136/hrt.2006.107763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Jadhav ST, Ferrell WR, Petrie JR, et al. Microvascular function, metabolic syndrome, and novel risk factor status in women with cardiac syndrome X. Am J Cardiol. 2006;97:1727–1731. doi: 10.1016/j.amjcard.2005.12.069. [DOI] [PubMed] [Google Scholar]
- 31.Kaski JC. Cardiac syndrome X in women: the role of oestrogen deficiency. Heart. 2006;92(Suppl 3):iii5–iii9. doi: 10.1136/hrt.2005.070318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kaski JC. Overview of gender aspects of cardiac syndrome X. Cardiovasc Res. 2002;53:620–626. doi: 10.1016/S0008-6363(01)00460-6. [DOI] [PubMed] [Google Scholar]
- 33.Kayikcioglu M, Saygi S, Azarsiz E, et al. Serum paraoxonase 1 activity and oxidative markers of LDL in patients with cardiac syndrome X. Acta Cardiol. 2007;62:245–249. doi: 10.2143/AC.62.3.2020812. [DOI] [PubMed] [Google Scholar]
- 34.Kidawa M, Krzeminska-Pakula M, Peruga JZ, et al. Cardiological syndrome X Non-invasive assessment of endothelial function and arterial compliance. Kardiol Pol. 2003;59:385–396. [PubMed] [Google Scholar]
- 35.Kolasilska-Kloch W, Lesniak W, Leszczynska-Golabek I, et al. Exercise tolerance and hyperinsulinemia in cardiac syndrome X. Folia Med Cracov. 2004;45:13–23. [PubMed] [Google Scholar]
- 36.Kolasinska-Kloch W, Furgala A, Krolczyk G, et al. Cardiac syndrome X-autonomic system disorders. Folia Med Cracov. 2004;45:19–29. [PubMed] [Google Scholar]
- 37.Lanza GA. Cardiac syndrome X: a critical overview and future perspectives. Heart. 2007;93:159–166. doi: 10.1136/hrt.2005.067330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lanza GA, Buffon A, Sestito A, et al. Relation between stress-induced myocardial perfusion defects on cardiovascular magnetic resonance and coronary microvascular dysfunction in patients with cardiac syndrome X. J Am Coll Cardiol. 2008;51:466–472. doi: 10.1016/j.jacc.2007.08.060. [DOI] [PubMed] [Google Scholar]
- 39.Lanza GA, Sestito A, Cammarota G, et al. Assessment of systemic inflammation and infective pathogen burden in patients with cardiac syndrome X. Am J Cardiol. 2004;94:40–44. doi: 10.1016/j.amjcard.2004.03.027. [DOI] [PubMed] [Google Scholar]
- 40.Lanza GA, Sestito A, Sgueglia GA, et al. Effect of spinal cord stimulation on spontaneous and stress-induced angina and ‘ischemia-like’ ST-segment depression in patients with cardiac syndrome X. Eur Heart J. 2005;26:983–989. doi: 10.1093/eurheartj/ehi089. [DOI] [PubMed] [Google Scholar]
- 41.Lee BK, Durairaj A, Mehra A, et al. Microcirculatory dysfunction in cardiac syndrome X: role of abnormal blood rheology. Microcirculation. 2008;15:451–459. doi: 10.1080/10739680701797090. [DOI] [PubMed] [Google Scholar]
- 42.Leichman JG, Lavis VR, Aguilar D, et al. The metabolic syndrome and the heart—a considered opinion. Clin Res Cardiol. 2006;95(Suppl 1):i134–i141. doi: 10.1007/s00392-006-1119-7. [DOI] [PubMed] [Google Scholar]
- 43.Leu HB, Lin CP, Lin WT, et al. Circulating mononuclear superoxide production and inflammatory markers for long-term prognosis in patients with cardiac syndrome X. Free Radic Biol Med. 2006;40:983–991. doi: 10.1016/j.freeradbiomed.2005.10.047. [DOI] [PubMed] [Google Scholar]
- 44.Li JJ, Zhu CG, Nan JL, et al. Elevated circulating inflammatory markers in female patients with cardiac syndrome X. Cytokine. 2007;40:172–176. doi: 10.1016/j.cyto.2007.09.005. [DOI] [PubMed] [Google Scholar]
- 45.Mao JY, Ge YB, Wang HH, et al. Summary of 32 patients with cardiac syndrome X treated by TCM therapy of regulating qi relieving chest stuffiness and promoting blood circulation. Chin J Integr Med. 2007;13:17–21. doi: 10.1007/s11655-007-0017-9. [DOI] [PubMed] [Google Scholar]
- 46.Masci PG, Laclaustra M, Lara JG, et al. Brachial artery flow-mediated dilation and myocardial perfusion in patients with cardiac syndrome X. Am J Cardiol. 2005;95:1478–1480. doi: 10.1016/j.amjcard.2005.02.018. [DOI] [PubMed] [Google Scholar]
- 47.Nam CW, Kim KS, Lee YS, et al. The incidence of gastro-esophageal disease for the patients with typical chest pain and a normal coronary angiogram. Korean J Intern Med. 2006;21:94–96. doi: 10.3904/kjim.2006.21.2.94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Okyay K, Cengel A, Sahinarslan A, et al. Plasma asymmetric dimethylarginine and L-arginine levels in patients with cardiac syndrome X. Coron Artery Dis. 2007;18:539–544. doi: 10.1097/MCA.0b013e3282f08ece. [DOI] [PubMed] [Google Scholar]
- 49.On YK, Park R, Hyon MS, et al. Are low total serum antioxidant status and elevated levels of C-reactive protein and monocyte chemotactic protein-1 associated with cardiac syndrome X? Circ J. 2005;69:1212–1217. doi: 10.1253/circj.69.1212. [DOI] [PubMed] [Google Scholar]
- 50.Osamichi S, Kouji K, Yoshimaro I, et al. Myocardial glucose metabolism assessed by positron emission tomography and the histopathologic findings of microvessels in syndrome X. Circ J. 2004;68:220–226. doi: 10.1253/circj.68.220. [DOI] [PubMed] [Google Scholar]
- 51.Pasqui AL, Puccetti L, Di Renzo M, et al. Structural and functional abnormality of systemic microvessels in cardiac syndrome X. Nutr Metab Cardiovasc Dis. 2005;15:56–64. doi: 10.1016/j.numecd.2004.05.001. [DOI] [PubMed] [Google Scholar]
- 52.Petkow Dimitrow P, Krzanowski M, Nizankowski R, et al. Effect of verapamil on systolic and diastolic coronary blood flow velocity in asymptomatic and mildly symptomatic patients with hypertrophic cardiomyopathy. Heart. 2000;83:262–266. doi: 10.1136/heart.83.3.262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Pizzi C, Manfrini O, Fontana F, et al. Angiotensin-converting enzyme inhibitors and 3-hydroxy-3-methylglutaryl coenzyme A reductase in cardiac Syndrome X: role of superoxide dismutase activity. Circulation. 2004;109:53–58. doi: 10.1161/01.CIR.0000100722.34034.E4. [DOI] [PubMed] [Google Scholar]
- 54.Qian JY, Ge JB, Fan B, et al. Identification of syndrome X using intravascular ultrasound imaging and Doppler flow mapping. Chin Med J (Engl) 2004;117:521–527. [PubMed] [Google Scholar]
- 55.Russell SJ, Di Stefano EM, Naffati MT, et al. The effects of the angiotensin II receptor (type I) antagonist irbesartan in patients with cardiac syndrome X. Heart. 2007;93:253–254. doi: 10.1136/hrt.2006.089904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Sen N, Tavil Y, Yazici HU, et al. Coronary blood flow in patients with cardiac syndrome X. Coron Artery Dis. 2007;18:45–48. doi: 10.1097/MCA.0b013e32801104fc. [DOI] [PubMed] [Google Scholar]
- 57.Senen K, Ileri M, Alper A, et al. Increased levels of soluble adhesion molecules E-selectin and P-selectin in patients with cardiac syndrome X. Angiology. 2005;56:273–277. doi: 10.1177/000331970505600306. [DOI] [PubMed] [Google Scholar]
- 58.Sestito A, Maccallini A, Sgueglia GA, et al. Platelet reactivity in response to mental stress in syndrome X and in stable or unstable coronary artery disease. Thromb Res. 2005;116:25–31. doi: 10.1016/j.thromres.2004.10.002. [DOI] [PubMed] [Google Scholar]
- 59.Sgueglia GA, Sestito A, Spinelli A, et al. Long-term follow-up of patients with cardiac syndrome X treated by spinal cord stimulation. Heart. 2007;93:591–597. doi: 10.1136/hrt.2006.102194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Shmilovich H, Deutsch V, Roth A, et al. Circulating endothelial progenitor cells in patients with cardiac syndrome X. Heart. 2007;93:1071–1076. doi: 10.1136/hrt.2005.077909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Timurkaynak T, Balcioglu S, Arslan U, et al. Plasma homocysteine level in cardiac syndrome X and its relation with duke treadmill score. Saudi Med J. 2008;29:364–367. [PubMed] [Google Scholar]
- 62.Valeriani M, Sestito A, Le Pera D, et al. Abnormal cortical pain processing in patients with cardiac syndrome X. Eur Heart J. 2005;26:975–982. doi: 10.1093/eurheartj/ehi229. [DOI] [PubMed] [Google Scholar]
- 63.Vermeltfoort IA, Bondarenko O, Raijmakers PG, et al. Is subendocardial ischaemia present in patients with chest pain and normal coronary angiograms? A cardiovascular MR study. Eur Heart J. 2007;28:1554–1558. doi: 10.1093/eurheartj/ehm088. [DOI] [PubMed] [Google Scholar]
- 64.Yildiz M, Altun A, Ozbay G. Assessment of arterial distensibility in patients with cardiac syndrome X. Angiology. 2007;58:458–462. doi: 10.1177/0003319707305064. [DOI] [PubMed] [Google Scholar]
- 65.Zipes DP, Libby P, Bonow RO, et al. Braunwald’s heart disease: a textbook of cardiovascular medicine, 8 edn. Philadelphia: Saunders-Elsevier; 2008. [Google Scholar]
- 66.Zorc-Pleskovic R, Vraspir-Porenta O, Zorc M, et al. Inflammatory changes in small blood vessels in the endomyocardium of cardiac syndrome X in female patients with increased C-reactive protein. Folia Biol (Praha) 2008;54:30–32. [PubMed] [Google Scholar]


