Abstract
During the active tooth eruption process, structural changes in the lamina propria are necessary to provide extracellular matrix remodelling and for the establishment of the eruptive pathway. A large number of resident cells, recruited cells and proteases have been demonstrated in the eruptive process, but the participation of MMP-9 and mast cells has not yet been demonstrated. In this study, we set out to evaluate the intensity of MMP-9 immunoexpression, the frequency of mast cells and the correlation between the incidence of mast cells and bone resorption in different phases of tooth eruption. Fragments of maxilla containing first molars, obtained from 9-, 11-, 13- and 16-day-old rats, were fixed in 4% formaldehyde, decalcified and embedded in paraffin. Sagittal sections were stained with Masson’s trichrome or submitted to the tartrate-resistant acid phosphatase method for quantification of osteoclasts. Sections stained by 1% toluidine blue were used for quantification of metachromatic mast cells mm−2 of lamina propria. The expression of MMP-9 in the lamina propria was evaluated by immunohistochemistry. In the 9-day-old rats, the lamina propria contained few mast cells and occasional osteoclasts were found in the bone surface overlying the occlusal portion of the tooth germs. Otherwise, a significant increase in the number of mast cells was observed in the intra-osseous phase of tooth eruption (11-day-old rats), period in which numerous TRAP-positive osteoclasts were found in the bone surface. MMP-9 immunolabelling was detected in fibroblasts, mast cells and macrophage-like cells of the lamina propria in all ages studied. However, an enhanced immunolabelling was evident in the advanced phase of tooth eruption (16-day-old rats). During the intra-osseous phase, the parallel between the high frequency of both mast cells and osteoclasts suggests that mast cells could exert a paracrine function on the osteoclasts and then stimulate bone resorption. The immunoexpression of MMP-9 in different cells of lamina propria, including mast cells, indicates that this enzyme participates in the degradation of ECM, mainly during late phase of mucosal penetration. Thus mast cells and MMP-9 are involved in the complex process of degradation of the eruptive pathway extracellular matrix.
Keywords: eruptive pathway, immunohistochemistry, lamina propria, mast cells, metalloproteinase-9, oral mucosa
Introduction
Tooth eruption is a programmed and localized process that involves a complex cascade of cellular and molecular events which are precisely controlled (Wise et al. 2002). Usually, this process is divided into five stages: pre-eruptive movements, intra-osseous eruption, mucosal penetration, pre-occlusal eruption and post-occlusal eruption. The initial development of the tooth germs occurs within the bony crypt; however, when the dentine root begins to form, the tooth move towards the oral cavity by an active eruption process, involving bone resorption (Marks, 1995; Marks & Schroeder, 1996).
A coordinated bone remodelling process is observed in the intra-osseous phase to accommodate the growing and erupting tooth germs. In this stage, it has been demonstrated that enamel organ and dental follicle produce and release several molecules required for eruption. Factors such as epidermal growth factor (EGF), interleukin-1 (IL-1), colony-stimulating factor-1 (CSF-1) and monocyte chemotactic protein-1 (MCP-1) (Wise, 2009) stimulate the migration of mononuclear precursors of osteoclasts for occlusal portion of the bony crypt (Wise et al. 1985; Wise & Fan, 1989; Gorski, 1992). The influx of mononuclear precursors is accomplished by an increase in osteoclasts and intense bone resorption (Wise et al. 1985; Wise & Fan, 1989; Wise, 2009). After resorption of occlusal portion of the bony crypt, the cusps invade the oral mucosa, characterized as the stage of mucosal penetration. At this stage, the rate of eruption accelerates and, therefore, extensive changes in the cells and in the components of the extracellular matrix (ECM) may occur for establishment of the eruptive pathway and tissue remodelling.
During biological processes such as embryonic development, tissue morphogenesis, tissue remodelling, wound repair, inflammatory diseases and cancer, the role of enzymes, especially metalloproteinases (MMPs), is essential (Sternlicht & Werb, 2001; Gonçalves et al. 2008). MMPs are a family of structurally related zinc-dependent endopeptidases responsible for degradation of different macromolecular components of the ECM. These enzymes are synthesized with an N-terminal propeptide that needs to be removed to achieve proteolytic activity (Sternlicht & Werb, 2001; Tchougounova et al. 2005; Page-McCaw et al. 2007). At least 23 members of the MMP family have been characterized (Page-McCaw et al. 2007); among them, MMP-9, also known as gelatinase B, degrades denatured collagens, type IV and type V collagens, anchoring collagen type VII, fibronectin and elastin (Birkedal-Hansen et al. 1993; Tanaka et al. 1999). This MMP is produced by numerous cell types, including fibroblasts, macrophages (Ogata et al. 1992; Sternlicht & Werb, 2001; Norrby, 2002; Shimizu et al. 2005; Takahashi et al. 2006) and mast cells (Fang et al. 1999; Tanaka et al. 1999). Moreover, it has been suggested that MMP-9 plays a crucial role in the bone resorption, as osteoclast expresses high levels of MMP-9 (Okada et al. 1995; Okaji et al. 2003; Ishibashi et al. 2006).
It has been demonstrated that mast cells play a fundamental role in tissue homeostasis, remodelling and repair (Tchougounova et al. 2005). Mast cells stimulate other cells to release cytokines and enzymes, including MMPs, during tissue degradation and/or remodelling (Artuc et al. 2002). Moreover, these cells are also able to produce potential molecules to mediate breakdown of components of ECM such as transforming growth factor beta (TGF-β), tumour necrosis factor-α (TNF-α), interleukins (IL-1β, -3, -4, 6, -8 and -13), tryptase, chymase and MMPs (Steinsvoll et al. 2004). In the oral mucosa, Naesse et al. (2003) have demonstrated that mast cells produce MMP-1, MMP-2 and MMP-8 for degradation of extracellular matrix components of the gingival lamina propria. Therefore, it has been demonstrated that mast cells are essential in collagen breakdown and play important role in the tissue degradation and remodelling (Naesse et al. 2003; Steinsvoll et al. 2004).
It is known that a high number of proteases and resident and recruited cells participate in the eruptive process. However, MMP-9 and mast cells have not yet been demonstrated. The aim of the present study was to evaluate the presence of mast cells and MMP-9 in the lamina propria during tooth eruption of rat molars. For this purpose, quantitative analysis of these cells, a correlation with bone resorption and the intensity of MMP-9 immunoexpression in the lamina propria were also evaluated at different phases of tooth eruption.
Materials and methods
This study was performed in accordance with the principles of laboratory animal care (NIH publication 85–23, 1985). National laws on animal use were observed for the present study, which was authorized by the Ethical Committee for Animal Research of the São Paulo State University, Brazil (Araraquara Dental School-UNESP).
Twenty Holtzman male rats, 9, 11, 13 and 16 days old, were used in the present study. The animals were killed with chloral hydrate (600 mg kg−1), decapitated and the upper maxilla removed. With the help of a dissecting microscope the fragments of the maxilla containing the first molars were removed and placed in the fixative solution.
Paraffin embedding
The fragments of maxilla containing the oral mucosa overlaying the first molar germs were fixed in 4% formaldehyde (prepared from paraformaldehyde) buffered at pH 7.2 with 0.1 m sodium phosphate for 48 h at room temperature. Subsequently, the specimens were decalcified for 7 days in a 7% solution of ethylenediaminetetraacetic acid (EDTA) containing 0.5% formaldehyde, buffered at pH 7.2 with 0.1 m sodium phosphate. After decalcification, the specimens were dehydrated in graded concentrations of ethanol and embedded in paraffin. Sagittal sections, 6 μm thick, were stained with Masson’s trichrome for morphological analyses; sections stained with 1% toluidine blue were used for quantification of mast cells in the lamina propria of the eruptive pathway. The tartrate-resistant acid phosphatase (TRAP) reaction was used to quantify the osteoclast number in the bone surface. Sections adhered to silanized slides (3-aminopropylethoxysilane; Sigma-Aldrich Chemical Company, St. Louis, MO, USA) were submitted to immunohistochemistry for detection of MMP-9.
Number of mast cells in the lamina propria
The mast cells of the lamina propria overlaying the first upper molar germs were quantified in the toluidine blue-stained sections. Five non-serial sections per animal were used; the shortest distance between the sections was 60 μm. An eyepiece containing a square grid was attached to the light microscope ocular and the number of metachromatic mast cells with purple/violet granules was counted at × 400. The quantification was made in several square fields of the lamina propria in the five sections, totalling a standardized area of 0.8 mm2 of lamina propria/animal; thus, the number of mast cells mm−2 of lamina propria per animal was obtained.
Number of TRAP-positive osteoclasts in the bone surface overlaying the tooth germ
Sections of maxilla containing the first molar in eruption were submitted to TRAP, used as an osteoclast marker (Minkin, 1982; Cerri et al. 2003; Faloni et al. 2007). The sections were incubated in solution prepared by dissolving 8 mg naphthol AS-BI (Sigma Chemical Company) in 500 μL of N-N-dimethylformamide (Sigma Chemical Company), followed by addition of 50 mL of 0.2 m sodium acetate buffer (pH 5.0) containing 70 mg of Fast Red Salt (Sigma Chemical Company). Sodium tartrate (50 mm) was added and the medium was filtered. After incubation at 37 °C, the sections were washed in distilled water and counterstained with toluidine blue.
The number of TRAP-positive osteoclasts apposed to the bone surface overlaying the occlusal portion of the tooth germs was quantified in the 9- and 11-day-old rats. In 13- and 16-day-old rats, the number of osteoclasts was not quantified because the bone had yet been resorbed. In each animal, two sections were selected at intervals of at least 100 μm and the linear bone surface (in mm) was measured using an image analysis system (image-pro express 6.0; Olympus) at × 10. Multinucleated TRAP-positive osteoclasts on the bone surface were counted using a light microscope (Olympus, BX-51), at × 400 magnification (Faloni et al. 2007; Cruzoé-Souza et al. 2009).
Statistical analysis
The differences between the groups were statistically analyzed by sigmastat 2.0 software (Jandel Scientific, Sausalito, CA, USA) following one-way analysis of variance. The Tukey test was applied and the significance level accepted was P ≤ 0.05.
Immunohistochemistry for MMP-9
For detection of MMP-9, a mouse primary antibody anti-human MMP-9 (Calbiochem®; Biochemicals and Immunochemicals, USA) was used; this purified mouse monoclonal antibody recognizes both the ∼ 92-kDa latent and the ∼ 83-kDa active forms of MMP-9. The sections were deparaffinized and immersed in 0.001 m sodium citrate buffer pH 6.0 and maintained at 90–94 °C in a microwave oven for 30 min (Sasso-Cerri et al. 2005; Cruzoé-Souza et al. 2009). After a cooling-off period, the endogenous peroxidase was blocked with 3% hydrogen peroxide for 20 min. The sections were washed in 0.05 m Tris–HCl buffered saline at pH 7.2 (TBS) and treated with 2% bovine serum albumin (BSA) for 20 min at room temperature. Subsequently, the sections were incubated with the primary antibody anti-human MMP-9 diluted 1 : 500 in TBS for 16 h in a humidified chamber at 4 °C. After washings in TBS, the immunoreaction was detected by Vectastain Kit (Vector Laboratories, Inc., Burlingame, CA, USA). Sections were incubated at room temperature, with biotinylated anti-mouse IgG for 30 min, and, after washings in TBS, were incubated with avidin-biotin-peroxidase complex for 30 min. Peroxidase activity was revealed by 0.06% 3,3′-diaminobenzidine (Sigma-Aldrich, Chemie GmbH, Germany) in TBS; some sections were counterstained with Carazzi’s haematoxylin. As negative controls, the immunohistochemical reaction was performed replacing the primary antibody by BSA.
Results
Morphological aspects
Stained sections of maxillas of 9-day-old rats revealed that first molar germs exhibited coronal dentine and enamel at an advanced stage of formation; in the most cervical portion of the enamel organ, the onset of root formation with a thin layer of dentine matrix was observed (Fig. 1). The developing tooth germs were inside the bony crypt and therefore a bone trabeculae layer was interposed between the molar germs and oral mucosa; some multinucleated osteoclasts apposed to the bone surface, in the occlusal portion, were observed (Figs 1 and 2A). In the 11-day-old rats, the portions of the bony crypt covering the cusp tips of the first molars were partially resorbed (Fig. 2B). In 13-day-old rats, the cusp tips of the developing tooth germs were crossing the thin layer of lamina propria of the eruptive pathway (Fig. 2C). The first upper molars, in the 16-day-old rats, showed an advanced stage of tooth eruption; usually, the reduced enamel epithelium on the molar cusps was thick and fused with the oral epithelium. Consequently, the lamina propria was often restricted to small regions between the molar cusps (Fig. 2D).
Fig. 1.

Light micrograph of a sagittal section of the first molar tooth germ of 9-day-old rat. Developing tooth germ containing dental papilla (DP), dentine (D) and enamel (E) is located inside bony crypt (B). In the cervical region, the initial stage of root dentine formation (white arrows) is observed. Ob, odontoblasts; A, ameloblasts; LP, lamina propria; OE, oral epithelium. Masson’s trichrome. Bar: 200 μm.
Fig. 2.

Light micrographs of portions of first molar tooth germs, in different stages of eruption, of 9- (A), 11- (B), 13- (C) and 16-day-old (D) rats, stained with Masson's trichrome. (2A) A continuous layer of bone trabeculae (B) is interposed between the tooth germ (GE) and the lamina propria (LP). OE, oral epithelium; E, enamel. Bar: 100 μm. (2B) Cusp tips of the tooth molar germ in close proximity to lamina propria (LP); bone trabeculae (B) partially covers the occlusal portion of the tooth germ. E, enamel; D, dentine; OE, oral epithelium. Bar: 100 μm. (2C) The cusp tips of the tooth germ penetrate in the lamina propria (LP) which is restricted to a thin layer. OE, oral epithelium; E, enamel; ES, enamel space; D, dentine; RE, reduced enamel epithelium. Bar: 100 μm. (2D) The molar germ passing through the oral epithelium (OE). Note that the lamina propria (LP) is restricted to small regions. DP, dental papilla; D, dentine; ES, enamel space; RE, reduced enamel epithelium. Bar: 100 μm.
Toluidine blue-stained sections revealed metachromatic mast cells with purple/violet granules in the lamina propria of the eruptive pathway in all ages analyzed. However, numerous mast cells were found in the 11-day-old rats compared with other ages (Fig. 3A,B). In the 11-day-old rats, the increased density of mast cells coexisted with numerous TRAP-positive osteoclasts (red staining) apposed to the bone surface covering the occlusal portion of the tooth germs (Fig. 3C).
Fig. 3.

Light micrographs of portions of the eruptive pathway of the first molar stained with toluidine blue (A and B) and, submitted to the TRAP-method and counterstained with toluidine blue (C). (3A) Several mast cells (arrows) in the lamina propria (LP) of an 11-day-old-rat. Note the metachromasia (purple colour) in the mast cell (inset). D, dentine; ES, enamel space; RE, reduced enamel epithelium; B, bone; OE, oral epithelium. Bar: 60 μm; inset bar: 5 μm. (3B) Portion of erupting tooth germ of a 13-day-old-rat. Scarce mast cells (arrows) are seen in the lamina propria (LP). ES, enamel space; D, dentine; RE, reduced enamel epithelium; OE, oral epithelium. Bar: 60 μm. (3C) Several TRAP-positive osteoclasts (OC) on the bone surface (B) and mast cells (arrows) are seen in the lamina propria (LP) of an 11-day-old rat. OE, oral epithelium. Bar: 10 μm.
Numerical density of mast cells and number of TRAP-positive osteoclasts
According to Table 1, the number of mast cells in the lamina propria was 29.3, 81.5, 36.0 and 36.0 mast cells mm−2 in 9-, 11-, 13- and 16-day-old rats, respectively. Thus, a significant increase was observed in the 11-day-old rats in comparison with other ages. It is important to note that after 11 days of age, the number of mast cells decreased in both 13- and 16-day-old rats, but was higher than in 9-day-old rats.
Table 1.
Number of mast cells per mm2 of lamina propria of the eruptive pathway in 9-, 11-, 13- and 16-day-old rats.
| Rats | 9-day-old | 11-day-old* | 13-day-old | 16-day-old |
|---|---|---|---|---|
| 1 | 35.6 | 124.0 | 34.5 | 33.7 |
| 2 | 17.2 | 78.0 | 22.0 | 23.5 |
| 3 | 26.4 | 92.3 | 52.0 | 59.1 |
| 4 | 29.4 | 52.8 | 35.4 | 31.4 |
| 5 | 37.7 | 60.7 | 36.4 | 33.5 |
| Mean ± SD | 29.2 ± 8.1 | 81.5 ± 28.3 | 36.0 ± 10.6 | 36.2 ± 13.4 |
Statistically significant (P ≤ 0.05).
In the 9- and 11-day-rats, the number of TRAP-positive osteoclasts apposed to the bone surface overlaying the occlusal portion of the first molar was 3.3 and 10.8 osteoclasts mm−1 of bone surface, respectively. This significant increase was proportional to the increase in the number of mast cells from 9 to 11 days (Fig. 4).
Fig. 4.

Number of TRAP-positive osteoclasts mm−1 of alveolar bone surface and mast cells mm−2 of lamina propria in rats aged 9 and 11 days. The number of mast cells and osteoclasts increased around 30% from 9- to 11-day-old rats. *Statistically significant (P ≤ 0.05).
MMP-9 immunoreaction
Sections of maxilla containing the eruptive pathway of the first molar submitted to immunohistochemistry for MMP-9 detection exhibited positive immunolabelling in the cytoplasm of different cells of the lamina propria, characterized by brown stain (Figs 5A–F and 6A–G). In the initial phases (9- and 11-day-old rats), the immunolabelling was observed in occasional cells of the lamina propria (Fig. 5A–F); in addition, a conspicuous immunostaining in the osteoclasts apposed to the bone surface was seen in the occlusal portion (Fig. 5A,D). Otherwise, in the advanced phases of tooth eruption (13- and 16-day-old rats), a strong immunoreaction was often found in the cytoplasm of several cells of lamina propria (Fig. 6A–G). The cytoplasm of fibroblasts showed a positive reaction to MMP-9 immunohistochemistry in all ages studied. However, an enhanced positive immunolabelling was evident in the fibroblasts of the 13- and 16-day-old rats (Fig. 6B,C,F,G) compared with the initial periods (Fig. 5B,E,F). MMP-9 was also detected in the cytoplasm of mast cells in different phases of tooth eruption (Figs 5C,F and 6B,F). In the advanced phases of eruption (13- and 16-day-old rats), a conspicuous immunoreaction was observed in the cytoplasm of macrophage/monocyte-like cells (Fig. 6D,F) and a scarce extracellular matrix was surrounding the immunolabelled cells (Fig. 6F,G). In the alveolar bone sections, used as negative control, positive immunolabelling was not found (data not illustrated).
Fig. 5.

(A–F) Light micrographs of portions of oral mucosa of the eruptive pathway of first molars submitted to the immunohistochemistry for detection of MMP-9 (brown colour) and counterstained with haematoxylin. (5A) Fibroblasts (arrows) exhibit weak or negative MMP-9 immunolabelling in the lamina propria (LP) of a 9-old-day rat. MMP-9 immunolabelled osteoclasts (OC) are observed in the bone surface (B). OE, oral epithelium. Bar: 25 μm. 5B (outlined area of 5A) and 5C show, respectively, a fibroblast (arrows) and a mast cell (MC) strongly MMP-9 immunolabelled in the lamina propria (LP) of 9-day-old rats. Bar: 5 μm. (5D) Scarce cells with immunopositive cytoplasm (arrows) are observed in the lamina propria (LP) of 11-day-old rat. Multinucleated osteoclasts (OC) adjacent to bone surface (B) are positive; note a conspicuous immunostaining in the osteoclast cytoplasm (OC, inset). OE, oral epithelium. Bar: 30 μm; inset bar: 5 μm. In 5E (outlined area of 5D) and 5F, MMP-9-positive immunolabelling is observed in the fibroblasts (arrows) and mast cells (MC) of the lamina propria (LP) of 11-day-old rats. Bar: 5 μm.
Fig. 6.
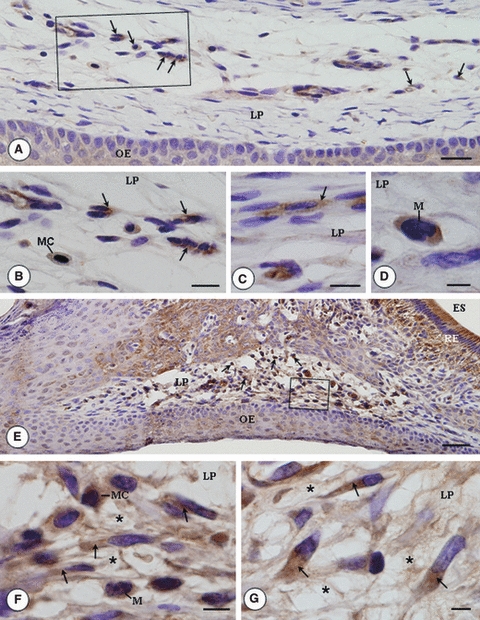
(A–G) Light micrographs of portions of oral mucosa of the eruptive pathway of first molars submitted to immunohistochemistry for detection of MMP-9 and counterstained with haematoxylin. (6A) Positive immunoreaction is observed in some cells (arrows) of the lamina propria (LP) of a 13-old-day rat. OE, oral epithelium. Bar: 25 μm. In 6B (outlined area of 6A), 6C and 6D, fibroblasts (arrows), mast cells (MC) and monocyte/macrophage-like cell (M) show cytoplasm strongly immunolabelled in the lamina propria (LP) of 13-day-old rats. (6B and 6C) Bar: 10 μm; (6D) bar: 5 μm. (6E) Strong positive immunoreaction is observed in numerous cells (arrows) of the lamina propria (LP) of a 16-day-old rat. OE, oral epithelium; RE, reduced enamel epithelium; ES, enamel space. Bar: 30 μm. In 6F (outlined area of 6E) and 6G, strong immunostaining is observed in the cytoplasm of fibroblasts (arrows), monocyte/macrophage-like cells (M) and mast cell (MC). Note that scarce components of extracellular matrix (asterisks) are observed in the lamina propria (LP). Bar: 5 μm.
Discussion
Morphological analysis revealed the presence of first upper molar germs in different phases of tooth eruption. In the 9- to 11-day-old rats, the tooth germs are in active process of eruption due to intense resorption of the bone covering the occlusal portion. This intense bone resorption may occur simultaneously to the movement of the tooth towards the oral cavity, i.e. the intra-osseous phase of tooth eruption (Gorski, 1992; Marks, 1995; Marks & Schroeder, 1996). Subsequently, in the 13- and 16-day-old rats, the bone that surrounded the occlusal portion of the molar germs was completely resorbed; the cusp tips of the first molars perforated the lamina propria, characterizing the mucosal penetration stage of the eruptive process (Marks, 1995; Marks & Schroeder, 1996).
It is known that the eruptive process depends on a coordinated and complex cascade of cellular and molecular events. There is strong evidence that the stellate reticulum and dental follicle produce several molecules required for eruption and, therefore, these structures seem to participate in the eruptive process (Gorski, 1992; Marks & Schroeder, 1996; Wise et al. 2002; Wise, 2009). In addition, an intense remodelling process in the lamina propria is required for the establishment of the eruptive pathway during tooth eruption. Our results showed that the lamina propria – interposed with tooth germ and oral epithelium – undergoes structural alterations. In the 9-day-old rats, the lamina propria contained few mast cells and occasional osteoclasts were found in the bone surface overlaying the occlusal portion of the tooth germs. Otherwise, in the 11-day-old rats, the number of mast cells in the lamina propria was significantly higher in comparison with 9-, 13- and 16-day-old rats and the number of TRAP-positive osteoclasts increased significantly from 9 to 11 days. During the intra-osseous phase of tooth eruption, the proportional increase in the number of mast cells in the lamina propria and osteoclasts indicates that mast cells could be involved in the bone resorption and, thereby, in the establishment of the eruptive pathway. Activated mast cells degranulate and thereby release several inflammatory mediators and growth factors, including histamine, cytokines such as tumour necrosis factor-α (TNF-α), vascular endothelial growth factor (VEGF) and various specific proteases (Artuc et al. 2002; Norrby, 2002; Naesse et al. 2003). There is evidence that mast cells may, in some circumstances, interfere with the regulation of bone resorption (Saffar & Klapisz-Wolikow, 1990; Dobigny & Saffar, 1991a,b; Lesclous et al. 2002, 2004; Fouilloux et al. 2006). The histamine, produced and released by mast cells, induces the production of IL-1, IL-6, macrophage colony-stimulation factor (M-CSF) and granulocyte macrophage colony-stimulating factor (GM-CSF) by haematopoietic cells (Mor et al. 1995) and stromal cells (Takamatsu & Nakano, 1998) via H2 receptors (Vannier & Dinarello, 1994; Mor et al. 1995; Jilka, 1998). These cytokines stimulate the osteoclast formation, differentiation and activity (Riggs, 2000; Phan et al. 2004). In addition, TNF-α also promotes osteoclast formation (Kobayashi et al. 2000) and VEGF upregulates the expression of receptor activator of nuclear factor kappa B (RANK) on osteoclast precursors (Yao et al. 2006). Considering that mast cell degranulation releases active factors for osteoclast differentiation and activity, the presence of numerous mast cells at the stage of intense bone resorption (intra-osseous stage) suggests a possible role of these cells in the recruitment of osteoclasts. This hypothesis is reinforced by the fact that the number of mast cells was reduced significantly after this stage and was maintained constant until the final stage of mucosal penetration (16 days old).
Studies have demonstrated the important role of MMPs in the degradation of the extracellular components during tissue maintenance and remodelling. Extracellular components such as collagen, anchoring collagen type VII, fibronectin and elastin are degraded by MMP-9 (Birkedal-Hansen et al. 1993; Tanaka et al. 1999). This enzyme has been detected in fibroblasts (Shimizu et al. 2005; Takahashi et al. 2006), mast cells (Fang et al. 1999; Tanaka et al. 1999) and macrophages (Ogata et al. 1992; Sternlicht & Werb, 2001; Norrby, 2002; Shimizu et al. 2005) of different tissues, indicating that this enzyme is secreted by these cells. Similar to these findings, our results revealed that MMP-9 is secreted by these same cell types in the lamina propria at different stages of tooth eruption. However, an enhanced immunolabelling was evident in the lamina propria of the advanced phase of tooth eruption (16-day-old rats). The mucosal penetration stage is characterized by fast speed of tooth eruption (Marks, 1995; Marks & Schroeder, 1996) and, therefore, the connective tissue of the eruptive pathway undergoes an intense process of degradation and/or remodelling. In fact, this stage was characterized by the presence of strongly MMP-9-immunolabelled cells surrounded by scarce collagen fibres and amorphous material, indicating that extracellular matrix was partially degraded. Thus, these findings indicate that MMP-9 participates in the process of degradation of the extracellular components of lamina propria during tooth eruption. Studies have demonstrated that the eruptive process is delayed in mice deficient in MT1-MMP, a membrane type 1-matrix metalloproteinase (Beertsen et al. 2002; Bartlett et al. 2003). Except for these few studies, the active participation of MMP-9 in the eruptive process has not yet been investigated. Thus, future studies are necessary to elucidate the actual role of MMP-9 in the eruptive process.
It has been demonstrated that mast cells are essential in collagen breakdown and play, therefore, an important role in tissue degradation and remodelling (Naesse et al. 2003; Steinsvoll et al. 2004). Mast cells produce MMP-1, MMP-2 and MMP-8 enzymes, which are responsible for degradation of extracellular matrix components of the gingival lamina propria (Naesse et al. 2003). In the present study, a high number of mast cells was observed in the 13- and 16-day-old rats. Moreover, these cells were also strongly MMP-9 immunostained in 16-day-old rats. We suggest that during the mucosal penetration stage of tooth eruption, mast cells participate in the degradation process of the extracellular matrix of the eruptive pathway.
Strong immunolabelling was also observed in osteoclasts located on the surface of the bone interposed with tooth germs and oral epithelium in the 9- and 11-day-old rats. MMP-9 in osteoclast is highly expressed and, therefore, it has been suggested that this MMP participates directly in the degradation of bone collagen (Reponen et al. 1994; Tezuka et al. 1994; Wucherpfennig et al. 1994; Okada et al. 1995; Ishibashi et al. 2006). Increased immunostaining for MMP-9 has been detected in osteoclasts during the development of different pathogenesis such as osteoclastomas and in Paget’s disease (Wucherpfennig et al. 1994). On the other hand, there is evidence from several studies that MMP-9 in osteoclasts seems to stimulate the recruitment of these cells, as inhibitors of MMP-9 suppress osteoclast migration (Blavier & Delaissé, 1995; Engsig et al. 2000; Xie et al. 2009).
In conclusion, our results indicate that MMP-9-positive mast cells are present in the lamina propria of the eruptive pathway in rat molars. The concomitant peak in the number of mast cells in the lamina propria and osteoclasts apposed to bone surface during the intra-osseous stage suggests a possible paracrine participation of mast cells in the process of bone resorption. The enhanced immunoexpression of MMP-9 in different cells of the lamina propria indicates that this metalloproteinase plays a role in the degradation of the extracellular components, mainly during the late phase of mucosal penetration. Investigation of the eruptive process in animals deficient in MMP-9 and/or mast cells would be useful to clarify the role of these components during tooth eruption.
Acknowledgments
The authors wish to thank Mr Luis Antônio Potenza and Mr Pedro Sérgio Simões for technical support. This research was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq – 307692/2006-0) and Fundação de Amparo à Pesquisa de São Paulo (FAPESP – 04/09898-0).
References
- Artuc M, Steckelings UM, Henz BM. Mast cell–fibroblast interactions: human mast cells as source and inducers of fibroblast and epithelial growth factors. J Invest Dermatol. 2002;118:391–395. doi: 10.1046/j.0022-202x.2001.01705.x. [DOI] [PubMed] [Google Scholar]
- Bartlett JD, Zhou Z, Skobe Z, et al. Delayed tooth eruption in membrane type-1 matrix metalloproteinase deficient mice. Connect Tissue Res. 2003;44:300–304. [PubMed] [Google Scholar]
- Beertsen W, Holmbeck K, Niehof A, et al. On the role of MT1-MMP, a matrix metalloproteinase essential to collagen remodeling, in murine molar eruption and root growth. Eur J Oral Sci. 2002;110:445–451. doi: 10.1034/j.1600-0722.2002.21384.x. [DOI] [PubMed] [Google Scholar]
- Birkedal-Hansen H, Moore WGI, Bodden MK, et al. Matrix metalloproteinases: a review. Crit Rev Oral Biol Med. 1993;4:197–250. doi: 10.1177/10454411930040020401. [DOI] [PubMed] [Google Scholar]
- Blavier L, Delaissé JM. Matrix metalloproteinases are obligatory for the migration of preosteoclasts to the developing marrow cavity of primitive long bones. J Cell Sci. 1995;108:3649–3659. doi: 10.1242/jcs.108.12.3649. [DOI] [PubMed] [Google Scholar]
- Cerri PS, Boabaid F, Katchburian E. Combined TUNEL and TRAP methods suggest that apoptotic bone cells are inside vacuoles of alveolar bone osteoclasts in young rats. J Periodontal Res. 2003;38:223–226. doi: 10.1034/j.1600-0765.2003.02006.x. [DOI] [PubMed] [Google Scholar]
- Cruzoé-Souza M, Sasso-Cerri E, Cerri PS. Immunohistochemical detection of estrogen receptor beta in alveolar bone cells of estradiol-treated female rats: possible direct action of estrogen on osteoclast life span. J Anat. 2009;215:673–681. doi: 10.1111/j.1469-7580.2009.01158.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dobigny C, Saffar JL. H1 and H2 histamine receptors modulate osteoclastic resorption by different pathways: evidence obtained by using receptor antagonists in a rat synchronized resorption model. J Cell Physiol. 1991a;173:10–18. doi: 10.1002/(SICI)1097-4652(199710)173:1<10::AID-JCP2>3.0.CO;2-M. [DOI] [PubMed] [Google Scholar]
- Dobigny C, Saffar JL. Partial inhibition of bone resorption by disodium cromoglycate in a synchronized model of bone remodelling. Agents Actions. 1991b;33:326–329. doi: 10.1007/BF01986581. [DOI] [PubMed] [Google Scholar]
- Engsig MT, Chen QJ, Vu TH, et al. Matrix metalloproteinase 9 and vascular endothelial growth factor are essential for osteoclast recruitment into developing long bones. J Cell Biol. 2000;151:879–889. doi: 10.1083/jcb.151.4.879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faloni APS, Sasso-Cerri E, Katchburian E, et al. Decrease in the number and apoptosis of alveolar bone osteoclasts in estrogen-treated rats. J Periodontal Res. 2007;42:193–201. doi: 10.1111/j.1600-0765.2006.00932.x. [DOI] [PubMed] [Google Scholar]
- Fang KC, Wolters PJ, Steinhoff M, et al. Mast cell expression of gelatinases A and B is regulated by kit ligand and TGF-β1. J Immunol. 1999;162:5528–5535. [PubMed] [Google Scholar]
- Fouilloux I, Duplan MB, Baroukh B, et al. Mast cell activation and degranulation occur early during induction of periosteal bone resorption. Bone. 2006;38:59–66. doi: 10.1016/j.bone.2005.07.026. [DOI] [PubMed] [Google Scholar]
- Gonçalves LDR, Oliveira G, Hurtado PA, et al. Expression of metalloproteinases and their tissue inhibitors in inflamed gingival biopsies. J Periodontal Res. 2008;43:570–577. doi: 10.1111/j.1600-0765.2008.01101.x. [DOI] [PubMed] [Google Scholar]
- Gorski JP. Current concepts of the biology of tooth eruption. Crit Rev Oral Biol Med. 1992;3:185–206. doi: 10.1177/10454411920030030201. [DOI] [PubMed] [Google Scholar]
- Ishibashi O, Niwa S, Kadoyama K, et al. MMP-9 antisense oligodeoxynucleotide exerts an inhibitory effect on osteoclastic bone resorption by suppressing cell migration. Life Sci. 2006;79:1657–1660. doi: 10.1016/j.lfs.2006.05.024. [DOI] [PubMed] [Google Scholar]
- Jilka RL. Cytokines, bone remodelling, and estrogen deficiency: a 1998 update. Bone. 1998;23:75–81. doi: 10.1016/s8756-3282(98)00077-5. [DOI] [PubMed] [Google Scholar]
- Kobayashi K, Takahashi N, Jimi E, et al. Tumor necrosis factor α stimulates osteoclast differentiation by a mechanism independent of the ODF/RANKL–RANK interaction. J Exp Med. 2000;191:275–285. doi: 10.1084/jem.191.2.275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lesclous P, Guez D, Saffar JL. Short-term prevention of osteoclastic resorption and osteopenia in ovariectomized rats treated with the H2 receptor antagonist cimetidine. Bone. 2002;30:131–136. doi: 10.1016/s8756-3282(01)00629-9. [DOI] [PubMed] [Google Scholar]
- Lesclous P, Guez D, Baroukh B, et al. Histamine participates in the early phase of trabecular bone loss in ovariectomized rats. Bone. 2004;34:91–99. doi: 10.1016/j.bone.2003.08.007. [DOI] [PubMed] [Google Scholar]
- Marks SC. The basic and applied biology of tooth eruption. Connect Tissue Res. 1995;32:149–157. doi: 10.3109/03008209509013718. [DOI] [PubMed] [Google Scholar]
- Marks SC, Schroeder HE. Tooth eruption: theories and facts. Anat Rec. 1996;245:3743–3793. doi: 10.1002/(SICI)1097-0185(199606)245:2<374::AID-AR18>3.0.CO;2-M. [DOI] [PubMed] [Google Scholar]
- Minkin C. Bone acid phosphatase: tartrate-resistant acid phosphatase as a marker of osteoclast function. Calcif Tissue Int. 1982;34:285–290. doi: 10.1007/BF02411252. [DOI] [PubMed] [Google Scholar]
- Mor S, Nagler A, Barak V, et al. Histamine enhances granulocyte-macrophage colony-stimulating factor and interleukin production by human peripheral blood mononuclear cells. J Leukoc Biol. 1995;58:445–450. doi: 10.1002/jlb.58.4.445. [DOI] [PubMed] [Google Scholar]
- Naesse EP, Schreurs O, Helgeland K, et al. Matrix metalloproteinases and their inhibitors in gingival mast cells in persons with and without human immunodeficiency virus infection. J Periodontal Res. 2003;38:575–582. doi: 10.1034/j.1600-0765.2003.00687.x. [DOI] [PubMed] [Google Scholar]
- Norrby K. Mast cells and angiogenesis. APMIS. 2002;110:355–371. doi: 10.1034/j.1600-0463.2002.100501.x. [DOI] [PubMed] [Google Scholar]
- Ogata Y, Enghild JJ, Negase H. Matrix metalloproteinase-3 (stromelysin) activates the precursor for the human matrix metalloproteinase-9. J Biol Chem. 1992;267:3581–3584. [PubMed] [Google Scholar]
- Okada Y, Naka K, Kawamura K, et al. Localization of matrix metalloproteinase 9 (92-kilodalton gelatinase/type IV collagenase – gelatinase B) in osteoclasts: implications for bone resorption. Lab Invest. 1995;72:311–322. [PubMed] [Google Scholar]
- Okaji M, Sakai H, Sakai E, et al. The regulation of bone resorption in tooth formation and eruption process in mouse alveolar crest devoid of cathepsin K. J Pharmacol Sci. 2003;91:285–294. doi: 10.1254/jphs.91.285. [DOI] [PubMed] [Google Scholar]
- Page-McCaw A, Ewald AJ, Werb Z. Matrix metalloproteinases and the regulation of tissue remodelling. Nat Rev Mol Cell Biol. 2007;8:221–233. doi: 10.1038/nrm2125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phan TC, Xu J, Zheng MH. Interaction between osteoblast and osteoclast: impact in bone disease. Histol Histopathol. 2004;19:1325–1344. doi: 10.14670/HH-19.1325. [DOI] [PubMed] [Google Scholar]
- Reponen P, Sahlberg C, Munaut C, et al. High expression of 92-kD type IV collagenase (gelatinase B) in the osteoclast lineage during mouse development. J Cell Biol. 1994;124:1091–1102. doi: 10.1083/jcb.124.6.1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riggs BL. The mechanisms of estrogen regulation of bone resorption. J Clin Invest. 2000;106:1203–1204. doi: 10.1172/JCI11468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saffar JL, Klapisz-Wolikow M. Changes in mast cell number during the activation phase of an induced synchronized remodelling sequence in the rat. Bone. 1990;11:369–372. doi: 10.1016/8756-3282(90)90093-e. [DOI] [PubMed] [Google Scholar]
- Sasso-Cerri E, Freymüller E, Miraglia SM. Testosterone-immunopositive primordial germ cells in the testis of the bullfrog, Rana catesbeiana. J Anat. 2005;206:519–523. doi: 10.1111/j.1469-7580.2005.00419.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimizu T, Kanai K, Asano K, et al. Suppression of matrix metalloproteinase production in nasal fibroblasts by tranilast, an antiallergic agent, in vitro. Mediators Inflamm. 2005;3:150–159. doi: 10.1155/MI.2005.150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinsvoll S, Helgeland K, Schenck K. Mast cells – a role in periodontal diseases? J Clin Periodontol. 2004;31:413–419. doi: 10.1111/j.1600-051X.2004.00516.x. [DOI] [PubMed] [Google Scholar]
- Sternlicht MD, Werb Z. How matrix metalloproteinases regulate cell behavior. Annu Rev Cell Dev Biol. 2001;17:463–516. doi: 10.1146/annurev.cellbio.17.1.463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi I, Onodera K, Nishimura M, et al. Expression of genes for gelatinases and tissue inhibitors of metalloproteinases in periodontal tissues during orthodontic tooth movement. J Mol Hist. 2006;37:333–342. doi: 10.1007/s10735-006-9060-7. [DOI] [PubMed] [Google Scholar]
- Takamatsu S, Nakano K. Regulation of interleukin-6, and macrophage colony-stimulating factor mRNA levels by histamine in stromal cell line (MC3T3-GP/PA6) Inflamm Res. 1998;47:221–226. doi: 10.1007/s000110050320. [DOI] [PubMed] [Google Scholar]
- Tanaka A, Arai K, Kitamura Y, et al. Matrix metalloproteinase-9 production, a newly identified function of mast cell progenitors, is downregulated by c-kit receptor activation. Blood. 1999;94:2390–2395. [PubMed] [Google Scholar]
- Tchougounova E, Lundequist A, Fajardo I, et al. A key role for mast cell chymase in the activation of pro-matrix metalloprotease-9 and pro-matrix metalloprotease-2. J Biol Chem. 2005;280:9291–9296. doi: 10.1074/jbc.M410396200. [DOI] [PubMed] [Google Scholar]
- Tezuka K-I, Nemoto K, Tezuka Y, et al. Identification of matrix metalloproteinase 9 in rabbit osteoclasts. J Biol Chem. 1994;269:15006–15009. [PubMed] [Google Scholar]
- Vannier E, Dinarello CA. Histamine enhances interleukin (IL)-1-induced IL-6 gene expression and protein synthesis via H2 receptors in peripheral blood mononuclear cells. J Biol Chem. 1994;269:9952–9956. [PubMed] [Google Scholar]
- Wise GE. Cellular and molecular basis of tooth eruption. Orthod Craniofac Res. 2009;12:67–73. doi: 10.1111/j.1601-6343.2009.01439.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wise GE, Fan W. Changes in the tartrate-resistant acid phosphatase cell population in dental follicle and bony crypts of rat molars during tooth eruption. J Dent Res. 1989;68:150–156. doi: 10.1177/00220345890680021001. [DOI] [PubMed] [Google Scholar]
- Wise GE, Marks SC, Jr, Cahill DR. Ultrastructural features of the dental follicle associated with formation of the tooth eruption pathway in the dog. J Oral Pathol. 1985;14:15–26. doi: 10.1111/j.1600-0714.1985.tb00461.x. [DOI] [PubMed] [Google Scholar]
- Wise GE, Frazier-Bowers S, D′Souza RN. Cellular, molecular, and genetic determinants of tooth eruption. Crit Rev Oral Biol Med. 2002;13:323–334. doi: 10.1177/154411130201300403. [DOI] [PubMed] [Google Scholar]
- Wucherpfennig AL, Li YP, Stetler-Stevenson WG, et al. Expression of 92 kD type IV collagenase/gelatinase B in human osteoclasts. J Bone Miner Res. 1994;9:549–556. doi: 10.1002/jbmr.5650090415. [DOI] [PubMed] [Google Scholar]
- Xie R, Kuijpers-Jagtman AM, Maltha JC. Osteoclast differentiation and recruitment during early stages of experimental tooth movement in rats. Eur J Oral Sci. 2009;117:43–50. doi: 10.1111/j.1600-0722.2008.00588.x. [DOI] [PubMed] [Google Scholar]
- Yao S, Liu D, Pan F, et al. Effect of vascular endothelial growth factor on RANK gene expression in osteoclast precursors and on osteoclastogenesis. Arch Oral Biol. 2006;51:596–602. doi: 10.1016/j.archoralbio.2005.12.006. [DOI] [PubMed] [Google Scholar]


