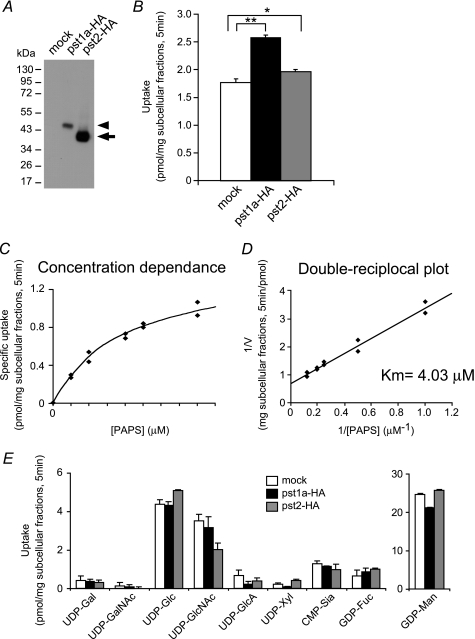
FIGURE 1.
Transport activities of PST-1a and PST-2 for PAPS and nucleotide sugars by yeast expression. A, Western blot analysis of the Golgi-rich subcellular fraction prepared from yeast cells expressing mock (left lane), HA-tagged PST-1a (center lane), and HA-tagged PST-2 (right lane) using anti-HA monoclonal antibody. The loaded amount of subcellular fraction proteins was 5 μg from the cells expressing mock or HA-tagged PST-1a and 0.5 μg from the cells expressing HA-tagged PST-2. The arrowhead and arrow indicate HA-tagged PST-1a and HA-tagged PST-2, respectively. B, PAPS uptake of PST-1a and PST-2. 200 μg of Golgi-rich subcellular fraction for each sample was incubated in 50 μl of reaction buffer containing 5 μm PAPS (mixture of 2 μm [35S]PAPS and 3 μm PAPS) at 25 °C for 5 min, and the incorporated radioactivity was measured. Values shown are the mean ± S.D. obtained from two independent experiments. Open bar, mock; solid bar, PST-1a-HA; gray bar, PST-2-HA. *, p < 0.05; **, p < 0.001; assessed by two-tailed Student's t test. C, substrate concentration dependence of PST-1a. 200 μg of Golgi-rich subcellular fraction for each sample was incubated in 50 μl of reaction buffer containing different concentrations of [35S]PAPS at 25 °C for 5 min, and the incorporated radioactivity was measured. Specific uptake of PST-1a was calculated by subtracting the value of the mock uptake from the values of PST-1a-HA uptake. D, double-reciprocal plot used to determine the Km value of PST-1a. E, nucleotide sugar uptake of PST-1a and PST-2. 100 μg of Golgi-rich subcellular fraction for each sample was incubated in 50 μl of reaction buffer containing 2 μm various nucleotide sugars at 25 °C for 5 min, and the incorporated radioactivity was measured. Values shown are the mean ± S.D. obtained from two independent experiments. Open bar, mock; solid bar, PST-1a-HA; gray bar, PST-2-HA.