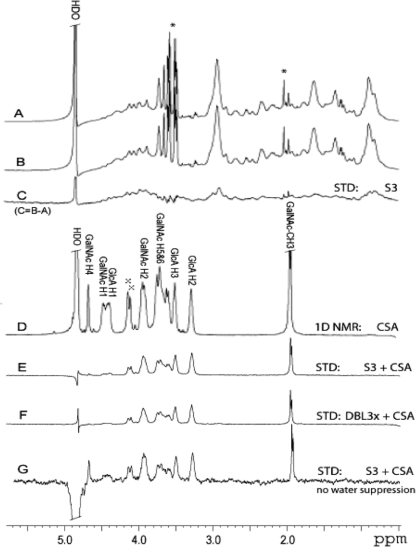
FIGURE 4.
S3 and DBL3x binding to CSA oligosaccharide observed by STD NMR. A–C, STD spectrum (C) of 60 μm S3 protein without CSA oligosaccharide at 20 °C, 1024 scans, obtained by subtraction (C = B − A) of the on- (A) from the off- (B) saturation spectra. Present in solution are some low molecular weight contaminants (asterisk), but note that their signals are completely cancelled in the difference spectrum in C. D, 1D 1H NMR spectrum of CSA oligosaccharide (20 °C, 16 scans, no water suppression). Residual signals from chondroitin-6-O-sulfate are labeled with x. Due to overlap, only selected resonances are labeled. E, STD spectrum of 1:100 molar ratio of S3 and CSA oligosaccharide. F, STD spectrum of 1:100 molar ratio of DBL3x and CSA oligosaccharide at 20 °C using 1024 scans each. G, same as E but the STD data were collected without water suppression at 10 °C to observe the STD signals for the GalNAc-H4, GalNAc-H1, and GlcA-H1 resonances that were obscured by water suppression in E. HDO, residual partially deuterated water.