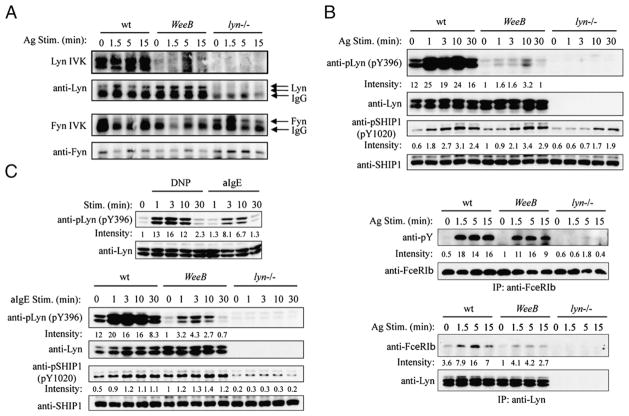
FIGURE 4.
Lyn activity is dramatically reduced in WeeB BMMCs, but phosphorylation levels of Lyn targets are only moderately impaired or normal. A, In vitro kinase assays for Lyn and Fyn were carried out after immunoprecipitation from cell lysates using the appropriate antisera. Equal loading was subsequently confirmed by immunoblotting using either the same blot or a duplicate blot derived from a portion of the immunoprecipitates. B, BMMCs were sensitized with IgE overnight and stimulated with 100 ng/ml DNP21-BSA for the indicated periods. Cell lysates were analyzed by SDS-PAGE followed by immunoblotting with the indicated antibodies. Immunoprecipitates of the FcεRIβ subunit and of Lyn were analyzed by immunoblotting with anti-phosphotyrosine (4G10) and anti-FcεRIβ, respectively, followed by reprobing of the blots with anti-FcεRIβ and anti-Lyn, respectively. C, Analysis of phospho-Lyn and phospho-SHIP1 after stimulation with 20 μg/ml anti-IgE (bottom panel) as described in A. Immunoblotting results are representative of three independent experiments, and kinase assays are representative of two independent experiments.