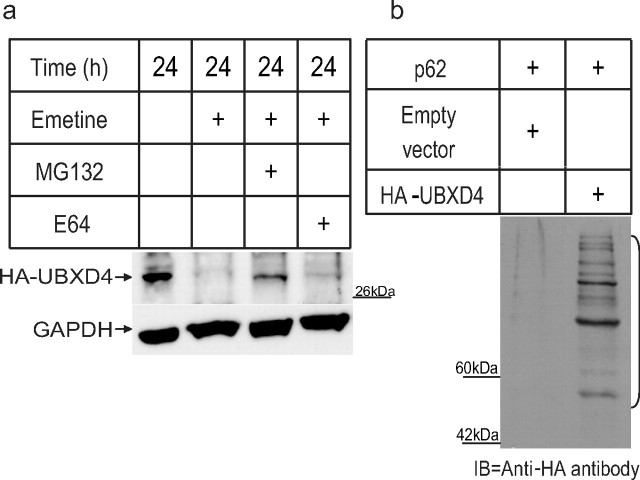
Figure 6.
UBXD4 is ubiquitinated and degraded by the 26S proteasome. a, A 24 h exposure to the proteasome blocker MG132 in the presence of the protein synthesis blocker, emetine, decreased the rate of degradation of UBXD4 in HEK293 cells transiently transfected with HA-tagged UBXD4. Exposure to the lysosome inhibitor E64 did not alter the degradation rate of UBXD4. b, A ladder of ubiquitinated, HA-tagged UBXD4 could be pulled down with p62 from α3β2 expressing HEK293 cells transiently transfected with HA-tagged UBXD4, but not with the HA-empty vector. The bracket on the right marks a ladder of bands corresponding to polyubiquitinated UBXD4. The above experiments were conducted twice with similar results each time.