Figure 2.
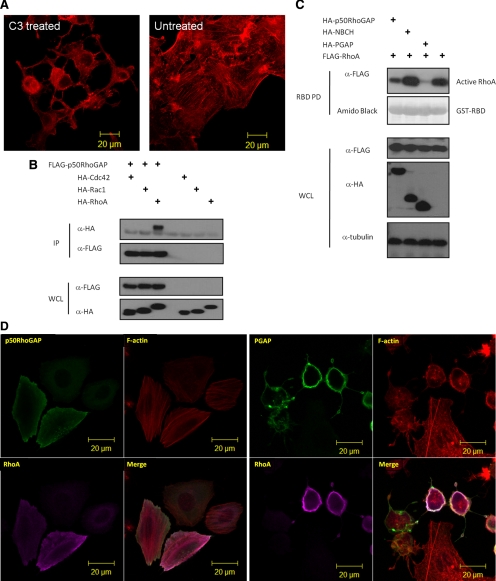
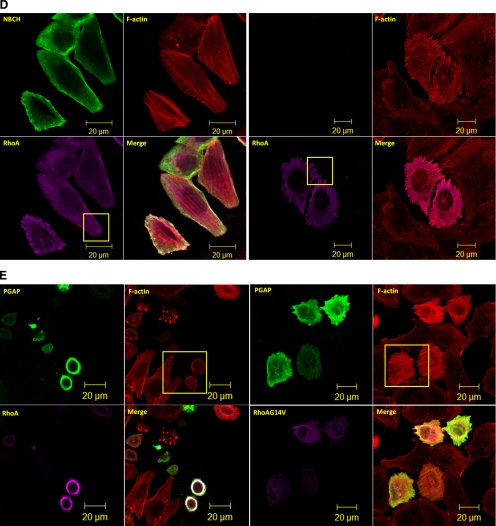
The GAP domain of p50RhoGAP induces cell rounding by inactivating Rho GTPases (A) HeLa cells were treated with Rho inhibitor C3 Transferase followed by rhodamine-conjugated phalloidin staining and confocal fluorescence microscopy analysis. (B) Cells were transfected with FLAG-p50RhoGAP in the presence or absence of HA-tagged expression constructs of Cdc42, Rac1, and RhoA. Lysates were immunoprecipitated (IP) with anti-FLAG beads, and the associated proteins were separated on SDS-PAGE, blotted, and probed with HA antibody. Expression of FLAG-p50RhoGAP and HA-tagged Cdc42, Rac1, and RhoA were verified by Western blot analyses for the whole cell lysates (WCL) using anti-FLAG (third panel) and anti-HA (bottom panel), respectively. The bound GTPase was detected by anti-HA (top panel), and equal loading of IP beads were verified by anti-FLAG (second panel). (C) To determine the Rho GTPase activity, HeLa cells were transfected with FLAG-tagged wild-type RhoA in the presence and absence of HA-tagged p50RhoGAP, NBCH, or PGAP mutants. Cell were lysed and incubated with GST fusion of the Rho-binding domain of rhotekin immobilized on beads as described in Materials and Methods Bound active RhoA were resolved on SDS-PAGE and detected by immunoblotting with FLAG-antibody (top panel). Equal loading of GST fusion proteins is shown in the second panel. (D) HeLa cells were transfected with plasmids encoding HA-RhoA alone or with FLAG-tagged full-length p50RhoGAP, PGAP, or NBCH mutants. Cells were then fixed after 20 h and subjected to confocal fluorescence microscopy as described in Materials and Methods The actin filaments were detected by direct costaining with rhodamine-conjugated phalloidin. (E) HeLa cells were cotransfected with HA-tagged PGAP mutant and wild-type or constitutively active RhoA-G14V. Cells were then fixed and images analyzed by confocal fluorescence microscopy after direct staining with rhodamine-conjugated phalloidin for actin filaments.