Abstract
The efficient and reproducible generation of differentiated progenitors from pluripotent stem cells requires the recapitulation of appropriate developmental stages and pathways. Here, we have used the combination of activin A, BMP4 and VEGF under serum-free conditions to induce hematopoietic differentiation from both embryonic and induced pluripotent stem cells, with the aim of modeling the primary sites of embryonic hematopoiesis. We identified two distinct Flk1-positive hematopoietic populations that can be isolated based on temporal patterns of emergence. The earliest arising population displays characteristics of yolk sac hematopoiesis, whereas a late developing Flk1-positive population appears to reflect the para-aortic splanchnopleura hematopoietic program, as it has reduced primitive erythroid capacity and substantially enhanced myeloid and lymphoid potential compared with the earlier wave. These differences between the two populations are accompanied by differences in the expression of Sox17 and Hoxb4, as well as in the cell surface markers AA4.1 and CD41. Together, these findings support the interpretation that the two populations are representative of the early sites of mammalian hematopoiesis.
Keywords: AGM, Hematopoiesis, Induced pluripotent stem cell, Embryonic stem cells, Sox17, Yolk sac, Mouse
INTRODUCTION
The capacity of embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs) to generate a broad range of differentiated progeny in culture provides a powerful model system for studying mammalian development as well as a novel source of cells for therapeutic applications (Murry and Keller, 2008; Rubin, 2008; Irion et al., 2009). Within this context, the generation of hematopoietic stem cells (HSCs) from pluripotent stem cells (PSCs) is of particular interest as the derivation of these cells in the differentiation cultures would provide a novel source of stem cells for transplantation and for modeling hematological diseases in vitro (McKinney-Freeman and Daley, 2007). Hematopoietic development is one of the best-studied differentiation programs in ESC cultures, as the blood cell lineages arise early even in poorly defined serum-based cultures (Keller, 2005). However, efficient development of transplantable HSCs from ESCs remains a challenge and this difficulty might in part reflect our inability to recapitulate the key developmental steps that establish this population in the embryo.
The embryonic hematopoietic system consists of distinct programs that develop at different sites and times during embryogenesis. The yolk sac (YS), which is the first site of hematopoiesis in the mammalian embryo, generates an early restricted hematopoietic program that is characterized by the transient development of a unique population of primitive erythrocytes, as well as macrophages and definitive erythroid, megakaryocyte and mast cell progenitors (Palis et al., 1999; Russell, 1979). Prior to the onset of circulation, the YS displays minimal lymphoid and HSC potential (Cumano et al., 1996; Cumano et al., 2001). This restriction appears to be stage specific, however, as the YS of older embryos does contain lymphoid progenitors and HSCs (Cumano et al., 1996; Yoder et al., 1997; Samokhvalov et al., 2007).
Following the onset of the YS program, hematopoiesis is initiated in a region known as the para-aortic splanchnopleura (P-Sp), which later contains the developing aorta, gonads and mesonephros (AGM) (Dieterlen-Lievre, 1975; Godin et al., 1993; Medvinsky et al., 1993). Recent studies indicate that P-Sp hematopoiesis is initiated through the direct transition of hemogenic endothelium to hematopoietic progenitors (Bertrand et al., 2010; Boisset et al., 2010; Kissa and Herbomel, 2010). The P-Sp-derived hematopoietic program is distinct from that of the early YS in that it gives rise to HSCs and lymphoid progenitors and is devoid of primitive erythroid potential (Cumano et al., 1996; Cumano et al., 2001; Medvinsky et al., 1996). These differences in developmental potential are associated with differential expression patterns of specific genes. In situ hybridization studies have shown that the cell surface marker AA4.1 (CD93 – Mouse Genome Informatics) is expressed in the P-Sp region but not in the YS prior to the onset of circulation (E7-8.5) (Petrenko et al., 1999). AA4.1 is found on the majority of P-Sp-derived HSCs and multipotential progenitors, whereas it is present only on a subset of myeloid/erythroid-restricted YS progenitors (Bertrand et al., 2005). At later stages of development, AA4.1 is found on lymphoid progenitors throughout the embryo (Yamane et al., 2009). Dependence on the transcription factor Sox17 may also distinguish YS and P-Sp hematopoiesis as Sox17-null animals contain some erythroid and myeloid progenitors but do not generate HSCs (Kim et al., 2007).
The onset of hematopoiesis in mouse ESC differentiation cultures appears to recapitulate early YS hematopoiesis, as this stage is restricted in potential and characterized by the emergence of a transient wave of primitive erythropoiesis (Keller, 2005). Although lymphoid progenitors have not been identified in this early phase of ESC-derived hematopoiesis, they have been detected in later-stage stromal-based cultures, suggesting that a P-Sp-like program is established under these conditions (Cho et al., 2001; Cho et al., 1999; Nakano et al., 1994; Schmitt and Zuniga-Pflucker, 2002; Tabatabaei-Zavareh et al., 2007; Yamane et al., 2009). The notion that P-Sp hematopoiesis can be generated from ESCs is further supported by observations that cells with properties of HSCs can be generated in ESC differentiation cultures following the enforced expression of either Hoxb4 or the combination of Cdx4 and Hoxb4 (Kyba et al., 2002; Wang et al., 2005b).
To be able to generate HSCs from ESCs, it is necessary to develop approaches that enable the specification and identification of P-Sp-like populations in the differentiation cultures. We have previously shown that temporal aspects of mesodermal specification observed in the mouse embryo are faithfully recapitulated in ESC cultures, enabling the isolation of hematopoietic and cardiovascular progenitors (Fehling et al., 2003; Kattman et al., 2006). Using a similar strategy in this study, we mapped hematopoietic development over time in differentiation cultures and identified distinct Flk1-positive (Flk1pos) hematopoietic populations (Flk1 is also known as Kdr – Mouse Genome Informatics) that display characteristics of YS and P-Sp hematopoiesis.
MATERIALS AND METHODS
ESC maintenance and differentiation
The T-EGFP/Rosa26-tdRFP (RFP.bry) ESCs were generated by targeting the tdRFP cDNA to the Rosa26 locus (Luche et al., 2007). RFP.bry, Sox17-EGFP mESCs (Kim et al., 2007) and the iPSC lines Sox2-EGFP and Oct4-EGFP (Stadtfeld et al., 2008) were cultured in serum-free media (Gadue et al., 2006). For differentiation, ESCs were dissociated and cultured in suspension in serum-free differentiation (SF-D) media without additional growth factors for 48 hours. Embryoid bodies (EBs) were then dissociated and reaggregated in SF-D with the addition of various growth factors or inhibitors as indicated. In most experiments, the EBs were harvested 30-32 hours later, the cells dissociated and the appropriate populations isolated by cell sorting. For reaggregation, sorted cells were either cultured at 250,000 cells/ml in 24-well ULA dishes (Costar) or at 30,000 cells/100 μl in 96-well ULA dishes. Human activin A, BMP4 and VEGF were purchased from R&D Systems; SB-431542 was obtained from Sigma.
Quantitative real-time PCR
Total RNA was prepared with the RNeasy Mini or Micro Kits (Qiagen) and treated with DNase (Qiagen). RNA (0.1-1 μg) was reverse transcribed using random hexamers and oligo(dT) with Superscript III reverse transcriptase (Invitrogen). Real-time quantitative (q) PCR was performed on a MasterCycler RealPlex (Eppendorf) using SYBR Green JumpStart ReadyMix (Sigma). The oligonucleotide sequences are listed in Table S2 in the supplementary material (all oligonucleotides from IDT). Genomic DNA standards were used to evaluate the efficiency of the PCR and calculate the copy number of each gene relative to the housekeeping gene Actb.
Flow cytometry and cell sorting
EBs generated from the ESC differentiations were stained using the following monoclonal antibodies from Pharmingen: CD41 (Itga2b) (1B5, hybridoma), Flk1 (89B3) (Kabrun et al., 1997), c-Kit (CD117), Sca1 (Ly6a) and AA4.1 (493). OP9 co-cultures were trypsinized and incubated with 2.4G2 monoclonal antibody to block FcγRIII/II and stained for CD45 (Ptprc), CD19, TCRβ, CD8, CD4, CD11b (Itgam) and Gr1 (Ly6g). All antibody stains were performed at 4°C in PBS containing 2% (v/v) FCS and in some cases also 0.02% (w/v) NaN3. The cells were acquired using a LSR II flow cytometer with HTS sampler option (Becton Dickinson) or sorted using a FACS ARIA II (Becton Dickinson). Analysis was performed using FlowJo (Tree Star).
Isolation of Flk1pos cells from mouse embryos
Timed pregnant Swiss Webster female mice were purchased from The Jackson Laboratory. YS and P-Sp/AGM regions were dissected and single-cell suspensions were stained with anti-Flk1 monoclonal antibody. The cells were sorted using a FACS ARIA II and mRNA was isolated and converted to cDNA as described above.
Hematopoietic progenitor assay
Hematopoietic precursors were plated in 1% (w/v) methylcellulose (Fluka) containing 10% (v/v) plasma-derived serum (PDS; Antech), 5% (v/v) protein-free hybridoma medium (PFHM-II; Invitrogen) and the following cytokines: kit ligand (KL) [1% (v/v) conditioned medium], thrombopoietin (TPO) [1% (v/v) conditioned medium], erythropoietin (2 U/ml), IL11 (25 ng/ml), IL3 [1% (v/v) conditioned medium], GM-CSF (Csf2) [1% (v/v) conditioned medium] and IL6 (5 ng/ml). Cultures were maintained at 37°C, 5% CO2. KL was derived from conditioned media (Genetics Institute). IL3 was from X63 AG8-653 myeloma cells transfected with a vector expressing IL3. Human VEGF, mouse IL6 and human IL11 were purchased from R&D Systems. Mean and standard errors of six independent experiments were calculated. Student's t-test was used to evaluate statistical significance.
OP9 co-culture
Irradiated (30 gray) OP9-EGFP or EGFP-DL1 cells were seeded 1 day prior to use on 0.1% (v/v) gelatin in 24- or 96-well tissue culture treated plates (Falcon). ESC-derived progenitors were then plated in αMEM (Invitrogen) with 20% (v/v) FCS containing 10 ng/ml mouse Flt3l, 10 ng/ml mouse IL7 (R&D Systems) and 2% (v/v) KL supernatant for the first week. Medium was changed every 5 to 7 days. KL was removed after 7 days. Tcrb locus rearrangement on genomic DNA was analyzed using the primers shown in Table S2 in the supplementary material.
RESULTS
Temporal development of hematopoietic progenitor populations
To recapitulate normal hematopoietic development in ESC/embryoid body (ES/EB) cell cultures, it is important to use agonists of signaling pathways that are known to regulate hematopoietic commitment in the early embryo. For this purpose, we focused on the nodal/TGFβ, BMP4 and VEGF pathways as they are known to play a role at different stages of mesoderm induction and hematopoietic specification in vivo (Conlon et al., 1994; Liu et al., 1999; Winnier et al., 1995) and have been shown to function in a similar capacity in vitro (Lengerke et al., 2007; Ng et al., 2005; Nostro et al., 2008). Using an ESC line carrying the enhanced green fluorescent protein cDNA targeted to the brachyury (T) gene [T-EGFP (Fehling et al., 2003)], we evaluated both T-EGFP and Flk1 expression over time to monitor mesoderm induction and specification. When cultured in serum-free medium in the absence of factors, neither T-EGFP nor Flk1 was induced in the EBs (Fig. 1A). By contrast, when differentiated in the presence of a combination of activin A, BMP4 and VEGF (AVB), greater than 80% (80.7±5.5, n=7) of the resulting EB cells expressed T-EGFP and of these typically more than 15% (15.8±2.9, n=7) co-expressed Flk1. The dramatic expansion of these populations to greater than 80% T-EGFP-positive and 15% Flk1-positive within 6-12 hours of their induction demonstrates the dynamic nature of differentiation in these cultures and highlights the importance of establishing detailed temporal profiles for the isolation of specific lineages.
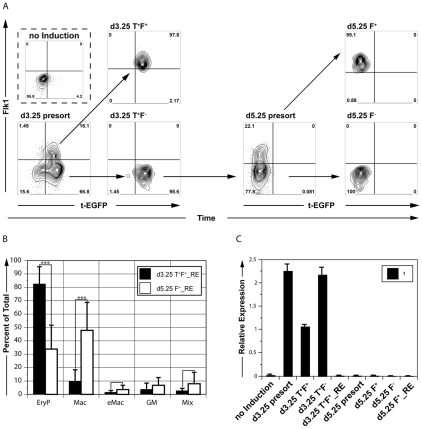
Fig. 1.
Induction of two distinct Flk1pos hematopoietic precursor populations from mouse ESCs using activin A, BMP4 and VEGF. (A) To the left are shown T-EGFP and Flk1 expression in day (d) 3.25 embryoid bodies (EBs) cultured in the absence (no induction) or presence (d3.25 presort) of inducers. The d3.25 population was segregated into d3.25 T+ F+ and d3.25 T+ F– fractions by cell sorting (T, brachyury; F, Flk1). The d3.25 T+ F– cells were cultured as aggregates for 48 hours and the d5.25 F+ and F– fractions isolated (shown to the right). (B) Hematopoietic progenitor potential of the d3.25 T+ F+ and d5.25 F+ populations aggregated for 24 hours and cultured in methylcellulose cultures. Primitive erythroid colonies (EryP) were scored at day 5 of culture, whereas the macrophage (Mac), erythroid-macrophage (eMac), granulocyte-macrophage (GM) and mixed (Mix) colonies were counted at days 7-8. Data are expressed as the mean of six independent experiments; error bars indicate ±s.d. ***, P<0.0002. (C) qRT-PCR analysis of T expression in all populations shown in A. d3.25 T+ F+_RE indicates d3.25 T+ F+ cells reaggregated for 24 hours; d5.25 T+ F+_RE indicates d5.25 F+ cells reaggregated for 24 hours. Data are presented as expression relative to that of the housekeeping gene Actb and are the mean of three independent experiments; error bars indicate ±s.d.
The earliest Flk1pos population that develops in serum-induced EBs displays predominantly primitive erythroid potential (Fehling et al., 2003; Keller, 2005). To assess the hematopoietic potential of the AVB-induced Flk1pos population, the cells were isolated from day (d) 3.25 EBs by flow cytometry, allowed to aggregate and then cultured for an additional 24 hours in serum-free medium in the presence of kit ligand [KL; also known as stem cell factor (SCF)] and VEGF. The majority of hematopoietic progenitors in these aggregates were of the primitive erythroid lineage (82%), demonstrating that the potential of the d3.25 Flk1pos population induced under serum-free conditions was similar to that of the serum-induced population and reflective of YS hematopoiesis (Fig. 1B) (Fehling et al., 2003). Low numbers (<10% of total) of macrophage, bi-potential erythroid-macrophage and multipotential progenitors were also detected. Taken together, these findings indicate that the AVB combination efficiently induces hematopoietic development in mouse ESC differentiation cultures.
If hematopoietic development in the ESC model recapitulates the establishment of the hematopoietic system in the early embryo, we reasoned that a mesoderm-derived population equivalent to the P-Sp should develop shortly following the appearance of the YS program (Parameswaran and Tam, 1995). To identify a later-developing hematopoietic program, we focused on the d3.25 Flk1-negative (Flk1neg) population and cultured these cells for 48 hours in the presence of AVB. These d3.25 T-EGFP+ Flk1neg cells did give rise to a substantial (24.8±5.8% of all cells, n=6) second Flk1pos population (d5.25 presort; Fig. 1A). Expression of T, evaluated by flow cytometry for EGFP or by qRT-PCR (Fig. 1A,C), declined during this culture period, demonstrating that this population represented a developmental stage beyond mesoderm.
When isolated and cultured in the presence of VEGF and KL for 24 hours, the d5.25 Flk1pos population displayed hematopoietic potential that differed from that of the early-developing Flk1pos population. In contrast to the d3.25 T-EGFP+ Flk1pos cells, the d5.25 Flk1pos population generated significantly fewer primitive erythroid progenitors (33.8% versus 82.5% of all colonies, respectively), a higher percentage of macrophage progenitors (47.8% versus 9.8%), and more bi-potential erythroid-macrophage (3.5% versus 1.5%) and multipotential (8% versus 2.6%) progenitors (Fig. 1B). The d5.25 Flk1neg population contained few hematopoietic progenitors (data not shown). The fact that the percentage of Flk1pos cells (d3.25, 15%; d5.25, 25%) and the frequency of progenitors were similar at both time points suggests that these changes are not simply reflective of a decline in primitive erythropoiesis over time (total colony number: d3.25, 409±197; d5.25, 442±98; P=0.6, n=5, expressed as number of colonies per 10,000 input cells; see Fig. S2 in the supplementary material). Rather, they support the interpretation that the two Flk1pos populations represent the temporal development of distinct hematopoietic progenitors in AVB-induced mouse ESC-derived EBs.
Gene expression analysis of sorted populations
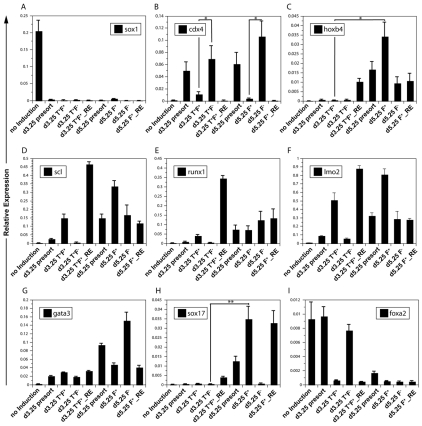
To further characterize these two hematopoietic progenitor populations, freshly isolated Flk1pos and Flk1neg cells, as well as populations derived from them were subjected to qRT-PCR gene expression analyses. These analyses included the following populations: (1) d3.25 non-induced EBs; (2) the d3.25 induced EBs (presort); (3) the T+ Flk1pos (T+ F+) fraction from d3.25 EBs; (4) the T+ Flk1neg (T+ F–) fraction from d3.25 EBs; (5) the d3.25 T+ Flk1pos cells following 24 hours of aggregation culture (d3.25 T+ F+_RE; RE refers to reaggregated); (6) the presorted d5.25 population generated from the T+ Flk1neg cells; (7,8) the Flk1pos and Flk1neg fractions isolated from the d5.25 cells (d5.25 F+ and d5.25 F–, respectively); and (9) the population derived from the d5.25 Flk1pos cells following 24 hours of aggregation culture (d5.25 F+_RE). These different populations were analyzed for expression of genes associated with hematopoietic induction [Cdx4, Hoxb4 (Davidson et al., 2003)] and hematopoietic commitment [Scl (Tal1 – Mouse Genome Informatics), Runx1, Lmo2, Gata3 (Robb et al., 1996; Wang et al., 1996)]. We also included genes indicative of neuroectoderm (Sox1) and definitive endoderm (Foxa2) development to monitor the efficiency of primitive streak (PS)/mesoderm induction. Sox17 was evaluated as it has been shown to be expressed in fetal liver HSCs in addition to definitive endoderm (Kim et al., 2007).
Expression of Sox1 was detected only in EBs cultured in the absence of PS inducers, indicating differentiation to the neuroectoderm lineage (Fig. 2A). In the induced EBs, Cdx4, a member of the caudal-related homeobox gene family, displayed a striking pattern, as its expression appeared to be preferentially restricted to the Flk1neg fractions from both time points (Fig. 2B, fractions 4 and 8). Expression of Hoxb4 was detected at highest levels in the d5.25 Flk1pos fraction generated from the d3.25 T+ F– Cdx4+ population. Hoxb4 expression appeared to distinguish the two Flk1pos populations, as it was found in the second but not the first population (Fig. 2C). Genes essential for hematopoietic development in the embryo, including Scl, Runx1 and Lmo2, were broadly expressed in all Flk1pos cells or derivatives thereof, a pattern consistent with their role in both YS and P-Sp/AGM hematopoietic commitment (Fig. 2D-F). Gata3, a gene required for T lymphocyte and kidney development (Pandolfi et al., 1995), also displayed a relatively broad expression pattern (Fig. 2G). The expression pattern of Sox17 was one of the most restricted as it was detected only in the d5.25 Flk1pos cells and the population derived from them (Fig. 2H, fractions 7 and 9). As with Hoxb4, expression of Sox17 also distinguished the two Flk1pos populations. Expression of Sox17 was not reflective of the emergence of definitive endoderm, as these populations did not express the pan-endoderm marker Foxa2 (Fig. 2I). Taken together, these expression studies document molecular differences between the two Flk1pos hematopoietic populations consistent with the interpretation that they are representative of distinct hematopoietic programs.
Fig. 2.
Gene expression analyses of different progenitor populations. (A-I) The sorted cell populations isolated in Fig. 1 were analyzed for the expression of the indicated genes by qRT-PCR. Data are presented as expression relative to that of Actb and are the mean of three independent experiments; error bars indicate ±s.d. *, P<0.2; **, P<0.01.
Flow cytometric analysis of the Flk1-derived populations
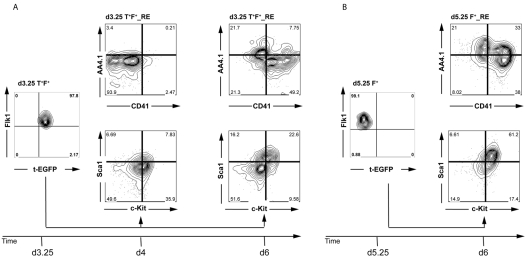
We next analyzed the two Flk1pos populations and cells derived from them for expression of AA4.1, CD41, c-Kit and Sca1, as combinations of these markers can distinguish early YS- and P-Sp/AGM-derived hematopoiesis (Bertrand et al., 2005; de Bruijn et al., 2002). The analyses included d3.25- and d5.25-derived populations cultured for 24 hours, as well as a d3.25 population cultured for 3 days as a control for the total culture period of the d5.25-derived cells. Cells from the d3.25 fraction cultured for 1 day did not express CD41 or AA4.1 (Fig. 3A). More than 35% of the population expressed c-Kit, but less than 8% of the cells co-expressed Sca1. By contrast, a substantial portion of the d5.25 Flk1pos-derived population expressed either CD41 (38%) or AA4.1 (21%) or both (>30%) (Fig. 3B). The majority (>60%) of these cells also co-expressed Sca1 and c-Kit. The differences observed between the two Flk1pos populations were not simply due to time in culture, as the patterns observed on the d3.25 T+ F+-derived population cultured for 3 days differed from those found on the d5.25-derived population (Fig. 3A). The most notable differences were the smaller size of the subpopulations that co-express AA4.1/CD41 and Sca1/c-Kit in the d3.25-derived population. In summary, cell surface marker analysis of these fractions suggests an enrichment of P-Sp/AGM-like hematopoiesis in the d5.25 Flk1pos fraction.
Fig. 3.
Phenotypic analyses of the populations derived from the two Flk1pos populations. (A) Analysis of d3.25 Flk1pos-derived populations cultured for either 1 day in the presence of KL and VEGF (d4) or for 2 days in activin, BMP4 and VEGF and then for 1 day in kit ligand (KL) and VEGF (d6). (B) Analysis of d5.25 Flk1pos-derived populations cultured for 1 day in the presence of KL and VEGF. Data are representative of two independent experiments and gates were set on unstained controls.
Lymphoid potential of the Flk1-derived populations
As a final evaluation of the two Flk1pos populations, we compared their capacity to generate cells of the lymphoid lineages (Cho et al., 1999; Nakano et al., 1994; Schmitt and Zuniga-Pflucker, 2002; Schmitt et al., 2004). Within 3 weeks of culture, B cell and T cell development was observed on the OP9 and OP9-DL1 stromal cells, respectively (see Fig. S3A,B in the supplementary material).
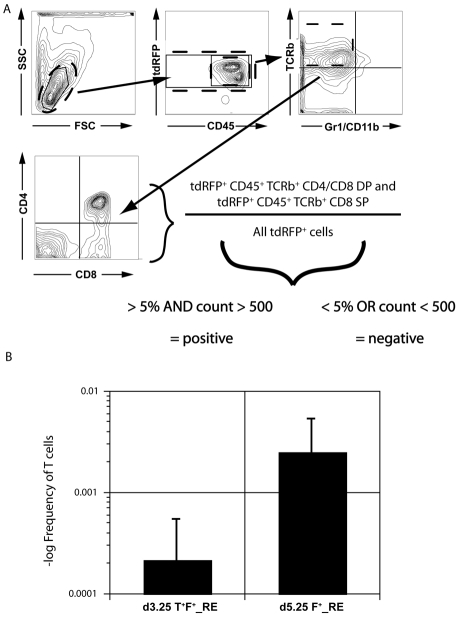
To better quantify the lymphoid potential of the two Flk1pos populations, we carried out a limiting dilution study to estimate the frequency of T lymphoid progenitors in these groups. For these analyses, d3.25 T+ F+_RE or d5.25 F+_RE cells generated from a T-EGFP cell line engineered to express the red fluorescent protein (tdRFP) from the Rosa26 locus (Luche et al., 2007) were seeded at different densities on OP9-DL1 stromal cells in microtiter plates. Cultures were harvested at 3 weeks and analyzed by flow cytometry. The frequency of wells containing T cell progenitors was calculated from the ratio of tdRFP+ T cells (defined by CD45+ TCRβ+ CD4+ CD8+ or CD45+ TCRβ+ CD8+) to the total number of tdRFP+ cells (Fig. 4A). Analyses from six different experiments revealed that the average frequency of T cell progenitors in the second Flk1pos population was approximately 1 in 400 cells (Fig. 4B). The frequency of T cell progenitors in the population derived from the first Flk1pos fraction was approximately 1 in 4600 (14.2±6.5-fold lower). This difference was not due to total induction time in culture, as the d3.25-derived population cultured for 3 days prior to the OP9-Dl1 co-culture (under the same conditions as their d3.25 T+ F– counterpart) showed a substantially lower frequency of T cell progenitors (approximately 1 in 12,000; n=2; data not shown), despite increased CD41 and AA4.1 expression (Fig. 3A). The findings from this lymphoid progenitor frequency analysis add further support to the interpretation that the d5.25 population represents the equivalent of the P-Sp hematopoietic program.
Fig. 4.
T cell potential of the Flk1pos progenitors. (A,B) d3.25 T+ F+_RE or d5.25 F+_RE cells generated from a T-EGFP ESC line expressing tdRFP were seeded over a range of densities on OP9-DL1 stroma cells and cultured for 3 weeks. Media were changed every week. Following culture, the cells were harvested, stained and (A) analyzed for expression of CD45, TCRβ, CD4 and CD8 by flow cytometry and (B) the frequency of T cell progenitors in the two populations (1/4680 for d3.25 versus 1/401 for d5.25) determined. Positive wells were those that contained at least 500 tdRFP+ cells (as a measure of total deposition), of which more than 5% expressed T cell markers as defined by the expression of both CD45+ and TCRβ+ and either CD8 alone or both CD4 and CD8. The frequency of T cell progenitors was then determined using L-Calc software (Stem Cell Technologies). Data are presented as the frequency of T cell progenitors and graphed on a logarithmic scale (n=6).
Sox17 expression distinguishes Flk1pos subpopulations
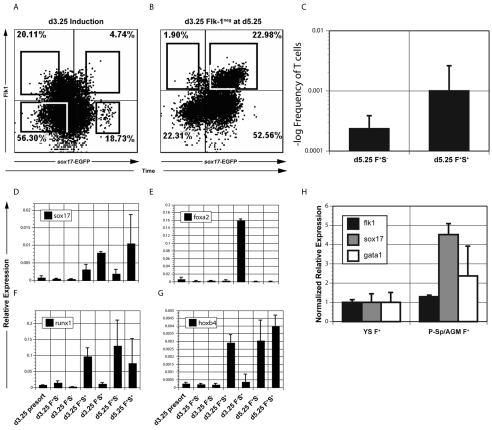
To determine whether the differential expression of Sox17 could be used to distinguish the Flk1pos populations, we used an ESC line in which EGFP was targeted to the Sox17 locus (Kim et al., 2007). The Sox17-EGFP cells responded well to the AVB induction protocol and generated a d3.25 Flk1pos population with similar efficiency to that observed with the T-EGFP cells (Fig. 5A,B). Two small distinct Sox17-positive populations were observed at this stage: an Flk1pos population that expressed an intermediate level of Sox17-EGFP (Sox17-EGFPint) and an Flk1neg population that expressed high levels of Sox17-EGFP (Sox17-EGFPhigh). The latter population is likely to represent a small amount of definitive endoderm induced under these conditions as it expressed Foxa2 in addition to Sox17 (Fig. 5D,E). Four different fractions were isolated from the d3.25 EBs: Flk1neg/Sox17-EGFPneg; Flk1neg/Sox17-EGFPhigh; Flk1pos/Sox17-EGFPneg (d3.25 F+ S–); and Flk1pos/Sox17-EGFPint (d3.25 F+ S+). The entire Flk1neg population was reaggregated and cultured in AVB media for an additional 48 hours. The Flk1neg cells generated a substantial second Flk1pos population, most of which expressed intermediate levels of Sox17-EGFP (Fig. 5B). The majority of the d5.25 Flk1neg population also expressed intermediate levels of Sox17-EGFP. The Flk1neg/Sox17-EGFPhigh population was not detected at this stage.
Fig. 5.
Isolation of Sox17-EGFP-positive cells from d3.25 and d5.25 populations. (A) Flow cytometric analyses showing Flk1 and Sox17-EGFP expression in d3.25 EBs. The four fractions indicated by the black boxes (d3.25 F+ S–, d3.25 F– S–, d3.25 F+ S+ and d3.25 F– S+; F, Flk1; S, Sox17) were isolated by cell sorting. (B) Flow cytometric analyses showing Flk1 and Sox17-EGFP expression in the d5.25 reaggregated population. Boxes indicate the Flk1pos populations (d5.25 F+ S+ and d5.25 F+ S–) isolated by cell sorting. (C) d5.25 F+ S+ and d5.25 F+ S– cells were plated in the limiting dilution OP9-DL1 co-culture assay for 3 weeks. The cells were then harvested and analyzed for the expression of T cell markers. (D-G) The sorted cell populations isolated in A and B were analyzed for the expression of the indicated genes by qRT-PCR. Data are presented as expression relative to that of Actb and are the mean of two independent experiments; error bars indicate ±s.d. (H) Flk1pos cells were isolated from E9.0 mouse embryos by flow cytometric cell sorting. Samples were analyzed for the expression of the indicated genes by qRT-PCR. Expression was standardized to that of Actb and are presented relative to the value found in the yolk sac (set at 1). Data shown are the mean of three independent experiments; error bars indicate ±s.d.
The Flk1pos/Sox17-EGFPneg and Flk1pos/Sox17-EGFPint cells from d3.25 and d5.25 were isolated and reaggregated in the presence of VEGF and KL for 24 hours and then tested for progenitor potential in methylcellulose cultures. There was no significant difference in precursor frequency between the two samples from d3.25, whereas the d5.25 Sox17-EGFPint population contained fewer primitive erythroid and more macrophage progenitors than the corresponding Sox17-EGFPneg population (see Fig. S4 in the supplementary material). Analyses of T cell potential at d5.25 revealed that the Flk1pos/Sox17-EGFPint population contained a higher frequency of T cell progenitors than the Flk1pos/Sox17-EGFPneg fraction. (1/988 versus 1/4205; n=4; Fig. 5C). Expression analyses (qRT-PCR) of the different fractions showed that Sox17 was expressed at the highest levels in the Sox17-EGFPint fractions, indicating that the EGFP reporter faithfully tracks endogenous Sox17 expression. Runx1 expression segregated to the Flk1pos/Sox17-EGFPint fraction at d3.25 and was detected in both d5.25 Flk1pos populations, whereas Hoxb4 was expressed at highest levels in the Sox17-EGFPint fractions at both time points. Taken together, these findings demonstrate that Sox17 is expressed in the Flk1pos progenitor population that develops following the onset of primitive hematopoiesis, displays lymphoid, myeloid and erythroid potential and co-expresses Runx1 and Hoxb4.
To determine whether different levels of Sox17 distinguish the different sites of hematopoietic development in the early embryo, we isolated Flk1pos cells from the YS and P-Sp/AGM regions of E9.0 embryos by cell sorting and analyzed them for Sox17 expression. As shown in Fig. 5H, the levels of Sox17 were significantly higher (P=0.0012) in the P-Sp-derived Flk1pos population than in the corresponding population isolated from the YS. Both populations expressed similar levels of Flk1. Expression of Gata1 indicates that this P-Sp/AGM population has hematopoietic potential. The observations from these embryo studies are consistent with findings from the ESC model and demonstrate that the YS- and P-Sp-derived Flk1pos populations express different levels of Sox17.
Regulation of d5.25 Flk1pos/Sox17-EGFPint development
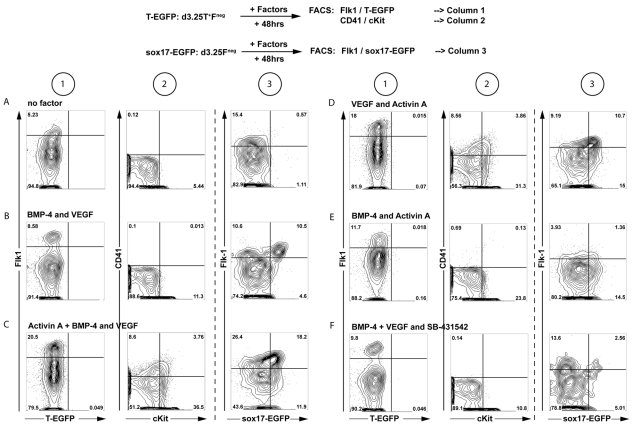
The d5.25 Flk1pos population used in the above studies was generated from d3.25 Flk1neg cells cultured in the presence of AVB. As this culture step is crucial for the development of this P-Sp-like progenitor, we investigated the role of different signaling pathways at this stage. For these studies, d3.25 Flk1neg cells from two different ESC lines (T-EGFP and Sox17-EGFP ESCs) were cultured in different factor combinations for 48 hours and the resulting aggregates analyzed for the presence of Flk1pos T-EGFP+ or Flk1pos Sox17-EGFPint progenitors and CD41+ c-Kit+ hematopoietic populations (Fig. 6A). In the absence of any factors, the d3.25 Flk1neg cells generated a modest size (5-16%) Flk1pos population with no detectable Sox17-EGFP expression. They did not give rise to any CD41 single-positive or CD41+ c-Kit+ cells, suggesting little, if any hematopoietic potential (Fig. 6A). The addition of BMP4 and VEGF to the d3.25 Flk1neg aggregation cultures promoted the development of an Flk1pos/Sox17-EGFPint population (9-31%; Fig. 6B). As with the population generated in the absence of added factors, the BMP4/VEGF-induced population did not give rise to CD41+ c-Kit+ hematopoietic cells at this stage. Activin A added together with BMP4 and VEGF substantially increased the size of the Flk1pos/Sox17-EGFPint population and also induced the development of CD41+ and CD41+ c-Kit+ populations (Fig. 6C). VEGF and activin A together also generated a substantial Flk1pos/Sox17-EGFPint population (18-35%; Fig. 6D) as well as the CD41+ and CD41+ c-Kit+ populations. In the absence of VEGF, together BMP4 and activin A induced a small Flk1pos/Sox17-EGFPint population and no CD41+ c-Kit+ cells (5-16% Flk1pos; Fig. 6E). These findings are consistent with the known requirement of VEGF for hematopoietic induction of Flk1pos cells (Choi et al., 1998; Nostro et al., 2008; Park et al., 2004; Pearson et al., 2008). The addition of SB-431542, a small-molecule inhibitor of the TGFβ1/activin/nodal pathway, to the BMP4 plus VEGF cultures decreased the size of the Flk1pos/Sox17-EGFPint population, but not of the overall Flk1pos population, indicating that the emergence of the Sox17-expressing cells in these conditions is likely to be dependent on endogenous nodal signaling (Fig. 6F).
Fig. 6.
Effect of activin A, BMP4 and VEGF on the generation of the secondary Flk1pos population. d3.25 T+ F– cells generated from either T-EGFP ESCs (columns 1+2) or Sox17-EGFP ESCs (column 3) were isolated, reaggregated in the indicated conditions for 48 hours and then analyzed for expression of T-EGFP, Sox17-EGFP, Flk1, c-Kit and CD41 (see schematic overview, top). SB-431542 is a small-molecule inhibitor of TGF-β1/activin/nodal signaling. BMP4, 10 ng/ml; activin A, 3ng/ml; VEGF, 5ng/ml; SB-431542, 6 μM final concentration. (A) StemPro 34 (SP34) media alone. (B-F) Reaggregation in the presence of (B) BMP4 and VEGF; (C) Activin A, BMP4 and VEGF; (D) VEGF and activin A; (E) BMP4 and activin A; (F) BMP4, VEGF and SB-431542. Quadrant labels were removed if zero (n=2 for both cell lines).
Given the known role of activin A signaling in PS induction and mesoderm formation (Gadue et al., 2006; Nostro et al., 2008), we were interested in determining whether the effect of activin A observed in these studies might be mediated at the level of mesoderm induction and/or expansion. To address this, d3.25 T+ F+ and d3.25 T+ F– populations were cultured as aggregates in the presence or absence of low levels of activin A for 24 hours and then analyzed for expression of T-EGFP. T+ F– cells cultured in the presence of activin A maintained a larger T-EGFP+ population than those cultured without it, suggesting that signaling through activin A sustains PS/mesodermal potential (see Table S1 in the supplementary material). Collectively, these observations demonstrate that the development of d5.25 Flk1pos cells from the d3.25 T-EGFP+ Flk1neg population is regulated by a combination of signaling pathways, including activin A/TGFβ, BMP and VEGF.
Hematopoietic development from iPSCs
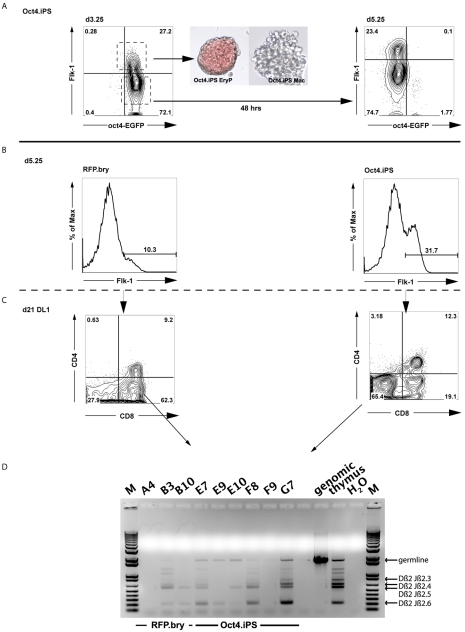
To determine whether the approach developed in this study is applicable to other pluripotent cell lines, we induced two different mouse iPSC lines [with EGFP targeted either to the Oct4 (Pou5f1 – Mouse Genome Informatics) or Sox2 locus] with AVB and analyzed the EBs for hematopoietic and lymphoid potential. Both iPSC lines responded well to the inducers and generated a substantial Flk1pos population by d3.25 (Fig. 7A; data not shown). The d3.25 Flk1pos and Flk1neg populations were isolated from the Oct4-EGFP-derived EBs and used for hematopoietic progenitor analysis and for the generation of d5.25 populations, respectively. Following 48 hours of culture, the Flk1neg cells gave rise to d5.25 Flk1pos and Flk1neg populations (Fig. 7A). The d5.25 Flk1pos cells were isolated and analyzed for hematopoietic potential. The patterns of hematopoietic progenitor development were similar to those observed for the ESC-derived fractions, as primitive erythroid colonies predominated the d3.25 Flk1pos cultures when compared with the d5.25 Flk1pos population (47.5% versus 31.5% of total), whereas macrophage colonies were more abundant in the cultures generated from the later-stage population (22.1% versus 54.1% of total) (see Fig. S5 in the supplementary material). For the analysis of their lymphoid potential, we implemented a simplified sorting strategy, providing access to the late-stage Flk1pos cells with only one cell-sorting step. For this approach, AVB-induced d3.25 EBs were dissociated and the entire population was reaggregated for 24 hours in the presence of activin A and then for an additional 24 hours in the presence of AVB. This protocol also yielded Flk1pos cells at d5.25, which were isolated and analyzed for their hematopoietic potential (Fig. 7B). This d5.25 Flk1pos population displayed T lymphoid potential from both iPSCs and ESCs (Fig. 7C). Analysis of these samples revealed the expected Tcrb locus rearrangement (Fig. 7D). These findings demonstrate that iPSCs do respond in a comparable fashion to ESCs to the inducers used for hematopoietic induction.
Fig. 7.
Generation and characterization of Flk1pos populations from iPSCs. (A) Oct4-EGFP iPSCs were differentiated in the presence of activin A, BMP4 and VEGF. Flk1pos and Flk1neg cells isolated at d3.25. Flk1pos cells were reaggregated for 24 hours and their hematopoietic potential was tested in a colony-forming assay. A primitive erythroid (EryP) and a macrophage (Mac) colony are shown, generated from the d3.25 Flk1pos population (40× magnification). Flk1neg cells were reaggregated for 48 hours and analyzed by flow cytometry at d5.25 for the emergence of a secondary Flk1pos population. (B) Flow cytometric analyses demonstrating the presence of an Flk1pos population in d5.25 aggregates generated from Oct4-EGFP iPSCs and tdRFP/T-EGFP (RFP.bry) cells. Both cell types were differentiated in the presence of activin A, BMP4 and VEGF. After dissociation at d3.25, the cells were reaggregated without cell sorting and cultured for an additional 48 hours, at which time they were sorted based on the expression of Flk1. (C) Flow cytometric profile demonstrating the generation of CD4+ CD8+ lymphocytes from the d5.25 Flk1pos ESC- and iPSC-derived progenitors following culture on OP9-Dl1 cells for 3 weeks. (D) PCR analyses showing T cell receptor gene rearrangements in the developing T cell populations from either the ESC progenitors (B3, B10) or the iPSC progenitors (E7, E9, E10, F8 and G7). M, 1 kb DNA ladder.
DISCUSSION
The efficient and reproducible generation of specific cell types from ESCs is dependent on accurately recapitulating in the differentiation cultures the key regulatory pathways that control the development of the corresponding lineage in the early embryo. Establishing the hematopoietic programs in such cultures requires not only activation of specific signaling pathways at appropriate stages, but also the ability to track the temporal changes associated with the emergence of the different sites of blood cell production in the embryo. In this study, we used reporter ESC lines carrying the EGFP cDNA targeted to either the T or Sox17 locus to follow the development of mesoderm subpopulations. With this approach we were able to identify two distinct Flk1pos progenitor populations that display hematopoietic potential indicative of YS and P-Sp development (see Fig. S1 in the supplementary material).
We initially identified the putative P-Sp progenitor as an Flk1pos cell that emerges in the ESC differentiation cultures following the development of a YS-like hematopoietic progenitor. These late-arising progenitors display substantially more lymphoid potential, have reduced primitive erythroid capacity and give rise to populations that are phenotypically similar to P-Sp-derived HSCs. An absolute separation of primitive erythroid and lymphoid potential in this system is difficult as the generation of Flk1pos mesoderm represents a continuum and there is no spatial separation of the developing populations as there is in the early embryo. The presence of Flk1 on the putative ESC-derived P-Sp population is consistent with studies in mouse and zebrafish showing that this receptor is expressed on hemogenic endothelium (Bertrand et al., 2010; Shalaby et al., 1997) and with transplantation experiments demonstrating that at least a subpopulation of AGM-derived HSCs is Flk1 positive (North et al., 2002). Lineage-tracing studies in both mouse and zebrafish clearly demonstrate that the majority of hematopoietic cells in the adult are derived from progenitors that at one stage expressed Flk1, strongly supporting the notion that the P-Sp hematopoietic program is established from an Flk1-positive progenitor (Ema et al., 2006; Bertrand et al., 2010; Lugus et al., 2009).
Although our studies show that Flk1 marks distinct hematopoietic populations in the ESC cultures, their isolation based solely on temporal patterns of Flk1 expression is challenging because the kinetics of differentiation can vary from cell line to cell line or following induction with different protocols in the same cell line. Our PCR analyses demonstrating that Sox17 is preferentially expressed in the second Flk1pos population indicated that the two populations do have different molecular profiles and that Sox17 might provide a marker of P-Sp hematopoiesis. Analysis of populations generated from the Sox17-EGFP reporter ESC line confirmed these findings and showed that Sox17-EGFP was found at highest levels in the d5.25 Flk1pos population. The fact that the d5.25 Flk1pos/Sox17-EGFPint population is enriched for lymphoid potential suggests that expression of Sox17 does mark the P-Sp-like progenitor population. A role for Sox17 in P-Sp-derived hematopoiesis in vivo is supported by in situ hybridization studies demonstrating that SoxF group transcription factors, including Sox17, are expressed in the ventral wall of the dorsal aorta of the P-Sp/AGM (Liao et al., 2009; Matsui et al., 2006) and by transplantation experiments showing that Sox17 is found in the majority of fetal liver-derived HSCs and is required for their development (Kim et al., 2007). Our analyses on the embryo populations clearly demonstrate that the P-Sp-derived Flk1pos cells express Sox17, suggesting that it might provide a marker for the emergence of the definitive hematopoietic program in vivo. The overlapping patterns of Sox17 and Hoxb4 in the d5.25 Flk1pos population is of particular interest in the context of Cdx4, as it has been shown that transient expression of Cdx4 followed by enforced expression of Hoxb4 in ESC-derived hematopoietic cells leads to the induction of cells with properties of HSCs (Wang et al., 2005a). Expression of Cdx4 in the d3.25 T+ Flk1neg fraction, followed by the endogenous upregulation of Hoxb4 expression in the d5.25 Flk1pos/Sox17int population derived from it, might reflect the normal molecular progression required to establish the P-Sp hematopoietic program and cells with HSC potential.
Although the d5.25 Flk1pos population displays properties of the P-Sp progenitor, attempts to repopulate Rag2–/– Il2rg–/– mice with these unmanipulated cells have thus far been unsuccessful (not shown). In this regard, they might be representative of the earliest P-Sp-derived hematopoietic progenitors that display limited repopulating potential. The repopulating capacity of these progenitors does increase with time in vivo and in P-Sp explants in vitro (Medvinsky and Dzierzak, 1996; Taoudi et al., 2008), suggesting that additional maturation steps are required to promote the differentiation of these pre-HSC progenitors to functional HSCs (Medvinsky and Dzierzak, 1996; Taoudi et al., 2008). Studies with human ESCs have indicated that interactions with appropriate stromal cell lines in vitro might provide the appropriate maturation signals required for the generation of HSCs (Ledran et al., 2008; Narayan et al., 2006; Wang et al., 2005a). Although the development of long-term repopulating multipotent HSCs under these conditions remains to be demonstrated, these findings do highlight the potential importance of interactions with the appropriate niche/cellular microenvironment for the differentiation and maturation of HSCs from PSCs.
The observation that the defined conditions used for the generation of the two Flk1pos hematopoietic populations from ESCs could also be used to induce the development of comparable populations from iPSCs is important, as it demonstrates that the principles of embryonic development can be applied to these PSCs as well, even in the absence of fluorescent marker genes. As the isolation of the d5.25 Flk1pos population with this approach is not dependent on the use of reporter ESCs, we expect it to be translatable to the human ESC (hESC) model. Studies with hESCs suggest that both YS- and P-Sp-derived hematopoiesis are initiated in the differentiation cultures (Galic et al., 2006; Galic et al., 2009; Tian et al., 2006; Timmermans et al., 2009; Vodyanik et al., 2005; Wang et al., 2005a). The onset of YS hematopoiesis has been defined by the emergence of a KDR-positive hemangioblast (Kennedy et al., 2007), whereas the development of a VE-cadherin+ CD45– hemogenic population might be reflective of the P-Sp program (Wang et al., 2004).
In summary, the findings reported here have identified distinct ESC-derived Flk1pos hematopoietic progenitors that display characteristics of the YS and P-Sp hematopoietic populations found in the early embryo. Detailed analysis of the emergence of the second Flk1pos population enabled the identification and isolation of intermediate populations that represent different stages in the progression of mesoderm to the putative P-Sp progenitor. Access to these populations provides a unique opportunity to investigate the regulatory pathways that control the development of this hematopoietic progenitor as well as those that promote its maturation to derivative hematopoietic progeny, including the HSC. Translation of this approach to human PSCs will represent an important step forward in modeling human hematopoiesis in culture as well as in accessing different cell populations for transplantation therapies.
Supplementary Material
Acknowledgments
We thank Andrea Ditadi for skillful dissection of the mouse embryos; Juan Carlos Zúñiga-Pflücker for providing the OP9 cell lines; Konrad Hochedlinger for the Sox1-EGFP and Oct4-EGFP iPSCs; the members of the G.M.K. laboratory for critical discussions of the manuscript; and the UHN/SickKids Flow Cytometry Facility for cell sorting. S.J.M. is an Investigator of the HHMI. This work was supported by National Institutes of Health grants R01 HL 48834-09 to G.M.K. and a fellowship from the Leopoldina Stiftung der Naturforscher Halle, Germany, to S.I. Deposited in PMC for release after 6 months.
Competing interests statement
The authors declare no competing financial interests.
Supplementary material
Supplementary material for this article is available at http://dev.biologists.org/lookup/suppl/doi:10.1242/dev.042119/-/DC1
References
- Bertrand J. Y., Giroux S., Golub R., Klaine M., Jalil A., Boucontet L., Godin I., Cumano A. (2005). Characterization of purified intraembryonic hematopoietic stem cells as a tool to define their site of origin. Proc. Natl. Acad. Sci. USA 102, 134-139 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bertrand J. Y., Chi N. C., Santoso B., Teng S., Stainier D. Y., Traver D. (2010). Haematopoietic stem cells derive directly from aortic endothelium during development. Nature 464, 108-111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boisset J. C., van Cappellen W., Andrieu-Soler C., Galjart N., Dzierzak E., Robin C. (2010). In vivo imaging of haematopoietic cells emerging from the mouse aortic endothelium. Nature 464, 116-120 [DOI] [PubMed] [Google Scholar]
- Cho S. K., Webber T. D., Carlyle J. R., Nakano T., Lewis S. M., Zuniga-Pflucker J. C. (1999). Functional characterization of B lymphocytes generated in vitro from embryonic stem cells. Proc. Natl. Acad. Sci. USA 96, 9797-9802 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cho S. K., Bourdeau A., Letarte M., Zuniga-Pflucker J. C. (2001). Expression and function of CD105 during the onset of hematopoiesis from Flk1(+) precursors. Blood 98, 3635-3642 [DOI] [PubMed] [Google Scholar]
- Choi K., Kennedy M., Kazarov A., Papadimitriou J. C., Keller G. (1998). A common precursor for hematopoietic and endothelial cells. Development 125, 725-732 [DOI] [PubMed] [Google Scholar]
- Conlon F. L., Lyons K. M., Takaesu N., Barth K. S., Kispert A., Herrmann B., Robertson E. J. (1994). A primary requirement for nodal in the formation and maintenance of the primitive streak in the mouse. Development 120, 1919-1928 [DOI] [PubMed] [Google Scholar]
- Cumano A., Dieterlen-Lievre F., Godin I. (1996). Lymphoid potential, probed before circulation in mouse, is restricted to caudal intraembryonic splanchnopleura. Cell 86, 907-916 [DOI] [PubMed] [Google Scholar]
- Cumano A., Ferraz J. C., Klaine M., Di Santo J. P., Godin I. (2001). Intraembryonic, but not yolk sac hematopoietic precursors, isolated before circulation, provide long-term multilineage reconstitution. Immunity 15, 477-485 [DOI] [PubMed] [Google Scholar]
- Davidson A. J., Ernst P., Wang Y., Dekens M. P., Kingsley P. D., Palis J., Korsmeyer S. J., Daley G. Q., Zon L. I. (2003). cdx4 mutants fail to specify blood progenitors and can be rescued by multiple hox genes. Nature 425, 300-306 [DOI] [PubMed] [Google Scholar]
- de Bruijn M. F., Ma X., Robin C., Ottersbach K., Sanchez M. J., Dzierzak E. (2002). Hematopoietic stem cells localize to the endothelial cell layer in the midgestation mouse aorta. Immunity 16, 673-683 [DOI] [PubMed] [Google Scholar]
- Dieterlen-Lievre F. (1975). On the origin of haemopoietic stem cells in the avian embryo: an experimental approach. J. Embryol. Exp. Morphol. 33, 607-619 [PubMed] [Google Scholar]
- Ema M., Takahashi S., Rossant J. (2006). Deletion of the selection cassette, but not cis-acting elements, in targeted Flk1-lacZ allele reveals Flk1 Expression in multipotent mesodermal progenitors. Blood 107, 111-117 [DOI] [PubMed] [Google Scholar]
- Fehling H. J., Lacaud G., Kubo A., Kennedy M., Robertson S., Keller G., Kouskoff V. (2003). Tracking mesoderm induction and its specification to the hemangioblast during embryonic stem cell differentiation. Development 130, 4217-4227 [DOI] [PubMed] [Google Scholar]
- Gadue P., Huber T. L., Paddison P. J., Keller G. M. (2006). Wnt and TGF-beta signaling are required for the induction of an in vitro model of primitive streak formation using embryonic stem cells. Proc. Natl. Acad. Sci. USA 103, 16806-16811 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galic Z., Kitchen S. G., Kacena A., Subramanian A., Burke B., Cortado R., Zack J. A. (2006). T lineage differentiation from human embryonic stem cells. Proc. Natl. Acad. Sci. USA 103, 11742-11747 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galic Z., Kitchen S. G., Subramanian A., Bristol G., Marsden M. D., Balamurugan A., Kacena A., Yang O., Zack J. A. (2009). Generation of T lineage cells from human embryonic stem cells in a feeder free system. Stem Cells 27, 100-107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Godin I. E., Garcia-Porrero J. A., Coutinho A., Dieterlen-Lievre F., Marcos M. A. (1993). Para-aortic splanchnopleura from early mouse embryos contains B1a cell progenitors. Nature 364, 67-70 [DOI] [PubMed] [Google Scholar]
- Irion S., Nostro M. C., Kattman S. J., Keller G. M. (2009). Directed differentiation of pluripotent stem cells: from developmental biology to therapeutic applications. Cold Spring Harbor Symp. Quant. Biol. 73, 101-110 [DOI] [PubMed] [Google Scholar]
- Kabrun N., Buhring H. J., Choi K., Ullrich A., Risau W., Keller G. (1997). Flk-1 expression defines a population of early embryonic hematopoietic precursors. Development 124, 2039-2048 [DOI] [PubMed] [Google Scholar]
- Kattman S. J., Huber T. L., Keller G. M. (2006). Multipotent Flk-1+ cardiovascular progenitor cells give rise to the cardiomyocyte, endothelial, and vascular smooth muscle lineages. Dev. Cell 11, 723-732 [DOI] [PubMed] [Google Scholar]
- Keller G. (2005). Embryonic stem cell differentiation: emergence of a new era in biology and medicine. Genes Dev. 19, 1129-1155 [DOI] [PubMed] [Google Scholar]
- Kennedy M., D'Souza S. L., Lynch-Kattman M., Schwantz S., Keller G. (2007). Development of the hemangioblast defines the onset of hematopoiesis in human ES cell differentiation cultures. Blood 109, 2679-2687 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim I., Saunders T. L., Morrison S. J. (2007). Sox17 dependence distinguishes the transcriptional regulation of fetal from adult hematopoietic stem cells. Cell 130, 470-483 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kissa K., Herbomel P. (2010). Blood stem cells emerge from aortic endothelium by a novel type of cell transition. Nature 464, 112-115 [DOI] [PubMed] [Google Scholar]
- Kyba M., Perlingeiro R. C., Daley G. Q. (2002). HoxB4 confers definitive lymphoid-myeloid engraftment potential on embryonic stem cell and yolk sac hematopoietic progenitors. Cell 109, 29-37 [DOI] [PubMed] [Google Scholar]
- Ledran M. H., Krassowska A., Armstrong L., Dimmick I., Renstrom J., Lang R., Yung S., Santibanez-Coref M., Dzierzak E., Stojkovic M., et al. (2008). Efficient hematopoietic differentiation of human embryonic stem cells on stromal cells derived from hematopoietic niches. Cell Stem Cell 3, 85-98 [DOI] [PubMed] [Google Scholar]
- Lengerke C., McKinney-Freeman S., Naveiras O., Yates F., Wang Y., Bansal D., Daley G. Q. (2007). The Cdx-Hox pathway in hematopoietic stem cell formation from embryonic stem cells. Ann. NY Acad. Sci. 1106, 197-208 [DOI] [PubMed] [Google Scholar]
- Liao W. P., Uetzmann L., Burtscher I., Lickert H. (2009). Generation of a mouse line expressing Sox17-driven cre recombinase with specific activity in arteries. Genesis 47, 476-483 [DOI] [PubMed] [Google Scholar]
- Liu P., Wakamiya M., Shea M. J., Albrecht U., Behringer R. R., Bradley A. (1999). Requirement for Wnt3 in vertebrate axis formation. Nat. Genet. 22, 361-365 [DOI] [PubMed] [Google Scholar]
- Luche H., Weber O., Nageswara Rao T., Blum C., Fehling H. J. (2007). Faithful activation of an extra-bright red fluorescent protein in `knock-in' cre-reporter mice ideally suited for lineage tracing studies. Eur. J. Immunol. 37, 43-53 [DOI] [PubMed] [Google Scholar]
- Lugus J. J., Park C., Ma Y. D., Choi K. (2009). Both primitive and definitive blood cells are derived from Flk-1+ mesoderm. Blood 113, 563-566 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsui T., Kanai-Azuma M., Hara K., Matoba S., Hiramatsu R., Kawakami H., Kurohmaru M., Koopman P., Kanai Y. (2006). Redundant roles of Sox17 and Sox18 in postnatal angiogenesis in mice. J. Cell Sci. 119, 3513-3526 [DOI] [PubMed] [Google Scholar]
- McKinney-Freeman S. L., Daley G. Q. (2007). Towards hematopoietic reconstitution from embryonic stem cells: a sanguine future. Curr. Opin. Hematol. 14, 343-347 [DOI] [PubMed] [Google Scholar]
- Medvinsky A., Dzierzak E. (1996). Definitive hematopoiesis is autonomously initiated by the AGM region. Cell 86, 897-906 [DOI] [PubMed] [Google Scholar]
- Medvinsky A. L., Samoylina N. L., Muller A. M., Dzierzak E. A. (1993). An early pre-liver intraembryonic source of CFU-S in the developing mouse. Nature 364, 64-67 [DOI] [PubMed] [Google Scholar]
- Medvinsky A. L., Gan O. I., Semenova M. L., Samoylina N. L. (1996). Development of day-8 colony-forming unit-spleen hematopoietic progenitors during early murine embryogenesis: spatial and temporal mapping. Blood 87, 557-566 [PubMed] [Google Scholar]
- Murry C. E., Keller G. (2008). Differentiation of embryonic stem cells to clinically relevant populations: lessons from embryonic development. Cell 132, 661-680 [DOI] [PubMed] [Google Scholar]
- Nakano T., Kodama H., Honjo T. (1994). Generation of lymphohematopoietic cells from embryonic stem cells in culture. Science 265, 1098-1101 [DOI] [PubMed] [Google Scholar]
- Narayan A. D., Chase J. L., Lewis R. L., Tian X., Kaufman D. S., Thomson J. A., Zanjani E. D. (2006). Human embryonic stem cell-derived hematopoietic cells are capable of engrafting primary as well as secondary fetal sheep recipients. Blood 107, 2180-2183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ng E. S., Azzola L., Sourris K., Robb L., Stanley E. G., Elefanty A. G. (2005). The primitive streak gene Mixl1 is required for efficient haematopoiesis and BMP4-induced ventral mesoderm patterning in differentiating ES cells. Development 132, 873-884 [DOI] [PubMed] [Google Scholar]
- North T. E., de Bruijn M. F., Stacy T., Talebian L., Lind E., Robin C., Binder M., Dzierzak E., Speck N. A. (2002). Runx1 expression marks long-term repopulating hematopoietic stem cells in the midgestation mouse embryo. Immunity 16, 661-672 [DOI] [PubMed] [Google Scholar]
- Nostro M. C., Cheng X., Keller G. M., Gadue P. (2008). Wnt, activin, and BMP signaling regulate distinct stages in the developmental pathway from embryonic stem cells to blood. Cell Stem Cell 2, 60-71 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palis J., Roberston S., Kennedy M., Wall C., Keller G. (1999). Development of erythroid and myeloid progenitors in the yolk sac and embryo proper of the mouse. Development 126, 5073-5084 [DOI] [PubMed] [Google Scholar]
- Pandolfi P. P., Roth M. E., Karis A., Leonard M. W., Dzierzak E., Grosveld F. G., Engel J. D., Lindenbaum M. H. (1995). Targeted disruption of the GATA3 gene causes severe abnormalities in the nervous system and in fetal liver haematopoiesis. Nat. Genet. 11, 40-44 [DOI] [PubMed] [Google Scholar]
- Parameswaran M., Tam P. P. (1995). Regionalisation of cell fate and morphogenetic movement of the mesoderm during mouse gastrulation. Dev. Genet. 17, 16-28 [DOI] [PubMed] [Google Scholar]
- Park C., Afrikanova I., Chung Y. S., Zhang W. J., Arentson E., Fong Gh G., Rosendahl A., Choi K. (2004). A hierarchical order of factors in the generation of FLK1- and SCL-expressing hematopoietic and endothelial progenitors from embryonic stem cells. Development 131, 2749-2762 [DOI] [PubMed] [Google Scholar]
- Pearson S., Sroczynska P., Lacaud G., Kouskoff V. (2008). The stepwise specification of embryonic stem cells to hematopoietic fate is driven by sequential exposure to Bmp4, Activin A, bFGF and VEGF. Development 135, 1525-1535 [DOI] [PubMed] [Google Scholar]
- Petrenko O., Beavis A., Klaine M., Kittappa R., Godin I., Lemischka I. R. (1999). The molecular characterization of the fetal stem cell marker AA4. Immunity 10, 691-700 [DOI] [PubMed] [Google Scholar]
- Robb L., Elwood N. J., Elefanty A. G., Köntgen F., Li R., Barnett L. D., Begley C. G. (1996). The scl gene product is required for the generation of all hematopoietic lineages in the adult mouse. EMBO J. 15, 4123-4129 [PMC free article] [PubMed] [Google Scholar]
- Rubin L. L. (2008). Stem cells and drug discovery: the beginning of a new era? Cell 132, 549-552 [DOI] [PubMed] [Google Scholar]
- Russell E. S. (1979). Hereditary anemias of the mouse: a review for geneticists. Adv. Genet. 20, 357-459 [PubMed] [Google Scholar]
- Samokhvalov I. M., Samokhvalova N. I., Nishikawa S. (2007). Cell tracing shows the contribution of the yolk sac to adult haematopoiesis. Nature 446, 1056-1061 [DOI] [PubMed] [Google Scholar]
- Schmitt T. M., Zuniga-Pflucker J. C. (2002). Induction of T cell development from hematopoietic progenitor cells by delta-like-1 in vitro. Immunity 17, 749-756 [DOI] [PubMed] [Google Scholar]
- Schmitt T. M., de Pooter R. F., Gronski M. A., Cho S. K., Ohashi P. S., Zuniga-Pflucker J. C. (2004). Induction of T cell development and establishment of T cell competence from embryonic stem cells differentiated in vitro. Nat. Immunol. 5, 410-417 [DOI] [PubMed] [Google Scholar]
- Shalaby F., Ho J., Stanford W. L., Fischer K. D., Schuh A. C., Schwartz L., Bernstein A., Rossant J. (1997). A requirement for Flk1 in primitive and definitive hematopoiesis and vasculogenesis. Cell 89, 981-990 [DOI] [PubMed] [Google Scholar]
- Stadtfeld M., Maherali N., Breault D. T., Hochedlinger K. (2008). Defining molecular cornerstones during fibroblast to iPS cell reprogramming in mouse. Cell Stem Cell 2, 230-240 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabatabaei-Zavareh N., Vlasova A., Greenwood C. P., Takei F. (2007). Characterization of developmental pathway of natural killer cells from embryonic stem cells in vitro. PLoS ONE 2, e232 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taoudi S., Gonneau C., Moore K., Sheridan J. M., Blackburn C. C., Taylor E., Medvinsky A. (2008). Extensive hematopoietic stem cell generation in the AGM region via maturation of VE-cadherin+CD45+ pre-definitive HSCs. Cell Stem Cell 3, 99-108 [DOI] [PubMed] [Google Scholar]
- Tian X., Woll P. S., Morris J. K., Linehan J. L., Kaufman D. S. (2006). Hematopoietic engraftment of human embryonic stem cell-derived cells is regulated by recipient innate immunity. Stem Cells 24, 1370-1380 [DOI] [PubMed] [Google Scholar]
- Timmermans F., Velghe I., Vanwalleghem L., De Smedt M., Van Coppernolle S., Taghon T., Moore H. D., Leclercq G., Langerak A. W., Kerre T., et al. (2009). Generation of T cells from human embryonic stem cell-derived hematopoietic zones. J. Immunol. 182, 6879-6888 [DOI] [PubMed] [Google Scholar]
- Vodyanik M. A., Bork J. A., Thomson J. A., Slukvin I. I. (2005). Human embryonic stem cell-derived CD34+ cells: efficient production in the coculture with OP9 stromal cells and analysis of lymphohematopoietic potential. Blood 105, 617-626 [DOI] [PubMed] [Google Scholar]
- Wang L., Li L., Shojaei F., Levac K., Cerdan C., Menendez P., Martin T., Rouleau A., Bhatia M. (2004). Endothelial and hematopoietic cell fate of human embryonic stem cells originates from primitive endothelium with hemangioblastic properties. Immunity 21, 31-41 [DOI] [PubMed] [Google Scholar]
- Wang L., Menendez P., Cerdan C., Bhatia M. (2005a). Hematopoietic development from human embryonic stem cell lines. Exp. Hematol. 33, 987-996 [DOI] [PubMed] [Google Scholar]
- Wang Q., Stacy T., Binder M., Marin-Padilla M., Sharpe A. H., Speck N. A. (1996). Disruption of the Cbfa2 gene causes necrosis and hemorrhaging in the central nervous system and blocks definitive hematopoiesis. Proc. Natl. Acad. Sci. USA 93, 3444-3449 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y., Yates F., Naveiras O., Ernst P., Daley G. Q. (2005b). Embryonic stem cell-derived hematopoietic stem cells. Proc. Natl. Acad. Sci. USA 102, 19081-19086 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winnier G., Blessing M., Labosky P. A., Hogan B. L. (1995). Bone morphogenetic protein-4 is required for mesoderm formation and patterning in the mouse. Genes Dev. 9, 2105-2116 [DOI] [PubMed] [Google Scholar]
- Yamane T., Hosen N., Yamazaki H., Weissman I. L. (2009). Expression of AA4.1 marks lymphohematopoietic progenitors in early mouse development. Proc. Natl. Acad. Sci. USA 106, 8953-8958 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoder M. C., Hiatt K., Dutt P., Mukherjee P., Bodine D. M., Orlic D. (1997). Characterization of definitive lymphohematopoietic stem cells in the day 9 murine yolk sac. Immunity 7, 335-344 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.