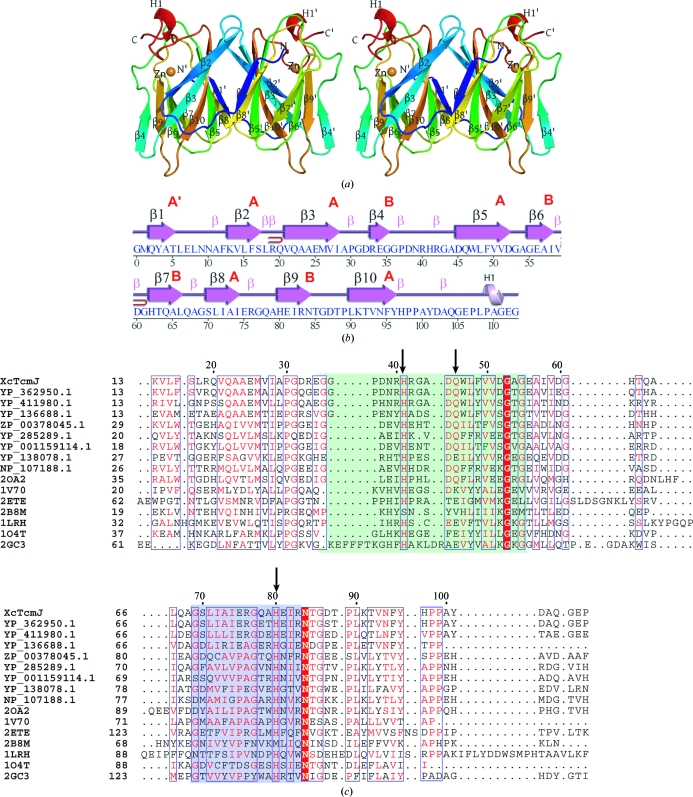
Figure 1.
Crystal structure of XcTcmJ from X. campestris pv. campestris. (a) Stereo ribbon diagram of the XcTcmJ dimer color coded from the N-terminus (blue) to the C-terminus (red). Helices H1 and H1′ and β-strands (β1–β10, β1′–β10′) are indicated. A Zn atom in the vicinity of the putative active site of each monomer is shown as a sphere (orange). (b) Diagram showing the secondary-structure elements of XcTcmJ superimposed on its primary sequence. The 310-helices, β-strands and β-turns are indicated. The β-sheets are indicated in red letters. The β-hairpins are depicted as red loops. (c) Multiple sequence alignments for XcTcmJ and related proteins. The first four protein sequences belong to XcTcmJ (GenBank NP_636471) and its putative orthologs (GenBank YP_362950, YP_411980 and YP_136688). Sequences 5–16 belong to proteins that are similar but have a different pattern of conserved residues (GenBank or PDB codes ZP_00378045, YP_285289, YP_001159114, YP_138078, NP_107188, 2oa2, 1v70, 1o4t, 1lrh and 2gc3). The positions of the three residues (His41, Gln46 and His80) within coordination distance of the Zn atom in the putative active site in the XcTcmJ structure are labeled with arrows. The two conserved cupin sequence motifs (Dunwell et al., 2004 ▶) are shaded in green and blue.