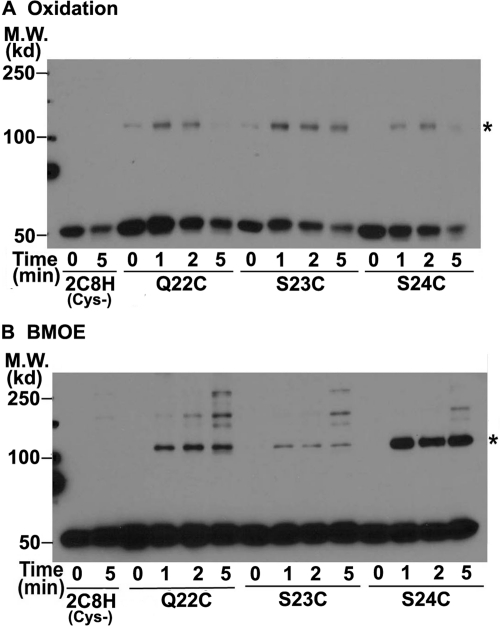
Fig. 4.
2C8H with Cys present at positions 22, 23, and 24 in the linker sequence of CYP2C8 can form cross-linked dimers. To reduce the formation of higher order complexes, the 2C8H(Cys-) mutant in which 7 Cys were mutated was used, and Cys was substituted at either residue 22, 23, or 24. The proteins were expressed in bacteria, cross-linked by oxidation of Cys (A) or BMOE treatment (B), and analyzed as described in the legend to Fig. 2. Asterisks denote the expected mobility of the 2C8H dimer. M.W., molecular weight.