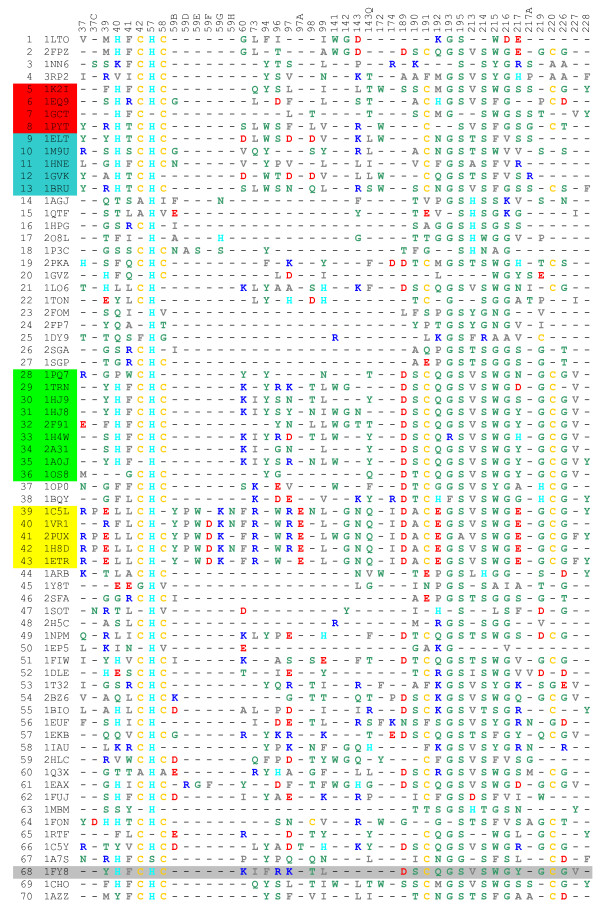
Figure 1.
The IFR positions and their occupancy by amino acids shown for 70 serine proteases bound to BPTI. The rows show amino acids occupying IFR positions for specific serine protease (the PDB ID given in the second column). The top row shows the sequence numbers in structurally aligned serine proteases (only IFRs are listed) for all enzymes in complex with the BPTI. The complexes were created by the procedure we named "hard docking", except for the case of 1fy8.pdb for which we have experimental data (row marked with light gray background). The color code for amino acids is as follows: Residues: AVLIMFP are colored grey [containing: small hydrophobic residues (A and P) and large hydrophobic residues]; Residues: STYNQWG are colored green [containing: polar residues]; Residues: D and E are colored red [containing: negatively charged residues]; Residues: R and K are colored blue [containing: positively charged residues]; Residue: H is colored cyan [containing: positively charged residue which is neutral at neutral pH]; Residue: C is colored yellow [containing: disulphide bridge forming residue]. {See text for details about amino acid classification} The rows are occupied by following serine protease sub-families: Tryptase(1-2), Chymase (3-4), Chymotrypsin (5-8) {red background}, Elastase (9-13){blue background}, Exfoliative toxin (14-15), Glutamyl endopeptidase (16-18), Kallikrein (19-22), NS3 protease (23-25), Streptogrisin (26-27), Trypsin (28-36){green background}, Venom (37-38), Thrombin (39-43){yellow background}, Miscellaneous-Prokaryotes (44-48), Miscellaneous-Virus (49-50), Miscellaneous (51-67), Native Complex (68-70) in respective row order.