Fig. (2). Basement Membrane Assembly.
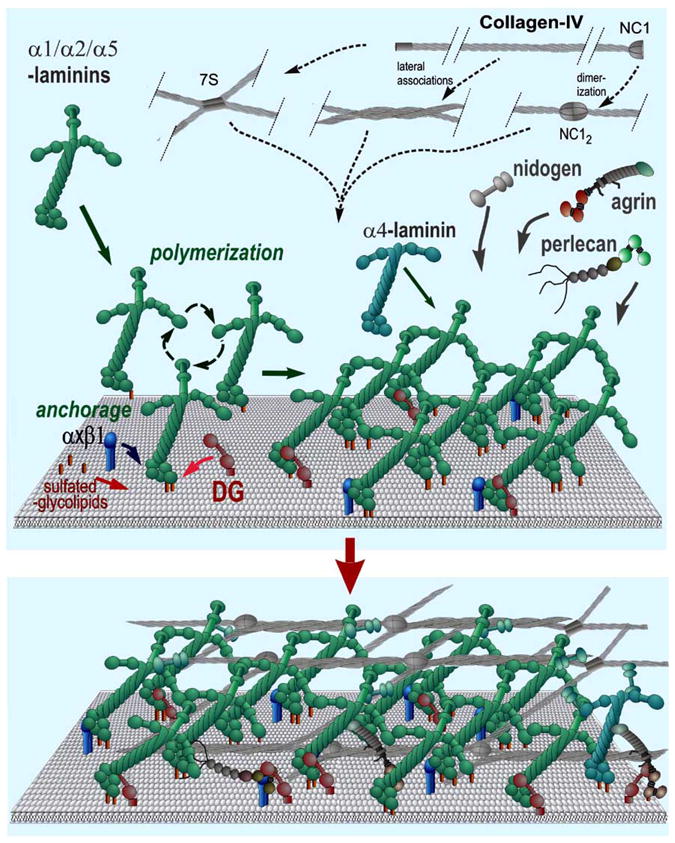
In this simplified model, laminins become anchored to the cell surface through their LG domains. Anchorage is further enhanced through binding of the α-LN domain to sulfated glycolipids. If the laminin has three LN domains it polymerizes, creating a “nascent” scaffolding. Nidogens, type IV collagens, perlecan, and agrin, are incorporated into this initial matrix by binding to laminin (or by binding through a nidogen bridge). The type IV collagen self-assembles into a covalently-crosslinked network. The non-laminin components provide crucial stability and increase ligand complexity. The basement membrane ligands interact with integrins and dystroglycan and the heparan sulfates of agrin and perlecan enable the tethering of tissue-specific growth factors.
