Abstract
Background
Up to 90% of neonates with congenital or perinatal cytomegalovirus (CMV) infection are asymptomatic, and little is known about CMV-associated thrombocytopenia after the neonatal period. We investigated the clinical findings of a series of infants diagnosed with CMV infection and thrombocytopenia.
Methods
From July 2005 to July 2008, infants aged younger than 6 months with thrombocytopenia were screened for CMV infection, using CMV IgM. Those who were positive for CMV IgM were then tested for CMV IgG via polymerase chain reaction (PCR) for CMV and CMV pp65 Ag and urine culture. Brain magnetic resonance imaging (MRI) and otologic and ophthalmologic evaluations were also performed.
Results
Twenty-one patients aged between 1 and 6 months (11 boys and 10 girls) were admitted and tested for CMV infection. Six patients (28.6%) were positive for CMV IgM; these were also positive for CMV IgG, CMV PCR, and urine culture, and 4 were also positive for CMV pp65 Ag. The median platelet count at admission was 6,500/µL (range, 2,000-105,000/µL). One patient (16.7%) was diagnosed with Evans syndrome and had calcifications on brain MRI. One patient had unilateral sensorineural hearing loss.
Conclusion
Thrombocytopenia can be the main clinical manifestation of otherwise asymptomatic CMV infection after the neonatal period, and close follow-up of neurodevelopmental sequelae is needed.
Keywords: Thrombocytopenia, Cytomegalovirus, Infant
INTRODUCTION
Cytomegalovirus (CMV) infection, the most common congenital infection, affects approximately 1% of all live births [1-3]. Congenital CMV infection is usually transmitted to the fetus in utero, whereas perinatal infection occurs during the intrapartum period or immediately after birth [1]. The diagnosis of congenital CMV infection is established by isolating the virus from urine, saliva, or tissue during the first 3 weeks of life [1]. Only about 1-15% of neonates with congenital CMV infection have symptoms at birth [4]; approximately 90% of infected newborns show no overt signs of infection. Thus, congenital CMV infection is easily overlooked [1, 4]. Subsequently, 10-20% of asymptomatic newborns develop late sequelae, particularly sensorineural hearing loss (SNHL) and neurodevelopmental disorders [4, 5].
Thrombocytopenia is an important prognostic factor in patients with CMV infection. Rivera et al. reported that CMV-induced neonatal thrombocytopenia is associated with symptoms at birth [6]. Benoist et al. demonstrated that the presence of abnormalities on prenatal ultrasound examination and thrombocytopenia appear to be strong, independent prenatal predictors of a poor outcome [3]. Petechiae are usually, but not always, present in patients with thrombocytopenia, and platelet counts in the first few weeks of life range from 2,000 to 125,000/µL [7]. Therefore, thrombocytopenia may be overlooked in the neonatal period, and it may persist or worsen after the neonatal period. However, little data are available on the association between congenital or perinatal CMV infection and thrombocytopenia during early infancy.
Therefore, we evaluated the clinical manifestations and neurodevelopmental outcomes of CMV-induced thrombocytopenia in infants aged younger than 6 months.
MATERIALS AND METHODS
1. Patients
From July 2005 to July 2008, patients with thrombocytopenia who were between 1 and 6 months of age were tested for IgM for Toxoplasma, syphilis, rubella, CMV, and herpes simplex virus (TORCH). Patients who tested positive for CMV IgM were further tested for CMV IgG via a polymerase chain reaction (PCR) test for CMV and CMV pp65 Ag and urine culture. Brain magnetic resonance imaging (MRI) and otologic and ophthalmologic evaluations were also performed. Thrombocytopenia was defined as a platelet count of less than 120,000/µL. If the platelet count recovered to normal levels (>150,000/µL) within 6 months after diagnosis, it was defined as the "acute form;" if thrombocytopenia persisted for longer than 6 months, it was defined as the "chronic form" [8]. Newborn infants with neonatal alloimmune thrombocytopenia and immune thrombocytopenic purpura (ITP) secondary to maternal ITP were excluded from this study.
2. Laboratory tests for CMV infection
CMV IgM and IgG in the patients' sera were detected using an enzyme immunoassay (Zeus Scientific, Branchburg, NJ, USA) and a microparticle enzyme immunoassay (AxSYM CMV IgG Assay; Abbott Laboratories, Abbott Park, IL, USA), respectively. For the CMV pp65 Ag assay, leukocytes were collected from anticoagulated blood by dextran agglutination and lysis of red blood cells. After centrifuging, 2×105 white blood cells were prepared on cytospin slides (Thermo Electron, Waltham, MA, USA) and fixed with formaldehyde. They were incubated with C10/C11 monoclonal antigen (Clonab CMV, Biotest, Dreieich, Germany) and stained with alkaline phosphatase/anti-alkaline phosphatase. Cells that stained red were considered to be positive for pp65 Ag, and the result was reported as the number of red-stained cells per 2×105 cells. For detecting CMV in urine culture, urine samples were collected from the patients and cultured, using the shell vial culture method (Chemicon, Temecula, CA, USA). Finally, EDTA-anticoagulated blood samples were used for the CMV PCR test, using an ABSOLUTE™ CMV PCR kit (BioSewoom, Seoul, South Korea) on an ABI 2720 Thermocycler (Applied Biosystems, Foster City, CA, USA).
3. Otologic and ophthalmologic evaluations
All patients underwent otologic and ophthalmologic tests after initial diagnosis of thrombocytopenia. The otologic evaluation included auditory brain stem evoked potentials (ABR) to detect SNHL. The ophthalmologic evaluation included a funduscopic evaluation for chorioretinitis, retinal scars, optic atrophy, and central vision loss. Patients with abnormal findings were followed at 3-month intervals.
4. Radiologic evaluations
All patients underwent brain MRI after the initial diagnosis of thrombocytopenia. In one patient who had been born prematurely (patient 4), the MRI findings were compared to those obtained at birth and the findings of brain ultrasonography were also considered.
RESULTS
1. Patients characteristics and clinical manifestations
Of the 21 patients in the study (11 boys and 10 girls), 6 (28.6%) were positive for CMV IgM (4 boys and 2 girls). The median duration of follow-up was 33 months (range, 16-51 months). All 6 patients tested positive for CMV IgG, CMV PCR, and CMV urine culture, while 4 tested positive for CMV pp65 Ag. The other tests for TORCH were all negative. The median gestational age at birth of the patients was 39 weeks and 3 days (range, 32 weeks, 3 days-40 weeks, and 5 days). All patients had birth weights appropriate for gestational age and normal head circumferences (median, 36.5 cm; range, 29-40 cm). No significant differences in gestational age or head circumference were observed between the CMV IgM-positive and CMV IgM-negative infants (median, 40 weeks and 36 cm, respectively). Except for one patient (patient 4) who was born prematurely (32 weeks and 3 days), no patient had a history of hospital admission during the neonatal period or had symptoms associated with CMV infection. The median age at diagnosis of thrombocytopenia was 2 months (range, 1.8-4 months), which did not differ from that of CMV IgM-negative patients (median, 2 months; range, 1.5-5 months) (Table 1). The median platelet count was 6,500/µL (range, 2,000-105,000/µL), and the median levels of aspartate transaminase (AST) and alanine transaminase (ALT) were 52.5 IU (range, 30-93 IU) and 40.5 IU (range, 25-97 IU), respectively, which did not differ significantly from the values for CMV IgM-negative infants [median, 56 IU (range, 28-122 IU) and 35 IU (range, 16-117 IU), respectively] (Table 2). The chief complaint was petechiae in all 21 patients; no patient had other histories of bleeding or hepatosplenomegaly. Patient 2's complete blood counts at birth revealed thrombocytopenia and anemia. If the patient had positive results for the direct Coomb's test, he/she was diagnosed with Evans syndrome. Patient 3 had a platelet count of 105,000/µL and had a history of petechiae 3 days before admission, which disappeared on admission. Except for this patient, all were treated with 1 g/kg/day intravenous immunoglobulin for 2 days or 400 mg/kg/day for 5 days until their platelet counts normalized. No patient had persistent thrombocytopenia for longer than 6 months (Table 3).
Table 1.
Comparisons of patients with thrombocytopenia according to CMV status.
Abbreviations: CMV, cytomegalovirus; GA, gestational age; HC, head circumference; Ht, height; WBC, white blood cell; Hb, hemoglobin; PLT, platelet.
Table 2.
Summary of test results in patients with CMV infection in infancy.
Abbreviations: PLT, platelet; AST, aspartate transaminase; ALT, alanine transaminase; D/ID, direct/indirect; CMV, cytomegalovirus; PCR, polymerase chain reaction.
Table 3.
Clinical manifestations of the patients.
Abbreviation: IVIG, intravenous immune globulin.
2. Otologic and ophthalmologic involvement
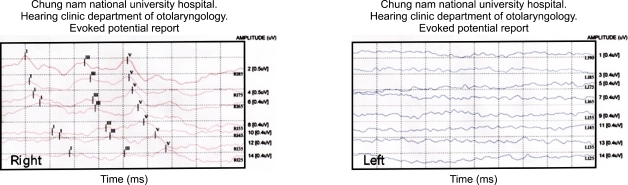
Patient 1 (1/6; 16.6%) had unilateral SNHL (Fig. 1). The remaining 5 patients had no otologic abnormalities at initial evaluation or at the 6- or 12-month follow-up evaluations. None of the 6 patients had ophthalmologic 1abnormalities. All 6 patients are still being followed regularly.
Fig. 1.
Patient 1 shows normal auditory brainstem response (ABR) on the right side (up to 25 dB) and minimal response on the left side.
3. Neurological manifestations
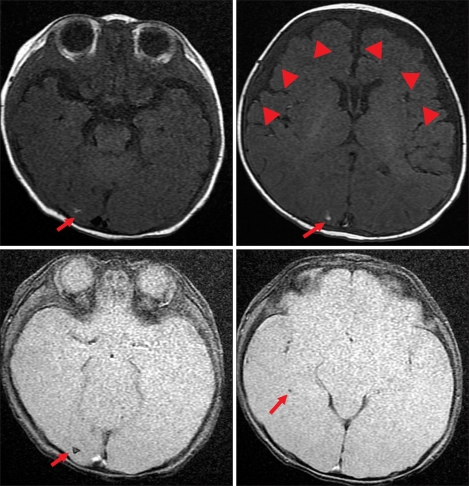
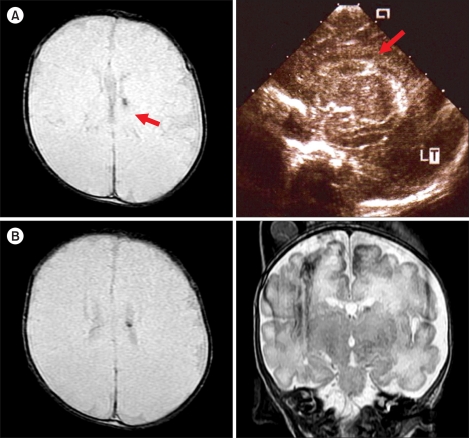
Of the 6 patients with CMV infection-associated thrombocytopenia, 2 (33.3%) had abnormal findings on brain MRI scans. Patient 2, who had Evans syndrome, had a calcified inflammatory granuloma in the right occipital lobe and a subdural effusion in the right frontotemporal convexity, which were considered sequelae of the CMV infection (Fig. 2). Patient 4, who was born prematurely, had a small nodular dark signal intensity lesion in the left caudothalamic notch, which was suspected to have resulted from a germinal matrix hemorrhage (Fig. 3). The findings of MRI and brain ultrasonography at birth in patient 4 did not change later and were considered to be a germinal matrix hemorrhage in prematurity. Both patients are currently being followed regularly and have had no neurological sequelae or other symptoms or signs.
Fig. 2.
Brain MRI of Patient 2 shows multiple small nodular T1-high and gradient echo-dark signal intensity lesions in the right occipital lobe (arrow). This finding is compatible with a calcified inflammatory granuloma as a sequela of previous CMV infection. Also note the subdural effusion and right frontotemporal convexity (arrowheads).
Fig. 3.
(A) Brain images of Patient 4 during the neonatal period. Red arrows point to the small nodular T2-weighted GRE-dark signal intensity lesion in the caudo-thalamic notch on the MRI scan (left) and left germinal matrix hemorrhage on brain sonogram (right). (B) Brain MRI scan of the patient obtained at the diagnosis of thrombocytopenia showed a small nodular T2-weighted axial GRE-(left) and coronal FSE (right)-dark signal intensity lesion in the caudo-thalamic notch (red arrows).
DISCUSSION
CMV is the most common cause of intrauterine infection, affecting 0.5-2.4% of live births; perinatal infection may occur from exposure to genital secretions at the time of delivery or from infected breast milk or close contact with an infected person [1, 3]. Currently, routine screening for CMV infection is not recommended in neonates because the overall risk of symptomatic CMV infection is low (0.2%) and because several limitations are imposed in screening all pregnant women: the positive predictive value is low, fetal diagnosis requires invasive amniocentesis, and the only available intervention at present is abortion [1, 5].
Few studies have reported on cases of asymptomatic CMV infection after the neonatal period. Although 85-90% of children with congenital CMV infection are asymptomatic during the neonatal period, some may subsequently develop symptomatic disease [4]. In fact, 10-20% of children with congenital CMV, either symptomatic or asymptomatic during the neonatal period, exhibit neurological deficits when followed up, which indicates that close follow-up evaluation, particularly of any neurological deficits, is warranted [4, 9]. None of the patients in this study had apparent clinical manifestations of CMV infection at birth, but they later developed thrombocytopenia, which was the chief reason for hospital admission.
The presence of IgM at birth is highly suggestive of a congenital infection, although its absence does not rule out the possibility of congenital infection. Typically, the diagnosis of congenital CMV infection is based on testing positive for IgM at 2-3 weeks of age; however, only 70% of patients who test positive for IgM are clinically significant cases of CMV infection. Direct demonstration of the virus' presence is needed for the diagnosis in neonates [4, 10]. However, patients with primary and secondary infection continue to test positive for IgM antibody for 6-18 months; hence, the IgM antibody test is useful in infants suspected of having CMV infection [5].
The major symptoms of CMV infection include intrauterine growth retardation, jaundice with direct hyperbilirubinemia, hepatosplenomegaly, petechiae or purpura, thrombocytopenia, and pneumonia. SNHL and ocular involvement are important late sequelae.
Petechiae are usually, but not always, accompanied by thrombocytopenia, and the platelet counts during the first few weeks of life range from 2,000 to 125,000/µL [1]. The thrombocytopenia usually resolves during the second week of life, but may persist and require treatment with repeated platelet transfusions. While several studies have reported on thrombocytopenia associated with CMV infection, the role of CMV infection in typical acute, self-limited, or persistent idiopathic immune thrombocytopenia (ITP) in children remains unclear. Several hypotheses exist to explain how CMV infection causes thrombocytopenia: direct cytotoxicity of CMV on hematopoietic cells, immune-mediated destruction of infected cells, or impaired bone marrow stromal function by CMV [11, 12]. In addition, CMV infection may induce nonspecific autoantibodies that cause antibody-mediated platelet destruction [13]. No apparent correlation exists between CMV clearance and the resolution of thrombocytopenia [11].
Evans syndrome has rarely been described in association with CMV infection; one report described 3 of 28 pediatric patients presenting with thrombocytopenia (11%) as being positive for CMV in the urine, using the shell vial assay [11]. Of these, 2 patients were diagnosed with ITP, and 1 patient had Evans syndrome [11]. One patient in our series had Evans syndrome; he had abnormal findings on brain MRI scans but no otologic or ophthalmologic manifestations.
Except for the patient with an initial platelet count 105,000/µL, all patients with CMV-induced thrombocytopenia in this study were treated with intravenous immunoglobulin; their platelet counts normalized within 3 months of the diagnosis, which is compatible with a diagnosis of the acute form of ITP. However, reports have described infants with CMV infection who experienced disease progression to chronic ITP, even with intravenous immunoglobulin therapy [14]. Therefore, long-term follow-up of our patients is needed to completely rule out chronic disease.
In cases of SNHL, the follow-up period is extremely important. A previous study reported that only 59% of the patients with SNHL diagnosed at 72 months of age had been correctly diagnosed at 12 months [15-17]. In addition, approximately half the cases with hearing loss due to congenital CMV infection were reported to be late-onset or progressive and therefore could not be detected at birth via newborn hearing screening tests [17, 18]. In our series, 1 patient developed unilateral SNHL. This patient, who is currently 2 years old, is undergoing regular follow-up evaluation. Ocular involvement with chorioretinitis is common in patients with CMV infection, and retinal scars, optic atrophy, and central vision loss have also been described [1, 4, 15]. In this study, however, no patients had ocular involvement.
We speculate that the CMV infections in our patients were acquired as congenital infections during late pregnancy or as perinatal infections. First, the patients did not have clinical signs of infection during early pregnancy, such as intrauterine growth retardation or microcephaly; however, they had signs of late infection, such as hepatitis, petechiae, and thrombocytopenia. Second, IgM persists for a long period, and 4 of 6 patients in our series were positive for CMV PCR and CMV pp65 Ag, which indicated recently acquired infection [8, 15, 19]. CMV infection during late pregnancy has a higher rate of neonatal infection but a lower rate of symptomatic manifestation than that during early pregnancy [20]. The transplacental infection rate during the first and second trimesters is around 45%, and the rate increases to 58-78% during the third trimester. Since neuronal growth and migration is completed before 26 weeks of gestation, transplacental infection during the third trimester is less frequently associated with symptomatic neurodevelopmental manifestations as compared to earlier infections [20, 21]. If the CMV infections in our series of patients were congenital infections, recurrent infection would have been more likely than primary infection, since all the patients were asymptomatic at birth and positive for CMV IgG but had no symptoms at admission other than those related to thrombocytopenia [22].
In conclusion, this study showed that thrombocytopenia can be the main clinical manifestation of otherwise asymptomatic CMV infection after the neonatal period. CMV infection needs to be considered in infants between 1 and 6 months old with thrombocytopenia, and close follow-up is needed to detect neurodevelopmental sequelae.
References
- 1.Aslam M, Anderson JL, Guglietti D, Cardwell D. CMV-induced neonatal thrombocytopenia: a case report and review of the literature. Am J Perinatol. 2007;24:429–434. doi: 10.1055/s-2007-984409. [DOI] [PubMed] [Google Scholar]
- 2.Romanelli RM, Magny JF, Jacquemard F. Prognostic markers of symptomatic congenital cytomegalovirus infection. Braz J Infect Dis. 2008;12:38–43. doi: 10.1590/s1413-86702008000100009. [DOI] [PubMed] [Google Scholar]
- 3.Benoist G, Salomon LJ, Jacquemard F, Daffos F, Ville Y. The prognostic value of ultrasound abnormalities and biological parameters in blood of fetuses infected with cytomegalovirus. BJOG. 2008;115:823–829. doi: 10.1111/j.1471-0528.2008.01714.x. [DOI] [PubMed] [Google Scholar]
- 4.Malm G, Engman ML. Congenital cytomegalovirus infections. Semin Fetal Neonatal Med. 2007;12:154–159. doi: 10.1016/j.siny.2007.01.012. [DOI] [PubMed] [Google Scholar]
- 5.Ornoy A, Diav-Citrin O. Fetal effects of primary and secondary cytomegalovirus infection in pregnancy. Reprod Toxicol. 2006;21:399–409. doi: 10.1016/j.reprotox.2005.02.002. [DOI] [PubMed] [Google Scholar]
- 6.Rivera LB, Boppana SB, Fowler KB, Britt WJ, Stagno S, Pass RF. Predictors of hearing loss in children with symptomatic congenital cytomegalovirus infection. Pediatrics. 2002;110:762–767. doi: 10.1542/peds.110.4.762. [DOI] [PubMed] [Google Scholar]
- 7.Walker A, Petheram SJ, Ballard L, Murph JR, Demmler GJ, Bale JF., Jr Characterization of human cytomegalovirus strains by analysis of short tandem repeat polymorphisms. J Clin Microbiol. 2001;39:2219–2226. doi: 10.1128/JCM.39.6.2219-2226.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lazzarotto T, Guerra B, Lanari M, Gabrielli L, Landini MP. New advances in the diagnosis of congenital cytomegalovirus infection. J Clin Virol. 2008;41:192–197. doi: 10.1016/j.jcv.2007.10.015. [DOI] [PubMed] [Google Scholar]
- 9.Ahlfors K, Ivarsson SA, Harris S. Report on a long-term study of maternal and congenital cytomegalovirus infection in Sweden. Review of prospective studies available in the literature. Scand J Infect Dis. 1999;31:443–457. doi: 10.1080/00365549950163969. [DOI] [PubMed] [Google Scholar]
- 10.Revello MG, Zavattoni M, Baldanti F, Sarasini A, Paolucci S, Gerna G. Diagnostic and prognostic value of human cytomegalovirus load and IgM antibody in blood of congenitally infected newborns. J Clin Virol. 1999;14:57–66. doi: 10.1016/s1386-6532(99)00016-5. [DOI] [PubMed] [Google Scholar]
- 11.Levy AS, Bussel J. Immune thrombocytopenic purpura: investigation of the role of cytomegalovirus infection. Br J Haematol. 2004;126:622–623. doi: 10.1111/j.1365-2141.2004.05068.x. [DOI] [PubMed] [Google Scholar]
- 12.Crapnell K, Zanjani ED, Chaudhuri A, Ascensao JL, St Jeor S, Maciejewski JP. In vitro infection of megakaryocytes and their precursors by human cytomegalovirus. Blood. 2000;95:487–493. [PubMed] [Google Scholar]
- 13.Toyoda M, Petrosian A, Jordan SC. Immunological characterization of anti-endothelial cell antibodies induced by cytomegalovirus infection. Transplantation. 1999;68:1311–1318. doi: 10.1097/00007890-199911150-00016. [DOI] [PubMed] [Google Scholar]
- 14.Yenicesu I, Yetgin S, Ozyürek E, Aslan D. Virus-associated immune thrombocytopenic purpura in childhood. Pediatr Hematol Oncol. 2002;19:433–437. doi: 10.1080/08880010290097233. [DOI] [PubMed] [Google Scholar]
- 15.Kylat RI, Kelly EN, Ford-Jones EL. Clinical findings and adverse outcome in neonates with symptomatic congenital cytomegalovirus (SCCMV) infection. Eur J Pediatr. 2006;165:773–778. doi: 10.1007/s00431-006-0172-6. [DOI] [PubMed] [Google Scholar]
- 16.Dollard SC, Grosse SD, Ross DS. New estimates of the prevalence of neurological and sensory sequelae and mortality associated with congenital cytomegalovirus infection. Rev Med Virol. 2007;17:355–363. doi: 10.1002/rmv.544. [DOI] [PubMed] [Google Scholar]
- 17.Grosse SD, Ross DS, Dollard SC. Congenital cytomegalovirus (CMV) infection as a cause of permanent bilateral hearing loss: a quantitative assessment. J Clin Virol. 2008;41:57–62. doi: 10.1016/j.jcv.2007.09.004. [DOI] [PubMed] [Google Scholar]
- 18.Fowler KB, Dahle AJ, Boppana SB, Pass RF. Newborn hearing screening: will children with hearing loss caused by congenital cytomegalovirus infection be missed. J Pediatr. 1999;135:60–64. doi: 10.1016/s0022-3476(99)70328-8. [DOI] [PubMed] [Google Scholar]
- 19.Fowler KB, Stagno S, Pass RF. Maternal age and congenital cytomegalovirus infection: screening of two diverse newborn populations, 1980-1990. J Infect Dis. 1993;168:552–556. doi: 10.1093/infdis/168.3.552. [DOI] [PubMed] [Google Scholar]
- 20.Revello MG, Gerna G. Pathogenesis and prenatal diagnosis of human cytomegalovirus infection. J Clin Virol. 2004;29:71–83. doi: 10.1016/j.jcv.2003.09.012. [DOI] [PubMed] [Google Scholar]
- 21.Gindes L, Teperberg-Oikawa M, Sherman D, Pardo J, Rahav G. Congenital cytomegalovirus infection following primary maternal infection in the third trimester. BJOG. 2008;115:830–835. doi: 10.1111/j.1471-0528.2007.01651.x. [DOI] [PubMed] [Google Scholar]
- 22.Kenneson A, Cannon MJ. Review and meta-analysis of the epidemiology of congenital cytomegalovirus (CMV) infection. Rev Med Virol. 2007;17:253–276. doi: 10.1002/rmv.535. [DOI] [PubMed] [Google Scholar]