Table 2.
Asymmetric alcoholysis of cyclic anhydrides 1 with QD-MN and (DHQD)2AQN* 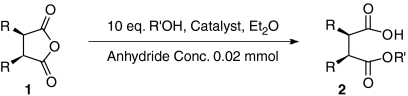
| Entry | Substrate | Catalyst (mol%) | Alcohol (10 eq.) | Temp (°C) | Time | Yield † | ee ‡ |
| 1 | QD-MN (10%) | CF3CH2OH | RT | 5 h | 91% | 94% | |
| 2 | (DHQD)2AQN (5%) | CF3CH2OH | RT | 9 h | 90% | 90% | |
| 3 | QD-MN (10%) | MeOH | RT | 9 h | 90% | 90% | |
| 4 | (DHQD)2 AQN (5%) | MeOH | RT | 5 h | 89% | 94% | |
| 5 | 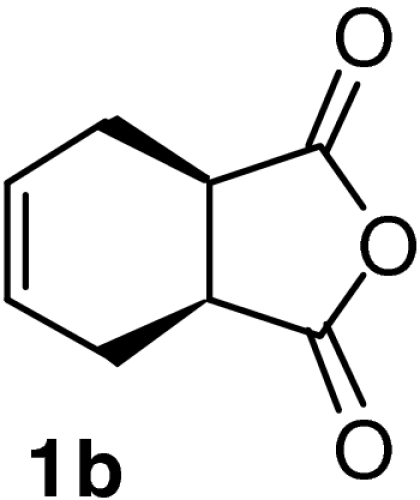 |
QD-MN (14%) | CF3CH2OH | RT | 6 h | 93% | 95% |
| 6 | (DHQD)2AQN (7%) | CF3CH2OH | RT | 5 h | 96% | 91% | |
| 7 | QD-MN (14%) | MeOH | RT | 8 h | 91% | 90% | |
| 8 | (DHQD)2AQN (7%) | MeOH | RT | 6 h | 92% | 94% | |
| 9 | 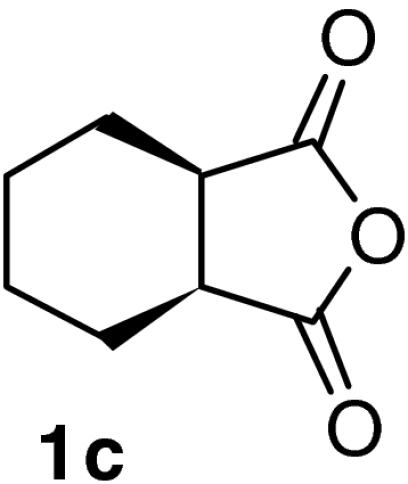 |
QD-MN (10%) | CF3CH2OH2 | RT | 7 h | 96% | 94% |
| 10 | (DHQD)2AQN (5%) | CF3CH2OH | RT | 7 h | 95% | 89% | |
| 11 | QD-MN (10%) | MeOH | RT | 8 h | 94% | 87% | |
| 12 | (DHQD)2AQN (5%) | MeOH | RT | 4 h | 95% | 93% | |
| 13 | 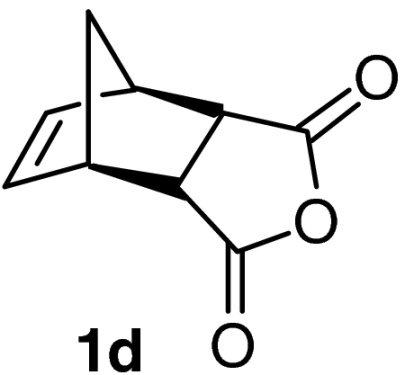 |
QD-MN (20%) | MeOH | RT | 36 h | 88% | 90% |
| 14 | (DHQD)2AQN (10%) | MeOH | RT | 12 h | 90% | 88% | |
| 15 | 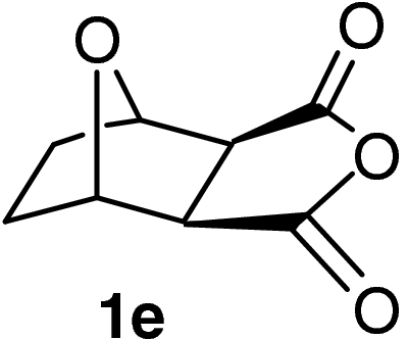 |
QD-MN (30%) | MeOH | −20 | 60 h | 91% | 95% |
| 16 | (DHQD)2AQN (15%) | MeOH | −20 | 120 h | 88% | 96% | |
| 17 | 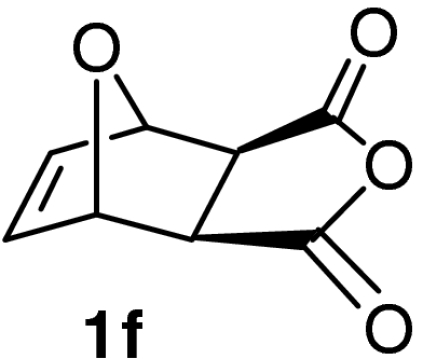 |
QD-MN (40%) | MeOH | −20 | 48 h | 87% | 92% |
| 18 | (DHQD)2AQN (20%) | MeOH | −20 | 96 h | 74% | 92% |
*The reaction was carried out with 1 (0.02 M in diethyl ether) and R’OH [10 equivalent (eq.)] in the presence of catalyst.
†Conversion was determined by 1H NMR.
‡See the SI Appendix for the determination of the ee value.
