Abstract
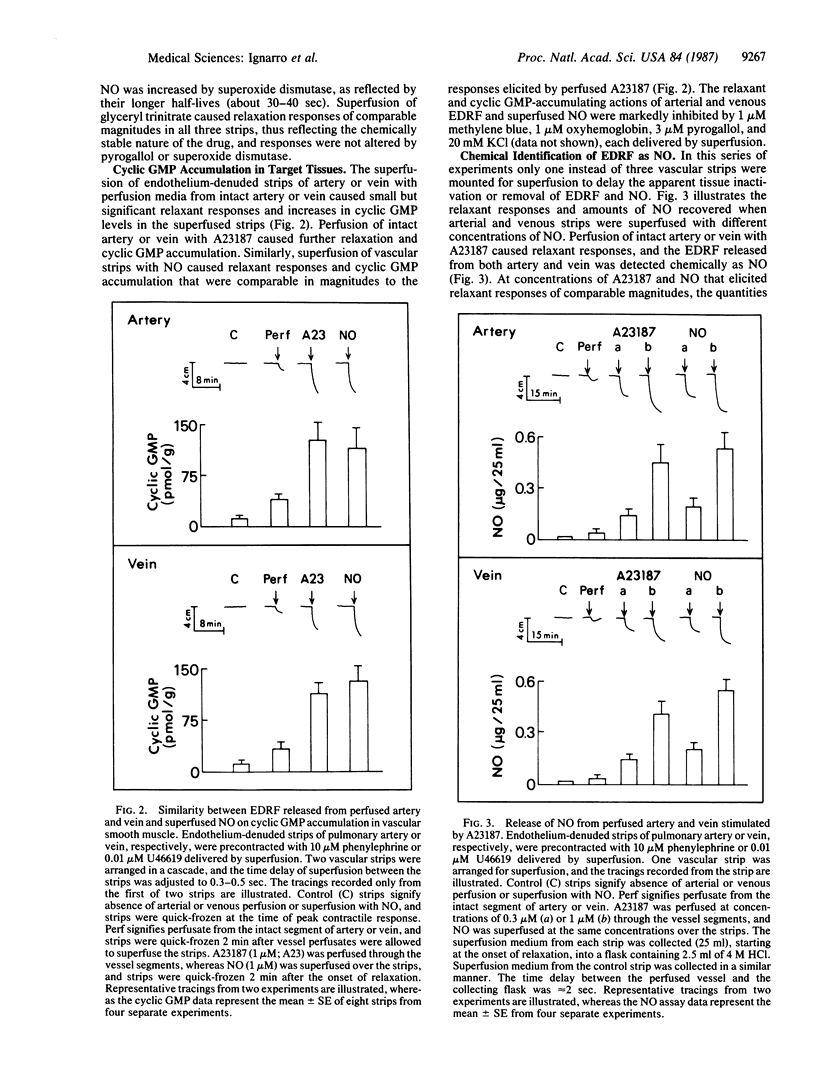
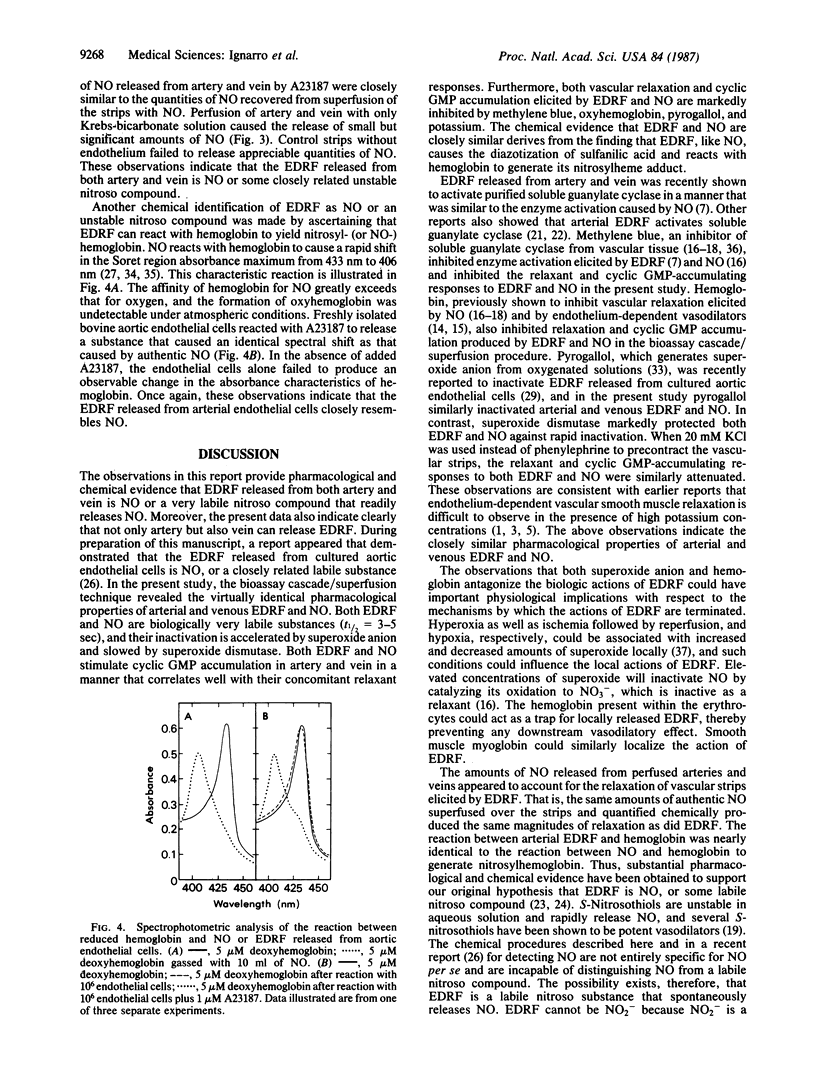
The objective of this study was to determine whether nitric oxide (NO) is responsible for the vascular smooth muscle relaxation elicited by endothelium-derived relaxing factor (EDRF). EDRF is an unstable humoral substance released from artery and vein that mediates the action of endothelium-dependent vasodilators. NO is an unstable endothelium-independent vasodilator that is released from vasodilator drugs such as nitroprusside and glyceryl trinitrate. We have repeatedly observed that the actions of NO on vascular smooth muscle closely resemble those of EDRF. In the present study the vascular effects of EDRF released from perfused bovine intrapulmonary artery and vein were compared with the effects of NO delivered by superfusion over endothelium-denuded arterial and venous strips arranged in a cascade. EDRF was indistinguishable from NO in that both were labile (t1/2 = 3-5 sec), inactivated by pyrogallol or superoxide anion, stabilized by superoxide dismutase, and inhibited by oxyhemoglobin or potassium. Both EDRF and NO produced comparable increases in cyclic GMP accumulation in artery and vein, and this cyclic GMP accumulation was inhibited by pyrogallol, oxyhemoglobin, potassium, and methylene blue. EDRF was identified chemically as NO, or a labile nitroso species, by two procedures. First, like NO, EDRF released from freshly isolated aortic endothelial cells reacted with hemoglobin to yield nitrosylhemoglobin. Second, EDRF and NO each similarly promoted the diazotization of sulfanilic acid and yielded the same reaction product after coupling with N-(1-naphthyl)-ethylenediamine. Thus, EDRF released from artery and vein possesses identical biological and chemical properties as NO.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BELL F. K., O'NEILL J. J., BURGISON R. M. Determination of the oil/water distribution coefficients of glyceryl trinitrate and two similar nitrate esters. J Pharm Sci. 1963 Jul;52:637–639. doi: 10.1002/jps.2600520706. [DOI] [PubMed] [Google Scholar]
- Beliner J. A. Regulation of endothelial cell DNA synthesis and adherence. In Vitro. 1981 Nov;17(11):985–992. doi: 10.1007/BF02618423. [DOI] [PubMed] [Google Scholar]
- Burton K. P., McCord J. M., Ghai G. Myocardial alterations due to free-radical generation. Am J Physiol. 1984 Jun;246(6 Pt 2):H776–H783. doi: 10.1152/ajpheart.1984.246.6.H776. [DOI] [PubMed] [Google Scholar]
- Chand N., Altura B. M. Acetylcholine and bradykinin relax intrapulmonary arteries by acting on endothelial cells: role in lung vascular diseases. Science. 1981 Sep 18;213(4514):1376–1379. doi: 10.1126/science.7268440. [DOI] [PubMed] [Google Scholar]
- Craven P. A., DeRubertis F. R. Requirement for heme in the activation of purified guanylate cyclase by nitric oxide. Biochim Biophys Acta. 1983 Jun 29;745(3):310–321. doi: 10.1016/0167-4838(83)90063-8. [DOI] [PubMed] [Google Scholar]
- De Mey J. G., Claeys M., Vanhoutte P. M. Endothelium-dependent inhibitory effects of acetylcholine, adenosine triphosphate, thrombin and arachidonic acid in the canine femoral artery. J Pharmacol Exp Ther. 1982 Jul;222(1):166–173. [PubMed] [Google Scholar]
- Diamond J., Chu E. B. Possible role for cyclic GMP in endothelium-dependent relaxation of rabbit aorta by acetylcholine. Comparison with nitroglycerin. Res Commun Chem Pathol Pharmacol. 1983 Sep;41(3):369–381. [PubMed] [Google Scholar]
- Edwards J. C., Ignarro L. J., Hyman A. L., Kadowitz P. J. Relaxation of intrapulmonary artery and vein by nitrogen oxide-containing vasodilators and cyclic GMP. J Pharmacol Exp Ther. 1984 Jan;228(1):33–42. [PubMed] [Google Scholar]
- Furchgott R. F. Role of endothelium in responses of vascular smooth muscle. Circ Res. 1983 Nov;53(5):557–573. doi: 10.1161/01.res.53.5.557. [DOI] [PubMed] [Google Scholar]
- Furchgott R. F., Zawadzki J. V. The obligatory role of endothelial cells in the relaxation of arterial smooth muscle by acetylcholine. Nature. 1980 Nov 27;288(5789):373–376. doi: 10.1038/288373a0. [DOI] [PubMed] [Google Scholar]
- Förstermann U., Mülsch A., Böhme E., Busse R. Stimulation of soluble guanylate cyclase by an acetylcholine-induced endothelium-derived factor from rabbit and canine arteries. Circ Res. 1986 Apr;58(4):531–538. doi: 10.1161/01.res.58.4.531. [DOI] [PubMed] [Google Scholar]
- Griffith T. M., Edwards D. H., Lewis M. J., Newby A. C., Henderson A. H. The nature of endothelium-derived vascular relaxant factor. Nature. 1984 Apr 12;308(5960):645–647. doi: 10.1038/308645a0. [DOI] [PubMed] [Google Scholar]
- Gruetter C. A., Barry B. K., McNamara D. B., Gruetter D. Y., Kadowitz P. J., Ignarro L. Relaxation of bovine coronary artery and activation of coronary arterial guanylate cyclase by nitric oxide, nitroprusside and a carcinogenic nitrosoamine. J Cyclic Nucleotide Res. 1979;5(3):211–224. [PubMed] [Google Scholar]
- Gruetter C. A., Barry B. K., McNamara D. B., Kadowitz P. J., Ignarro L. J. Coronary arterial relaxation and guanylate cyclase activation by cigarette smoke, N'-nitrosonornicotine and nitric oxide. J Pharmacol Exp Ther. 1980 Jul;214(1):9–15. [PubMed] [Google Scholar]
- Gruetter C. A., Gruetter D. Y., Lyon J. E., Kadowitz P. J., Ignarro L. J. Relationship between cyclic guanosine 3':5'-monophosphate formation and relaxation of coronary arterial smooth muscle by glyceryl trinitrate, nitroprusside, nitrite and nitric oxide: effects of methylene blue and methemoglobin. J Pharmacol Exp Ther. 1981 Oct;219(1):181–186. [PubMed] [Google Scholar]
- Gruetter C. A., Lemke S. M. Comparison of endothelium-dependent relaxation in bovine intrapulmonary artery and vein by acetylcholine and A23187. J Pharmacol Exp Ther. 1986 Sep;238(3):1055–1062. [PubMed] [Google Scholar]
- Gryglewski R. J., Moncada S., Palmer R. M. Bioassay of prostacyclin and endothelium-derived relaxing factor (EDRF) from porcine aortic endothelial cells. Br J Pharmacol. 1986 Apr;87(4):685–694. doi: 10.1111/j.1476-5381.1986.tb14586.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holzmann S. Endothelium-induced relaxation by acetylcholine associated with larger rises in cyclic GMP in coronary arterial strips. J Cyclic Nucleotide Res. 1982;8(6):409–419. [PubMed] [Google Scholar]
- Ignarro L. J., Adams J. B., Horwitz P. M., Wood K. S. Activation of soluble guanylate cyclase by NO-hemoproteins involves NO-heme exchange. Comparison of heme-containing and heme-deficient enzyme forms. J Biol Chem. 1986 Apr 15;261(11):4997–5002. [PubMed] [Google Scholar]
- Ignarro L. J., Burke T. M., Wood K. S., Wolin M. S., Kadowitz P. J. Association between cyclic GMP accumulation and acetylcholine-elicited relaxation of bovine intrapulmonary artery. J Pharmacol Exp Ther. 1984 Mar;228(3):682–690. [PubMed] [Google Scholar]
- Ignarro L. J., Degnan J. N., Baricos W. H., Kadowitz P. J., Wolin M. S. Activation of purified guanylate cyclase by nitric oxide requires heme. Comparison of heme-deficient, heme-reconstituted and heme-containing forms of soluble enzyme from bovine lung. Biochim Biophys Acta. 1982 Sep 17;718(1):49–59. doi: 10.1016/0304-4165(82)90008-3. [DOI] [PubMed] [Google Scholar]
- Ignarro L. J., Harbison R. G., Wood K. S., Kadowitz P. J. Activation of purified soluble guanylate cyclase by endothelium-derived relaxing factor from intrapulmonary artery and vein: stimulation by acetylcholine, bradykinin and arachidonic acid. J Pharmacol Exp Ther. 1986 Jun;237(3):893–900. [PubMed] [Google Scholar]
- Ignarro L. J., Harbison R. G., Wood K. S., Wolin M. S., McNamara D. B., Hyman A. L., Kadowitz P. J. Differences in responsiveness of intrapulmonary artery and vein to arachidonic acid: mechanism of arterial relaxation involves cyclic guanosine 3':5'-monophosphate and cyclic adenosine 3':5'-monophosphate. J Pharmacol Exp Ther. 1985 Jun;233(3):560–569. [PubMed] [Google Scholar]
- Ignarro L. J., Lippton H., Edwards J. C., Baricos W. H., Hyman A. L., Kadowitz P. J., Gruetter C. A. Mechanism of vascular smooth muscle relaxation by organic nitrates, nitrites, nitroprusside and nitric oxide: evidence for the involvement of S-nitrosothiols as active intermediates. J Pharmacol Exp Ther. 1981 Sep;218(3):739–749. [PubMed] [Google Scholar]
- Ignarro L. J., Wood K. S., Ballot B., Wolin M. S. Guanylate cyclase from bovine lung. Evidence that enzyme activation by phenylhydrazine is mediated by iron-phenyl hemoprotein complexes. J Biol Chem. 1984 May 10;259(9):5923–5931. [PubMed] [Google Scholar]
- Marklund S., Marklund G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem. 1974 Sep 16;47(3):469–474. doi: 10.1111/j.1432-1033.1974.tb03714.x. [DOI] [PubMed] [Google Scholar]
- Martin W., Villani G. M., Jothianandan D., Furchgott R. F. Blockade of endothelium-dependent and glyceryl trinitrate-induced relaxation of rabbit aorta by certain ferrous hemoproteins. J Pharmacol Exp Ther. 1985 Jun;233(3):679–685. [PubMed] [Google Scholar]
- Martin W., Villani G. M., Jothianandan D., Furchgott R. F. Selective blockade of endothelium-dependent and glyceryl trinitrate-induced relaxation by hemoglobin and by methylene blue in the rabbit aorta. J Pharmacol Exp Ther. 1985 Mar;232(3):708–716. [PubMed] [Google Scholar]
- Mülsch A., Böhme E., Busse R. Stimulation of soluble guanylate cyclase by endothelium-derived relaxing factor from cultured endothelial cells. Eur J Pharmacol. 1987 Mar 17;135(2):247–250. doi: 10.1016/0014-2999(87)90620-0. [DOI] [PubMed] [Google Scholar]
- Ohlstein E. H., Wood K. S., Ignarro L. J. Purification and properties of heme-deficient hepatic soluble guanylate cyclase: effects of heme and other factors on enzyme activation by NO, NO-heme, and protoporphyrin IX. Arch Biochem Biophys. 1982 Oct 1;218(1):187–198. doi: 10.1016/0003-9861(82)90335-6. [DOI] [PubMed] [Google Scholar]
- Palmer R. M., Ferrige A. G., Moncada S. Nitric oxide release accounts for the biological activity of endothelium-derived relaxing factor. Nature. 1987 Jun 11;327(6122):524–526. doi: 10.1038/327524a0. [DOI] [PubMed] [Google Scholar]
- Rapoport R. M., Murad F. Agonist-induced endothelium-dependent relaxation in rat thoracic aorta may be mediated through cGMP. Circ Res. 1983 Mar;52(3):352–357. doi: 10.1161/01.res.52.3.352. [DOI] [PubMed] [Google Scholar]
- Schwartz S. M. Selection and characterization of bovine aortic endothelial cells. In Vitro. 1978 Dec;14(12):966–980. doi: 10.1007/BF02616210. [DOI] [PubMed] [Google Scholar]
- VANE J. R. THE USE OF ISOLATED ORGANS FOR DETECTING ACTIVE SUBSTANCES IN THE CIRCULATING BLOOD. Br J Pharmacol Chemother. 1964 Oct;23:360–373. doi: 10.1111/j.1476-5381.1964.tb01592.x. [DOI] [PMC free article] [PubMed] [Google Scholar]


